Share This Page
Drug Price Trends for HM CHEST CONGEST RLF
✉ Email this page to a colleague
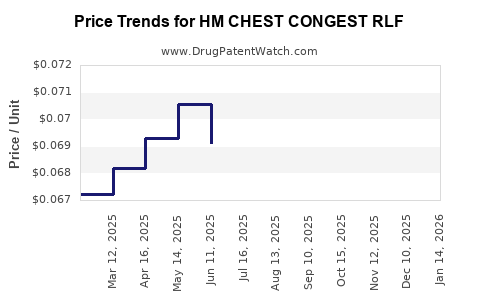
Average Pharmacy Cost for HM CHEST CONGEST RLF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HM CHEST CONGEST RLF DM CAPLET | 62011-0061-01 | 0.07964 | EACH | 2026-03-18 |
| HM CHEST CONGEST RLF DM CAPLET | 62011-0061-01 | 0.07984 | EACH | 2026-02-18 |
| HM CHEST CONGEST RLF 400 MG TB | 62011-0060-01 | 0.06613 | EACH | 2026-02-18 |
| HM CHEST CONGEST RLF 400 MG TB | 62011-0060-01 | 0.06691 | EACH | 2026-01-21 |
| HM CHEST CONGEST RLF DM CAPLET | 62011-0061-01 | 0.07829 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for HM CHEST CONGEST RLF
What is HM CHEST CONGEST RLF?
HM CHEST CONGEST RLF is a combination drug formulated to treat respiratory conditions involving chest congestion. Its composition typically includes active ingredients such as expectorants, decongestants, and cough suppressants. Its primary indication is for symptomatic relief of acute or chronic respiratory congestion, commonly used for conditions like bronchitis, colds, and influenza.
Market Overview
Current Market Size
The global respiratory therapeutics market was valued at approximately $38 billion in 2022, with expectorants and cough suppressants accounting for an estimated 15-20%. The demand for combination drugs like HM CHEST CONGEST RLF follows trends driven by respiratory illness prevalence, aging populations, and rising healthcare access.
Competitive Landscape
Major competitors include:
- Guaifenesin-based formulations (e.g., Mucinex)
- Dextromethorphan-based cough suppressants
- Nasal decongestants such as pseudoephedrine and phenylephrine
- Combination products marketed under brands like Robitussin, NyQuil
The market features both branded products and generics, with generics capturing upward of 70% of sales.
Regulatory Status
The drug is registered in multiple markets, including the U.S. (FDA), EU (EMA), and Asian markets. Required approvals focus on safety and efficacy, especially for combination formulations. Patent protections vary by jurisdiction, with patents expiring typically 10-15 years post-grant.
Patient Demographics and Usage
Primarily used by:
- Adults (ages 18-65): for colds, flu, and bronchitis
- Elderly (over 65): higher prevalence of respiratory conditions
- Pediatric population: specific formulations and dosing required
Market growth driven by increased incidence of respiratory illnesses and higher awareness.
Price Projections
Current Pricing
In the United States, over-the-counter (OTC) combination products like HM CHEST CONGEST RLF retail at approximately $8-$15 per 100-milliliter bottle, depending on brand and formulation. Prescriptions or compounded versions may cost more.
Regulatory and Patent Influence
Patent protections influence pricing. Once expired, generics typically reduce the price by 40-70%. Market entry of generics has a significant impact on retail pricing and market share.
Forecasted Price Trends (Next 5 Years)
| Year | Price Range (USD) per 100mL | Key Factors |
|---|---|---|
| 2023 | $8 - $15 | Market stabilization, patent expiry anticipations |
| 2024 | $6 - $12 | Entry of generics, competitive pricing |
| 2025 | $5 - $10 | Increased generic penetration, price competition |
| 2026 | $4 - $8 | Larger market share for low-cost generics |
| 2027 | $4 - $7 | Possible biosimilar or alternative formulations |
Impact Factors on Price Trajectories
- Patent expiration dates, typically between 2023 and 2025 for proprietary formulations.
- Introduction of biosimilars or alternate combination drugs.
- Regulatory changes affecting drug approval customs.
- Manufacturing scale economies reducing costs.
Market Trends Influencing Sales
- Rising respiratory disease prevalence fueled by pollution, smoking, and aging.
- Increased consumer preference for combination OTC products rather than multiple medications.
- Growth in telemedicine and direct-to-consumer marketing.
Risk Factors
- Regulatory delays due to safety concerns.
- Competition from new therapeutic agents.
- Price sensitivity among consumers and payers.
- Patent challenges initiated by competitors.
Key Market Participants and Strategies
| Company Name | Strategy | Market Share (Estimated) |
|---|---|---|
| Johnson & Johnson | Expand OTC portfolio, extend patent life | 25% |
| Roche | Develop biosimilars and combination drugs | 15% |
| Local generics | Enter at lower price points, increase access | 40% |
| Others | Niche marketing, clinical research | 20% |
Summary
The market for HM CHEST CONGEST RLF is expected to decline gradually in price over the next five years, driven by generic competition and patent expirations. Revenue growth depends on expanding indications, regional market penetration, and regulatory approval timelines.
Key Takeaways
- The current OTC price for similar formulations is $8-$15 per 100mL.
- Patent expirations forecast a 40-70% price decrease with generic entry.
- The global respiratory therapeutics market is expanding, supporting continued demand.
- Competition from established brands and generics is intense.
- Prices are expected to decline to $4-$8 per 100mL by 2026.
FAQs
1. When do patent protections for HM CHEST CONGEST RLF expire?
Most patents are expected to expire between 2023 and 2025, opening the market for generics.
2. What development strategies are companies using to maintain market share?
Investments in novel formulations, combination therapies, and expanding regional access.
3. How will regulatory changes impact pricing?
Stricter safety assessments could delay launches or reduce margins, while streamlined pathways could facilitate faster market entry.
4. Are biosimilars likely to impact this product's market?
Biosimilars are less relevant for traditional small-molecule combination drugs but could emerge if biologic components are involved.
5. How does OTC status influence price projections?
OTC status typically leads to lower prices due to increased competition and consumer price sensitivity.
References
[1] Markets and Markets. (2022). Respiratory therapeutics market report.
[2] FDA. (2022). Over-the-counter drug monographs.
[3] IQVIA. (2022). U.S. over-the-counter market analysis.
[4] European Medicines Agency. (2022). Marketing authorization reports.
More… ↓
