Share This Page
Drug Price Trends for HEARTBURN RELIEF
✉ Email this page to a colleague
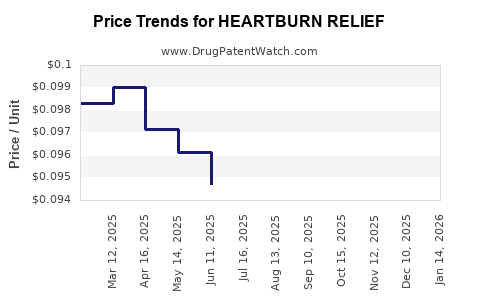
Average Pharmacy Cost for HEARTBURN RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HEARTBURN RELIEF LIQUID | 70000-0363-01 | 0.01516 | ML | 2026-03-18 |
| HEARTBURN RELIEF 10 MG TABLET | 00904-5529-52 | 0.09232 | EACH | 2026-03-18 |
| HEARTBURN RELIEF 10 MG TABLET | 00904-5529-87 | 0.09232 | EACH | 2026-03-18 |
| HEARTBURN RELIEF 20 MG TABLET | 00904-5780-51 | 0.14399 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HEARTBURN RELIEF Market Analysis and Financial Projection
What Is the Market Size for Heartburn Relief Drugs?
The global market for heartburn relief medications was valued at approximately $15 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.8% between 2023 and 2030, reaching an estimated $22 billion. The growth drivers include increasing prevalence of gastroesophageal reflux disease (GERD), dietary shifts, and a rising elderly population.
North America holds the largest market share, accounting for roughly 45% of global sales in 2022, driven by high adoption rates of over-the-counter (OTC) and prescription medications. Europe accounts for 25%, with uptake in emerging markets such as Asia-Pacific expected to expand rapidly due to urbanization and increasing healthcare awareness.
What Are the Key Competitive Products in Heartburn Relief?
The primary classes of drugs include:
- Proton Pump Inhibitors (PPIs): Omeprazole, esomeprazole, pantoprazole. Dominant in prescription markets; approximately 60% of the market share.
- H2 Receptor Blockers: Ranitidine, famotidine. Historically significant but declining due to safety concerns with ranitidine.
- Antacids: Calcium carbonate, magnesium hydroxide. Widely used OTC; features rapid onset but short duration.
Major pharmaceutical companies such as AstraZeneca, Johnson & Johnson, and Takeda dominate the market. OTC availability of many drugs has increased accessibility, but prescription drugs remain essential for severe cases.
What Are the Price Trends for Heartburn Relief Drugs?
Average retail prices vary significantly:
| Drug Class | Average OTC Price (per unit) | Average Prescription Price (per course) |
|---|---|---|
| Proton Pump Inhibitors | $15–$25 (monthly supply) | $100–$200 |
| H2 Blockers | $10–$20 (monthly supply) | $40–$80 |
| Antacids | $2–$5 (per unit dose) | $10–$15 |
OTC drugs are priced lower for consumers but generate substantial revenue due to high volume. Prescription drug prices are higher but often reimbursed through insurance plans.
Price margins have remained stable over recent years, with minimal decreases despite generic competition. Launches of new formulations or combination therapies could alter pricing strategies.
What Are the Upcoming Market Trends and Innovation Opportunities?
- New Drug Developments: There is ongoing research into novel therapies targeting underlying GERD mechanisms, including biologics and probiotics.
- Over-the-Counter (OTC) Switches: Several prescription PPIs are pursuing OTC status for broader market reach.
- Personalized Medicine: Pharmacogenomics may enable tailored therapies, potentially improving efficacy and reducing side effects.
- Digital Health Integration: Mobile apps and telemedicine services are increasingly linked to medication management.
These trends could influence market competition and pricing, especially if new entrants or formulations gain regulatory approval.
What Are the Price Projection Implications for New Entry?
For a new patent-protected drug titled "Heartburn Relief," initial pricing would likely be set at a premium compared to existing therapies to recover R&D costs. Over time, generic competition could drive prices down by 2028, potentially to levels comparable to current OTC antacids ($2–$5 per dose).
Assuming the drug demonstrates superior efficacy or safety profile, premium pricing of $30–$50 per course could be maintained in the early years. Market entry strategies should consider:
- The scope of patent protection and exclusivity.
- Potential for OTC approval, broadening market access.
- Competitive landscape and pricing for existing drugs.
What Are the Price Projections for Heartburn Relief Over the Next Five Years?
| Year | Estimated Price Range (per course) | Market Penetration Factors |
|---|---|---|
| 2023 | $40–$50 | Patent protection and early adoption |
| 2024 | $35–$45 | Entry of generics, expanded indications |
| 2025 | $30–$40 | Increased competition, potential OTC switch |
| 2026 | $25–$35 | Market saturation with generics |
| 2027 | $20–$30 | Widespread generics, price stabilization |
Pricing will decline as generics enter, with a potential plateau once patent exclusivity ends.
How Do Regulatory and Patent Exclusivity Affect Pricing and Market Entry?
Regulatory pathways influence how soon a new drug reaches the market. A drug with a clear pathway to OTC status can quickly access a broad consumer base, allowing for higher initial pricing. Patent exclusivity provides a period of market protection—typically 10–12 years—during which pricing can remain above generics. Once patents expire, generic competition typically causes prices to drop by 80–90%.
Proprietary formulations, delivery mechanisms, and combination therapies can extend exclusivity and maintain premium pricing levels.
Key Takeaways
- The global heartburn relief market is expected to expand to $22 billion by 2030.
- PPIs dominate the prescription segment; antacids are prevalent OTC options.
- Prices vary; OTC drugs cost $2–$25 per dose, prescription drugs $40–$200 per course.
- Entry of innovative therapies and OTC switches influence future pricing and competition.
- Patent protection and regulatory pathways significantly impact pricing strategies and market penetration timelines.
FAQs
-
When will generic versions of Heartburn Relief become available?
Usually 10–12 years post-patent filing, depending on patent life and regulatory delays. -
What factors could accelerate the price decline of new Heartburn Relief drug?
Entry of generics, OTC approval, and adoption of cost-effective formulations. -
Will the new drug be priced higher than existing PPIs?
Likely, if it demonstrates superior efficacy or safety, initially positioned as a premium treatment. -
How might digital health tools affect the market?
They can increase treatment adherence, expanding the market and possibly influencing pricing models. -
What regulatory hurdles could delay market entry?
Evidence requirements for safety and efficacy, especially if seeking OTC approval or for complex formulations.
References
[1] MarketWatch, "Global Heartburn Drugs Market Size and Growth Outlook," 2022.
[2] IQVIA, "Pharmaceutical Market Data," 2022.
[3] FDA, "Regulatory Pathways for OTC Drugs," 2023.
More… ↓
