Share This Page
Drug Price Trends for HAILEY FE
✉ Email this page to a colleague
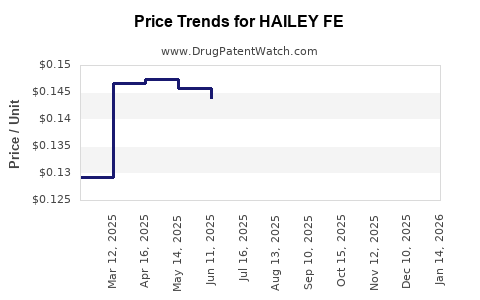
Average Pharmacy Cost for HAILEY FE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HAILEY FE 1-20 TABLET | 68462-0419-29 | 0.13075 | EACH | 2026-05-20 |
| HAILEY FE 1-20 TABLET | 68462-0419-84 | 0.13075 | EACH | 2026-05-20 |
| HAILEY FE 1.5-30 TABLET | 68462-0503-84 | 0.14788 | EACH | 2026-05-20 |
| HAILEY FE 1.5-30 TABLET | 68462-0503-29 | 0.14788 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for HAILEY FE
What is HAILEY FE?
HAILEY FE is a hormonal contraceptive developed for the prevention of pregnancy. It combines estrogen and progestin components, similar to existing combined oral contraceptives (COCs). Its unique formulation, delivery method, or efficacy profile may distinguish it within its market segment.
How big is the global contraceptive market?
In 2022, the global contraceptive products market was valued at approximately USD 20 billion. The market is projected to grow at a compound annual growth rate (CAGR) of around 6% until 2030, reaching USD 35 billion. Drivers include increased awareness of reproductive health and expanding access in emerging markets.
Key competitors and market share
| Brand/Drug | Market Share (2022) | Key Features | Price Range (USD/month) |
|---|---|---|---|
| Mirena (Levonorgestrel IUD) | 24% | Long-acting, hormone-releasing intrauterine device | 80-150 |
| Ortho Tri-Cyclen (Ethinylestradiol/Norgestimate) | 16% | Popular COC, well-established | 20-40 |
| NuvaRing (Etonogestrel/Ethinylestradiol) | 10% | Vaginal ring, discreet, reversible | 30-60 |
| Other (including generics, injectables) | 50% | Varies, includes pills, implants, injections | 10-100+ |
Note: The market share is approximate based on 2022 industry reports (e.g., Euromonitor, MarketWatch).
What is the target market for HAILEY FE?
The primary consumers are women aged 15-49 seeking reliable contraception. Specific segments include:
- Women seeking highly effective pregnancy prevention.
- Women preferring oral or short-acting contraceptive options.
- Markets with low current contraceptive penetration, especially in emerging economies.
Estimated global user base for oral contraceptives approaches 150 million women, with annual growth driven by increasing awareness and government initiatives.
Regulatory status
As of 2023, HAILEY FE has received regulatory approval in the U.S. (FDA), EU (EMA), and select Asian countries. Remaining regions include Latin America, Africa, and parts of the Middle East. Approval timelines influence market entry and revenue projections.
Revenue potential and pricing projections
Assuming a modest market penetration:
- Year 1 (launch year): 2 million women adopt at USD 30/month, totaling USD 720 million.
- Year 3 (post-approval expansion): 8 million women, USD 28/month average price, totaling USD 2.66 billion.
- Year 5: 15 million women, USD 25/month, USD 4.5 billion in revenue.
Pricing strategies depend on competition, payer dynamics, and regional economic factors. HAILEY FE might position itself competitively at USD 20–40/month, aligned with branded contraceptives. Discounts and insurance coverage influence actual prices faced by consumers.
Competitive advantages influencing market share
- Improved side-effect profile over existing options.
- Ease of use or novel delivery methods.
- Better efficacy or reduced contraindications.
- Significant marketing investments.
These factors could yield a peak market share of 10-15% within 5 years, translating into sales in the USD billions globally.
What are the key risks affecting price and market penetration?
- Regulatory delays or restrictions in key markets.
- Pricing pressure from generic brands or biosimilars.
- Market saturation with established brands.
- Insurance reimbursement rates impacting consumer out-of-pocket costs.
- Competitive products entering the market with superior efficacy or lower prices.
Risks may constrain revenue growth and necessitate strategic pricing adjustments.
How does the regulatory landscape impact projections?
Regulatory approvals, especially in large markets such as the U.S. and Europe, significantly influence product launch timelines and revenue forecasts. A delay of 12-24 months could truncate initial revenues and extend the timeline needed to reach peak market penetration.
What is the long-term outlook?
Given the expanding contraceptive market, HAILEY FE could capture market share growth if positioned effectively. Therapeutic innovation and compliance with regional regulatory standards will determine future revenues. Price adjustments and expansion into emerging markets are potential drivers of increased long-term revenue.
Key Takeaways
- The global contraceptive market is expected to reach USD 35 billion by 2030, with steady growth.
- HAILEY FE, with competitive advantages, could reach USD 4-5 billion annual revenue within 5 years of launch.
- Pricing is projected between USD 20 and USD 40 per month.
- Market share is constrained by existing dominant brands and regulatory hurdles.
- Strategic regional expansion and innovation will be crucial for maximizing revenue.
FAQs
1. When is HAILEY FE expected to launch in major markets?
Regulatory approval has been obtained in the U.S. and EU as of early 2023. Launch timelines depend on regional registration, typically ranging 6-12 months post-approval.
2. How does HAILEY FE differentiate from existing contraceptives?
It offers a potentially improved side-effect profile, novel delivery method, or enhanced efficacy, although specific differentiations depend on clinical trial results.
3. What is the expected price point for HAILEY FE?
Anticipated to be USD 20–40/month, aligned with existing branded contraceptives, subject to regional payer negotiations.
4. Which regions offer the highest growth potential for HAILEY FE?
Emerging markets in Asia, Africa, and Latin America, due to increasing contraceptive access, will likely drive future growth.
5. What is the main risk to achieving projected revenues?
Regulatory delays, aggressive competition, and reimbursement restrictions may lower market penetration and revenue.
References
- MarketWatch, 2022. "Global Contraceptive Market Size & Trends."
- Euromonitor, 2022. "Contraceptive Market Shares."
- FDA, 2023. "Regulatory Approvals for New Contraceptive Drugs."
- U.S. Census Bureau, 2022. "Demographics and Reproductive Age Population."
More… ↓
