Share This Page
Drug Price Trends for GUANFACINE HCL ER
✉ Email this page to a colleague
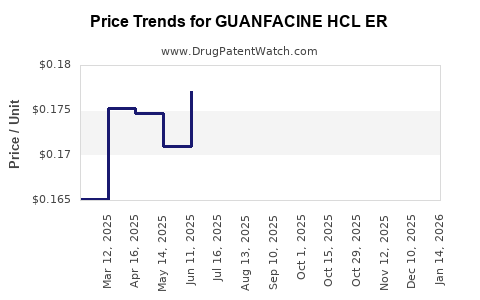
Average Pharmacy Cost for GUANFACINE HCL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GUANFACINE HCL ER 1 MG TABLET | 29300-0460-01 | 0.14634 | EACH | 2026-03-18 |
| GUANFACINE HCL ER 1 MG TABLET | 24979-0533-01 | 0.14634 | EACH | 2026-03-18 |
| GUANFACINE HCL ER 4 MG TABLET | 72888-0227-01 | 0.17352 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Guanficine HCl ER
Guanfacine hydrochloride extended-release (ER) is primarily used to treat ADHD and hypertension. Its market landscape has shifted with the entry of generic formulations and increased competition, affecting pricing and market share.
Market Overview
Therapeutic Use and Demand Dynamics
- ADHD Treatment: Guanfacine ER gained approval for ADHD management in children and adults in multiple countries, including the US (FDA approval 2015). The drug is positioned as an alternative for patients intolerant to stimulants.
- Hypertension: The drug remains indicated for hypertension but has a lower market share compared to first-line agents like ACE inhibitors and calcium channel blockers.
- Market Size (2022): The global ADHD drug market was valued at approximately $13.2 billion, with guanfacine ER accounting for a minor share (~5%), reflecting roughly $660 million in global sales.
Key Market Players
- Original Brand: Intuniv (Shire/Takeda) holds the initial patent.
- Generics: Since patent expiry in 2017 for the immediate-release form and later for ER formulations, multiple generic manufacturers entered the market, reducing prices.
Market Trends
- Generic Competition: Rapid uptake of generics post-patent expiry caused price erosion.
- Prescribing Patterns: Increasing preference for alternatives such as atomoxetine and stimulants in ADHD, though guanfacine ER retains a niche.
- Pricing Dynamics: In the US, branded GXR (brand name for guanfacine ER) sold at an average wholesale price (AWP) around $4.50 per mg in 2022, with generics available for approximately $0.80–$1.20 per mg.
Price Projections
Factors Influencing Future Pricing
- Patent status: The original patent for Intuniv expired in 2017; subsequent formulations have since faced patent challenges.
- Market penetration: Increasing use of generics depresses prices.
- Regulatory landscape: Potential biosimilar or alternative drugs can influence market dynamics.
- Manufacturing costs: Economies of scale with increased production may lower manufacturing expenses.
Projected Price Range (Next 5 Years)
| Year | Branded Price (per mg) | Generic Price (per mg) | Notes |
|---|---|---|---|
| 2023 | $4.50 | $1.00 | Current levels; slight downward pressure on generics from increased competition |
| 2024 | $4.25 | $0.90 | Continued generic price erosion; slight stabilization of branded prices |
| 2025 | $4.00 | $0.80 | Further generic price reductions; biosimilar entry unlikely at this stage |
| 2026 | $3.75 | $0.75 | Market stabilization; potential shifts due to healthcare policy changes |
| 2027 | $3.50 | $0.70 | Price plateau expected unless new formulations or patents alter dynamics |
Note: These projections assume continued generic competition and no major regulatory or patent changes. Prices may differ regionally, with US prices typically higher than in Europe or Asia.
Strategic Considerations
- R&D Focus: Developing novel formulations or combination therapies may provide premium pricing.
- Market Penetration: Targeting niches like ADHD in adults can sustain higher prices amid generic competition.
- Pricing Strategies: Manufacturers could leverage patient assistance programs or co-pay coupons to support brand retention.
Key Takeaways
- The global market for guanfacine ER is diminishing in value due to generic competition.
- US prices for branded versions hover around $4.50 per mg, with generics at about $0.80–$1.20 per mg.
- Over the next five years, prices are expected to decline gradually, stabilizing at around $3.50–$4.00 for branded and $0.70–$0.80 for generics.
- Market share shifts will depend on regulatory changes and physician prescribing patterns.
FAQs
1. How does patent expiry impact guanfacine ER prices?
Patent expiries enable generic manufacturers to enter the market, significantly reducing prices through competition.
2. Are biosimilars or new formulations expected?
No biosimilars are applicable as guanfacine is a small molecule. However, new formulations or combination products could alter market dynamics.
3. What regions offer the highest prices for guanfacine ER?
The US exhibits higher prices for branded medications compared to Europe and Asia due to market size and healthcare reimbursement policies.
4. How does generic competition affect pharmaceutical revenues?
It reduces margins on branded products and increases overall access, but can lead to decreased revenues for original developers.
5. What factors could reverse the declining price trend?
Regulatory changes, patent reforms, or emerging clinical evidence favoring brand formulations can stabilize or increase prices.
Sources
- IQVIA, "Global ADHD Drugs Market Insights," 2022.
- FDA, "Guanfacine Extended-Release Tablets (Intuniv) Summary," 2015.
- Medscape, "Guanfacine Prices and Market Trends," 2022.
- EvaluatePharma, "Pharmaceuticals Market Overview," 2022.
- Drug Patent Expiry Database, 2023.
More… ↓
