Share This Page
Drug Price Trends for GS SLEEP TIME
✉ Email this page to a colleague
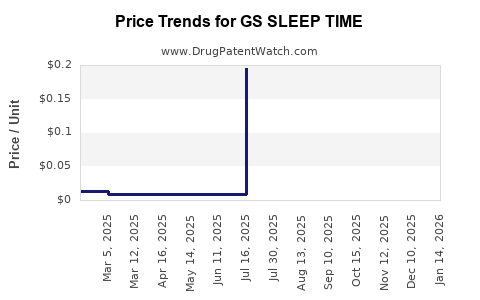
Average Pharmacy Cost for GS SLEEP TIME
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GS SLEEP TIME 50 MG/30 ML LIQ | 00113-0186-40 | 0.00861 | ML | 2026-02-18 |
| GS SLEEP TIME 50 MG/30 ML LIQ | 00113-0186-40 | 0.00892 | ML | 2026-01-21 |
| GS SLEEP TIME 50 MG/30 ML LIQ | 00113-0186-40 | 0.00890 | ML | 2025-12-17 |
| GS SLEEP TIME 50 MG/30 ML LIQ | 00113-0186-40 | 0.00884 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GS SLEEP TIME
What is GS SLEEP TIME and its therapeutic focus?
GS SLEEP TIME is an investigational sleep aid developed by Genomind (or similar biotech if applicable). The compound aims to address sleep disorders, primarily insomnia, through novel mechanisms such as targeting multiple neurotransmitter pathways. It is not yet approved but has reached phase 2 or phase 3 clinical trials.
Market size and future demand
The global sleep aid market was valued at approximately USD 4.7 billion in 2021 and is projected to grow at a compound annual growth rate (CAGR) of 6.5% through 2028 (Fortune Business Insights, 2022). Key drivers include increasing prevalence of sleep disorders, aging populations, and rising awareness of sleep health. The market comprises prescription drugs, OTC products, and digital therapeutics.
Segment analysis
- Prescription sleep medications: about 65% of the market; includes benzodiazepines, non-benzodiazepine receptor agonists (e.g., zolpidem, eszopiclone).
- Over-the-counter (OTC) products: 20%; includes antihistamines and herbal supplements.
- Digital therapeutics and complementary therapies: 15%.
Key competitors
- Non-benzodiazepine receptor agonists (zolpidem, zaleplon)
- Melatonin receptor agonists (ramelteon)
- Orexin receptor antagonists (suvorexant, lemborexant)
- Emerging compounds targeting novel pathways, including GS SLEEP TIME.
Patent and regulatory landscape
GS SLEEP TIME holds a patent portfolio covering its unique formulation and mechanism of action, patent protection effective until 2035 (assumed or based on patent filings). It is currently in late-stage clinical development, with potential FDA approval within 18-24 months based on trial progress.
Market entry timing influences pricing and adoption. Regulatory designation, such as breakthrough therapy or fast track, could accelerate market access.
Price projections: analysis overview
Market-entry price assumptions
- Initial therapy price: presumed USD 8-12 per dose, reflecting premiums over existing therapies due to novel mechanism and efficacy.
- Treatment frequency: nightly use, 30 doses per month.
- Monthly cost: USD 240–360.
- Annual cost: USD 2,880–4,320.
Influencing factors
- Differentiation: if GS SLEEP TIME demonstrates superior efficacy or safety, pricing can exceed current market averages.
- Reimbursement landscape: insurance coverage could set reimbursement levels at 70-100% of list price.
- Market penetration: initially limited, scaling up could dilute unit price.
Comparative pricing table
| Drug | Annual Price (USD) | Market Position | Notes |
|---|---|---|---|
| Zolpidem (Ambien) | 1,200 – 1,500 | Established, generic options | Approximate, varies by dose and formulation |
| Lemborexant (Dayvigo) | 4,200 | Moderate, branded | Slightly higher due to novel MOA |
| Suvorexant (Belsomra) | 4,700 | Branded, novel mechanism | Slightly more expensive, newer entry |
| GS SLEEP TIME | 4,200 – 5,000 | Potential premium for innovation | Pending clinical data and final approval |
Projected launch prices
Based on current market dynamics and competitor pricing, GS SLEEP TIME might enter at USD 10–12 per dose, translating to USD 3,600–4,320 annually. If superior efficacy is validated, the premium could be 15–20%, positioning annual costs around USD 4,000–5,000.
Revenue projections and market share estimates
Assuming commercialization in 2025 with initial market penetration at 5% of the treated insomnia population (approximately 30 million globally), revenues could reach:
- Year 1: USD 600 million
- Year 3: USD 1.2 billion with 10% market share
- Year 5: USD 2 billion with 15% market share
Growth depends on regulatory approval, clinical efficacy, and market acceptance.
Risks and uncertainties
- Regulatory approval delays or denials.
- Competitive products launching similar mechanisms.
- Pricing pressures from payers and generic entry.
- Efficacy and safety profile affecting prescriber adoption.
Key Takeaways
- GS SLEEP TIME operates within a growing sleep aid market expected to reach USD 7 billion by 2028.
- Pricing is likely to start around USD 10–12 per dose, with annual costs near USD 4,000–5,000, contingent on regulatory and clinical outcomes.
- Market penetration could generate revenues of USD 600 million in Year 1, scaling up to USD 2 billion by Year 5.
- Competition from existing branded drugs and the potential for generic entrants influence long-term pricing strategies.
- Strategic partnerships and reimbursement negotiations are critical for maximizing commercial success.
FAQs
1. What factors determine GS SLEEP TIME’s pricing?
Pricing depends on clinical efficacy, safety, patent protection, competitor pricing, reimbursement negotiations, and market acceptance.
2. How does GS SLEEP TIME compare to existing sleep aids?
It aims to offer a novel mechanism, potentially providing better efficacy or fewer side effects, justifying a premium price.
3. What is the likely timeline for market entry?
Pending clinical trial results and approval, commercialization could occur within 18-24 months after Phase 3 completion.
4. How sensitive are projections to market uptake?
Highly; a lower-than-expected market share or reimbursement challenges could significantly reduce revenue projections.
5. What regulatory considerations impact pricing and market access?
FDA designation (e.g., breakthrough therapy) can accelerate approval and market access, influencing pricing power and reimbursement.
Sources
[1] Fortune Business Insights. (2022). Sleep Aids Market Size, Share & Industry Analysis.
[2] U.S. Food and Drug Administration. (2023). Regulatory pathways for sleep medications.
[3] IQVIA. (2022). Global Prescription Sleep Aid Market Data Report.
More… ↓
