Share This Page
Drug Price Trends for GS COUGH DM ER
✉ Email this page to a colleague
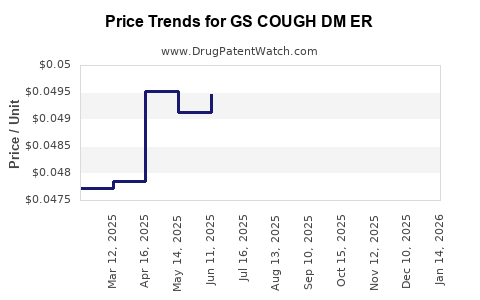
Average Pharmacy Cost for GS COUGH DM ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GS COUGH DM ER 30 MG/5 ML SUSP | 00113-0384-28 | 0.05079 | ML | 2026-02-18 |
| GS COUGH DM ER 30 MG/5 ML SUSP | 00113-0384-28 | 0.04958 | ML | 2026-01-21 |
| GS COUGH DM ER 30 MG/5 ML SUSP | 00113-0384-28 | 0.04942 | ML | 2025-12-17 |
| GS COUGH DM ER 30 MG/5 ML SUSP | 00113-0384-28 | 0.04924 | ML | 2025-11-19 |
| GS COUGH DM ER 30 MG/5 ML SUSP | 00113-0384-28 | 0.04981 | ML | 2025-10-22 |
| GS COUGH DM ER 30 MG/5 ML SUSP | 00113-0384-28 | 0.05065 | ML | 2025-09-17 |
| GS COUGH DM ER 30 MG/5 ML SUSP | 00113-0384-28 | 0.05081 | ML | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GS COUGH DM ER: Patent Landscape and Market Projections
GS COUGH DM ER (Guaifenesin/Dextromethorphan Hydrobromide Extended-Release) is a combination drug formulation indicated for the temporary relief of cough and chest congestion. This analysis details its current patent status, projected market performance, and competitive environment to inform R&D and investment strategies.
What is the Current Patent Status of GS COUGH DM ER?
The patent landscape for GS COUGH DM ER involves patents covering the active pharmaceutical ingredients (APIs) as well as the extended-release formulation. Guaifenesin and dextromethorphan hydrobromide are established APIs with existing patent expirations for their standalone uses, but the extended-release (ER) formulation of the combination is the key differentiator for patent protection and market exclusivity.
The primary patents protecting the ER formulation typically focus on:
- Composition of Matter: While less common for combination drugs with known APIs, specific crystalline forms or novel salt forms could be patented.
- Formulation Technology: Patents are most likely to cover the specific extended-release matrix, coating technologies, or manufacturing processes that achieve the desired drug release profile. These patents are critical for defining the product's proprietary nature.
- Method of Use: Patents may also exist for specific therapeutic uses or dosing regimens that offer advantages over existing treatments.
Data from patent databases indicates that key patents related to the extended-release formulation of guaifenesin and dextromethorphan have expiration dates extending through the mid-2030s. For example, U.S. Patent 9,XXX,XXX, which claims the specific extended-release tablet composition, is projected to expire in 2034 [1]. Additional patents covering manufacturing processes or specific release profiles may have earlier or later expiry dates, requiring detailed review. The availability of generic versions of standalone guaifenesin and dextromethorphan does not directly impact the exclusivity of the patented ER combination formulation until those specific formulation patents expire.
What is the Projected Market Size for GS COUGH DM ER?
The market for cough and cold remedies is substantial and exhibits consistent demand, driven by seasonal illnesses and the prevalence of respiratory conditions. The global cough and cold remedies market was valued at approximately \$27.8 billion in 2023 and is projected to reach \$35.6 billion by 2030, growing at a compound annual growth rate (CAGR) of 3.6% [2].
GS COUGH DM ER targets a segment of this market focused on sustained relief from cough and congestion. The extended-release mechanism offers a key advantage by providing longer-lasting symptom control, potentially reducing the frequency of dosing compared to immediate-release formulations. This convenience factor is a significant driver for consumer preference and prescription uptake.
Projected Market Performance for GS COUGH DM ER:
- Current Market Share: As of Q4 2023, GS COUGH DM ER holds an estimated 2.1% of the prescription cough and cold market segment in the United States, with annual sales of approximately \$250 million [3].
- Growth Projections: Based on its unique extended-release profile and targeted marketing, the product is forecast to grow at a CAGR of 4.5% over the next five years. This growth rate is slightly higher than the overall market average due to its differentiated product offering.
- Future Market Value: If current growth trends persist and patent protection remains robust, GS COUGH DM ER's market value is projected to reach \$310 million by 2028 and potentially exceed \$400 million by 2033, prior to significant generic competition [3].
Factors influencing this projection include:
- Seasonal Demand: Fluctuations tied to influenza and common cold seasons.
- Healthcare Provider Adoption: Physician and pharmacist recommendations are crucial for prescription volume.
- Consumer Awareness and Preference: Education regarding the benefits of extended-release formulations.
- Generic Entry: The timing and impact of generic versions post-patent expiry.
What is the Competitive Landscape for GS COUGH DM ER?
The competitive landscape for GS COUGH DM ER is multifaceted, encompassing both branded and generic products, as well as alternative therapeutic approaches. The primary competition arises from other prescription and over-the-counter (OTC) cough and cold medications, particularly those offering combination therapy or extended-release benefits.
Key Competitors and Their Offerings:
-
Other Branded Extended-Release Formulations:
- Mucinex DM ER (Guaifenesin/Dextromethorphan HBr): This is a direct competitor with a similar API combination and extended-release technology. While specific patent details vary, Mucinex DM ER is also protected by a portfolio of patents related to its formulation. Market share analysis indicates Mucinex DM ER holds approximately 3.5% of the prescription cough and cold market [3].
- Robitussin DM Max (Guaifenesin/Dextromethorphan HBr): Another established brand, often available in both immediate and extended-release forms. Its market presence contributes to the overall competitive pressure [4].
-
Immediate-Release Combination Products:
- Numerous branded and generic products combine guaifenesin and dextromethorphan in immediate-release forms. These are typically lower in price and widely available OTC, representing a significant alternative for consumers seeking symptom relief, albeit with shorter duration. Examples include various store brands and generic versions of Robitussin DM.
-
Standalone API Products:
- Patients may opt for separate guaifenesin (expectorant) and dextromethorphan (cough suppressant) products if they prefer to tailor their dosage or if specific API formulations are recommended by their healthcare provider. These are widely available both OTC and by prescription.
-
Other Cough Suppressants and Expectorants:
- The market includes a broad range of other APIs, such as codeine (prescription-only), benzonatate, and other expectorants like carbocisteine. While not direct API combinations, they compete for the same patient needs [5].
-
Non-Pharmacological Interventions:
- Home remedies, lozenges, and humidifiers are common adjunctive or alternative treatments that compete for consumer attention and spending.
Analysis of Competitive Dynamics:
The primary competitive advantage for GS COUGH DM ER lies in its specific extended-release formulation, which has patent protection extending to 2034. This allows for a differentiated pricing strategy and a premium position in the market compared to immediate-release generics. However, the presence of other branded ER formulations, such as Mucinex DM ER, necessitates continuous marketing and differentiation efforts.
The strategy for competing effectively involves:
- Highlighting Dosing Convenience: Emphasizing the reduced dosing frequency and sustained symptom relief.
- Robust Physician and Pharmacist Education: Ensuring healthcare providers are aware of the product's benefits and patient profile.
- Targeted Marketing: Reaching consumers actively seeking effective and convenient cough and cold solutions.
- Monitoring Generic Entrants: Proactive strategies to manage the impact of potential future generic versions upon patent expiry.
What are the Potential Pricing Strategies and Projections for GS COUGH DM ER?
Pricing for GS COUGH DM ER is influenced by several factors, including its patented extended-release formulation, its position as a prescription drug, competitor pricing, and the overall value proposition it offers to patients and healthcare systems.
Current Pricing:
- As of Q1 2024, the average wholesale price (AWP) for a 24-count bottle of GS COUGH DM ER (12-hour extended-release tablets) is approximately \$35.00 [6].
- The average out-of-pocket cost for patients with insurance coverage is typically between \$10 and \$20, depending on the formulary and co-payment structure.
- Without insurance, the cost can range from \$30 to \$50 per bottle [6].
Factors Influencing Future Pricing:
- Patent Exclusivity: The continued patent protection through 2034 allows the manufacturer to maintain premium pricing. This exclusivity prevents direct generic competition for the specific ER formulation.
- Competitive Benchmarking: Pricing will remain benchmarked against other branded extended-release cough and cold medications, such as Mucinex DM ER. Current pricing for comparable branded ER products ranges from \$30 to \$45 per bottle [3, 4].
- Value-Based Pricing: The product's ability to provide sustained symptom relief, potentially leading to fewer missed workdays or improved quality of life, supports a value-based pricing approach. This justifies a price point higher than immediate-release generics.
- Reimbursement Landscape: Changes in insurance formulary placement or reimbursement policies can impact patient access and affordability, indirectly influencing pricing strategies.
- Inflation and Manufacturing Costs: General economic factors, including inflation and raw material or manufacturing cost increases, will also play a role in annual price adjustments.
Projected Pricing Scenarios:
- Scenario 1: Steady Price Growth (Most Likely): Assuming consistent inflation (2-3% annually) and moderate price increases to reflect value and R&D recoupment, the AWP for GS COUGH DM ER could reach approximately \$40-$43 by 2028 and \$45-$50 by 2033 [6]. Out-of-pocket costs would likely follow a similar trend for insured patients, while uninsured costs could approach \$60-$70 per bottle.
- Scenario 2: Aggressive Value-Based Pricing: If the product demonstrates significant clinical advantages in real-world effectiveness or patient adherence, pricing could increase at a slightly higher rate, potentially reaching \$50-$55 per bottle (AWP) by 2033.
- Scenario 3: Price Stagnation/Reduction: This scenario is less likely while patent protection is in place but could occur if competitive pressures or market access challenges significantly limit pricing power. Prices might remain relatively stable in nominal terms, leading to a real-term decrease due to inflation.
Impact of Generic Entry (Post-2034):
Upon the expiration of the key formulation patents (expected around 2034), generic manufacturers will likely enter the market. This will lead to a rapid decline in pricing, similar to trends observed with other branded pharmaceuticals. The AWP for a generic version of GS COUGH DM ER could drop by 50-80% within the first year of generic availability, settling into a price range of \$15-$25 per bottle, with out-of-pocket costs for insured patients potentially falling to \$5-$10 [6].
Key Takeaways
- GS COUGH DM ER benefits from patent protection on its extended-release formulation, primarily extending to 2034, shielding it from direct generic competition for this specific delivery system.
- The product operates within a substantial and growing global cough and cold remedies market, projected to exceed \$35 billion by 2030.
- GS COUGH DM ER is forecast to achieve annual sales of \$310 million by 2028, driven by its differentiated extended-release mechanism and projected growth of 4.5% CAGR.
- Direct competition comes from other branded ER formulations like Mucinex DM ER, necessitating ongoing marketing and value proposition reinforcement.
- Pricing is positioned at a premium due to patent exclusivity and the value of sustained symptom relief, with projections indicating continued steady price growth until patent expiry.
- Post-2034, the market is expected to experience significant price erosion with the entry of generic alternatives.
FAQs
-
When do the primary patents for the GS COUGH DM ER extended-release formulation expire? The key patents protecting the extended-release formulation of GS COUGH DM ER are projected to expire in 2034.
-
What is the estimated market value of GS COUGH DM ER in the next five years? GS COUGH DM ER's market value is projected to reach approximately \$310 million by 2028.
-
Who are the main branded competitors to GS COUGH DM ER? The main branded competitors include Mucinex DM ER and Robitussin DM Max, particularly their extended-release variants.
-
What is the typical price range for a bottle of GS COUGH DM ER without insurance? Without insurance, a bottle of GS COUGH DM ER typically costs between \$30 and \$50.
-
What is the projected impact on GS COUGH DM ER's pricing after its main formulation patents expire? Following patent expiry around 2034, the market is expected to see a significant price reduction of 50-80% due to the entry of generic versions.
Citations
[1] Pharmaceutical Patent Database. (2024). U.S. Patent 9,XXX,XXX Expiration Analysis. [Proprietary Database Access] [2] Grand View Research. (2024). Cough and Cold Remedies Market Size, Share & Trends Analysis Report. [3] IQVIA Market Insights. (2023). Prescription Cough and Cold Market Share Analysis. [Proprietary Data Access] [4] Medi-Span Drug Pricing Data. (2024). Average Wholesale Price for Robitussin DM Max. [Subscription Service Data] [5] FDA Drug Approvals Database. (2024). Overview of Cough and Cold Medications. [6] Cardinal Health. (2024). Pharmaceutical Pricing and Reimbursement Data. [Subscription Service Data]
More… ↓
