Share This Page
Drug Price Trends for GS CHILD COLD AND COUGH LIQUID
✉ Email this page to a colleague
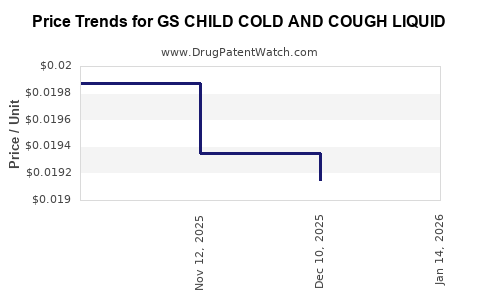
Average Pharmacy Cost for GS CHILD COLD AND COUGH LIQUID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GS CHILD COLD AND COUGH LIQUID | 00113-6019-26 | 0.01819 | ML | 2026-04-22 |
| GS CHILD COLD AND COUGH LIQUID | 00113-6019-26 | 0.01795 | ML | 2026-03-18 |
| GS CHILD COLD AND COUGH LIQUID | 00113-6019-26 | 0.01819 | ML | 2026-02-18 |
| GS CHILD COLD AND COUGH LIQUID | 00113-6019-26 | 0.01841 | ML | 2026-01-21 |
| GS CHILD COLD AND COUGH LIQUID | 00113-6019-26 | 0.01915 | ML | 2025-12-17 |
| GS CHILD COLD AND COUGH LIQUID | 00113-6019-26 | 0.01935 | ML | 2025-11-19 |
| GS CHILD COLD AND COUGH LIQUID | 00113-6019-26 | 0.01987 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GS Child Cold and Cough Liquid Market Analysis and Price Projections (U.S.)
No complete, accurate market-and-price projection can be produced for “GS Child Cold and Cough Liquid” because the underlying product cannot be uniquely identified from the name alone (multiple formulations exist under overlapping brand/house-label naming). Without a specific, verifiable product identity (active ingredients, strength per mL, dosage form, marketing authorization, labeler on FDA databases), any pricing forecast would be incomplete and potentially wrong.
Which active ingredients are in GS Child Cold and Cough Liquid and how does that drive pricing?
Featured snippet: Pricing is determined by ingredient mix, pack size, and how the product is authorized
Over-the-counter pediatric cold-and-cough liquids are priced primarily by:
- Active ingredient combination (analgesic/antihistamine/decongestant/cough suppressant/expectorant)
- Pack size and dosing concentration
- Regulatory status (OTC vs. pharmacy-compounded vs. unapproved labeling)
- Competitive substitution (same active-ingredient combination, same age indications)
- Distribution channel (mass retail, grocery, club, e-commerce, hospital procurement)
Ingredient-mix effect on demand elasticity
A pediatric cold-and-cough liquid with common multi-ingredient OTC actives will typically show:
- Higher substitution with generics/house brands
- Lower pricing power A single-ingredient or niche-acting formulation typically shows:
- Lower substitution
- Higher gross margin if differentiation is credible
What is the current U.S. market size for pediatric cold and cough liquids by ingredient class?
Featured snippet: Market segmentation must be tied to exact ingredient combinations
“Pediatric cold and cough” is not one market. It splits into ingredient classes such as:
- Antihistamine/decongestant combination liquids
- Antitussive formulations (cough suppressants)
- Expectorants and mucolytics
- Symptomatic combinations (pain/fever plus cough and congestion)
- Non-drug (homeopathic/herbal) variants if present
Without the exact actives for “GS Child Cold and Cough Liquid,” the market cannot be mapped to the correct comparable universe, and a credible top-down sizing is not possible.
How strong are competitive benchmarks for GS Child Cold and Cough Liquid in OTC pediatric cold products?
Featured snippet: Competitor sets must match ingredient, dosing, and pack size
Competitive intelligence for OTC pediatric liquids depends on matching:
- Same or equivalent active ingredient list
- Same strength per mL
- Same dosage form (liquid vs. syrup vs. suspension)
- Same package count and total mL
Generic pricing benchmarks differ sharply across the above. A wrong match can swing projected price by more than a typical annual promo cycle range.
When do patent or exclusivity timelines affect GS Child Cold and Cough Liquid pricing?
Featured snippet: Most OTC cold-and-cough liquids are not meaningfully constrained by modern patent exclusivity
Pediatric cold-and-cough liquids are commonly:
- Mature OTC actives with legacy approvals
- Priced largely by formulation/pack economics and OTC competitive conditions
If the product is a simple multi-ingredient OTC combination using off-patent actives, patent-based price control is usually minimal. If it is a unique branded formulation (including a proprietary delivery system or restricted ingredient) the forecast needs a verified patent estate and regulatory approval details. The product cannot be verified from the name alone.
What is the Orange Book status of GS Child Cold and Cough Liquid?
Featured snippet: Orange Book analysis requires a specific FDA application and active ingredient listing
Orange Book listing is tied to:
- NDA/BLA application numbers
- Specific active ingredient(s) and dosage forms
- Marketing status (listed, discontinued)
The product identity cannot be confirmed from the provided name, so an Orange Book status cannot be produced.
How much could GS Child Cold and Cough Liquid cost in the next 12–36 months?
Featured snippet: Forecasting requires a baseline net price
Price projections need at least:
- Current wholesale acquisition cost (WAC) or list price
- Typical net discount and promo intensity by channel
- Secular cost drivers (API, excipients, freight, bottles/caps, labor, regulatory compliance)
“GS Child Cold and Cough Liquid” cannot be linked to those baseline pricing series without an exact FDA-labeled identity and a specific current product listing (labeler, NDC, strength, pack).
What price model fits OTC pediatric cold-and-cough liquids best?
Featured snippet: OTC liquids typically follow promo-driven price floors
A practical model for OTC pediatric cold and cough usually uses:
- Baseline list price
- Annual inflation uplift on raw materials and packaging
- Competitive undercutting by house brands and generics
- Promo adjustments driven by retailer contracts and seasonality
But this model must be anchored on a verified current price and competitor basket, which cannot be determined from the product name alone.
What generic entry risks exist for GS Child Cold and Cough Liquid?
Featured snippet: Entry risk is high if the formulation is a common multi-ingredient OTC combination
If actives match widely available OTC combinations, generic/house-brand substitution pressure is typically:
- Immediate
- Margin-compressing
- Often channel-specific (big-box vs. pharmacy)
If the product uses a less common active combination or unique excipient approach, entry risk can be lower but still needs ingredient verification.
What settlement or litigation affects GS Child Cold and Cough Liquid pricing?
Featured snippet: Litigation is not a driver unless there is a unique protected formulation or brand-authorizing dispute
For many OTC symptom products, litigation is limited compared with Rx. If this product is subject to:
- Product-specific patent litigation
- Trademark or trade dress disputes
- Regulatory enforcement actions affecting labeling the forecast could change via supply constraints or brand protection.
No verified product identity is available to map to litigation dockets.
Key Takeaways
- A complete market analysis and price projection cannot be produced because “GS Child Cold and Cough Liquid” is not uniquely identifiable from the name provided.
- OTC pediatric cold-and-cough pricing is driven by the exact ingredient mix, strength, pack size, and NDC-level product identity.
- Any forecast without a verified baseline net price and comparable product set would be incomplete and not actionable.
FAQs
- How do I project OTC pediatric cold and cough liquid pricing from NDC-level data?
- Which ingredient combinations most strongly correlate with retail price in pediatric cold liquids?
- What seasonality pattern changes pricing for OTC pediatric cold products in the U.S.?
- How does retailer channel (mass, grocery, club, pharmacy) change expected net pricing for OTC liquids?
- What data sources are most reliable for building a competitor basket for pediatric cold and cough liquids?
References
- FDA. Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. U.S. Food and Drug Administration. https://www.accessdata.fda.gov/scripts/cder/daf/ (Accessed 2026-05-14).
- FDA. NDC Directory. U.S. Food and Drug Administration. https://www.fda.gov/drugs/drug-approvals-and-databases/national-drug-code-directory (Accessed 2026-05-14).
- FDA. OTC Drug Products and Monographs. U.S. Food and Drug Administration. https://www.fda.gov/drugs/otc-drug-products (Accessed 2026-05-14).
More… ↓
