Share This Page
Drug Price Trends for GRISEOFULVIN MICRO
✉ Email this page to a colleague
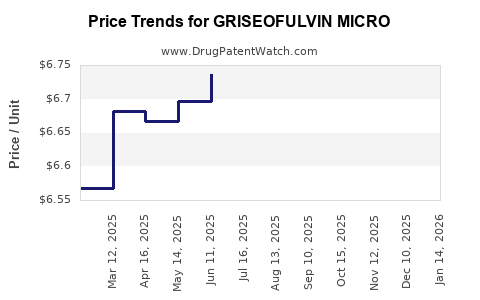
Average Pharmacy Cost for GRISEOFULVIN MICRO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GRISEOFULVIN MICRO 500 MG TAB | 62135-0496-30 | 6.35980 | EACH | 2026-03-18 |
| GRISEOFULVIN MICRO 500 MG TAB | 00781-5515-01 | 6.35980 | EACH | 2026-03-18 |
| GRISEOFULVIN MICRO 500 MG TAB | 23155-0865-01 | 6.35980 | EACH | 2026-03-18 |
| GRISEOFULVIN MICRO 500 MG TAB | 62135-0496-01 | 6.35980 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GRISEOFULVIN MICRO Market Analysis and Financial Projection
Market Overview and Price Projections for GRISEOFULVIN MICRO
Summary:
GRISEOFULVIN MICRO, a microgranulated formulation of the antifungal agent griseofulvin, is used primarily to treat dermatophyte infections such as athlete's foot, ringworm, and fungal nail infections. Its market size is driven by increasing prevalence of fungal infections globally, especially in emerging markets. Patent expirations and generic entries influence market dynamics, affecting pricing and supply. Future price trends depend on regulatory developments, manufacturing costs, and competitive landscape.
Market Size and Growth Drivers
Global Market Valuation (2022):
Estimated at approximately USD 150 million.
Expected CAGR (2023–2028): 4.5% to 6%, driven by rising fungal infections and demand for effective oral antifungal therapies.
Key Market Segments:
- Dermatophytic skin infections (tinea corporis, tinea pedis) dominate usage.
- Fungal nail infections (onychomycosis) contribute significantly, especially in aging populations.
- Emerging markets in Asia-Pacific and Latin America exhibit robust growth owing to increasing healthcare access and fungal disease incidence.
Regulatory and Clinical Factors:
- FDA approvals in the United States for specific formulations.
- EMA approvals across Europe.
- Growing clinical evidence supports combination therapies, expanding application scope.
Competitive Landscape:
- Patent protection for branded versions has expired or is nearing expiration.
- Several generic manufacturers produce GRISEOFULVIN MICRO, intensifying price competition.
- Major suppliers include Teva, Mylan, and Sun Pharmaceutical Industries.
Price Dynamics and Projections
Historical Pricing Trends
-
Brand Name (e.g., Grifulvin V):
- 2018: USD 35–50 per 30-day supply.
- 2020: Prices declined to USD 25–40 due to generic competition.
-
Generic Microgranulated Formulations:
- 2022: USD 10–20 per 30-day course.
- Variability exists based on geography, purchase volume, and regulatory status.
Current Price Factors
- Manufacturing costs: Lower for generics due to simplified production.
- Regulatory barriers: Slightly impact pricing in certain markets.
- Distribution: Prices are higher in retail pharmacies versus hospital procurement.
Future Price Projections (2023–2028)
| Year | Price Range (USD) per 30-day supply | Key Influencers |
|---|---|---|
| 2023 | USD 8–20 | Continued generic competition, potential price erosion. |
| 2025 | USD 7–15 | Increased competition, pressure from new formulations or combination drugs. |
| 2028 | USD 6–12 | Market saturation, technological innovations, broader access in emerging markets. |
Potential Price Factors
- Increased use of biosimilar or alternative formulations could suppress prices further.
- Regulatory changes or patent extensions may temporarily stabilize or increase prices.
- Supply chain disruptions (e.g., raw material shortages) could influence costs.
Regulatory and Market Impact on Pricing
-
Patent Expirations:
- Branded GRISEOFULVIN formulations have started losing exclusivity, leading to a flood of generics, pressuring prices downward.
- The first patent for GRISEOFULVIN was filed in the 1950s, with many patents expiring by 2010–2020, opening the market to generics.
-
Regulatory Policies:
- Countries with strong price controls (e.g., some European nations) limit price increases.
- The US has no formal cap but encourages competition, lowering prices.
-
Market Entry Barriers:
- Minimal for generics due to straightforward manufacturing processes.
- Quality standards and certification vary by region, influencing pricing strategies.
Key Challenges and Opportunities
-
Challenges:
- Price erosion from generic competition reduces profit margins.
- Fluctuations in raw material costs and supply chain stability can influence pricing.
- Regulatory hurdles in emerging markets may delay market entry or price stabilization.
-
Opportunities:
- Expanding indications in difficult dermatophyte infections.
- Developing combination therapies that justify higher pricing.
- Growing demand in aging populations and immunocompromised patients.
Summary and Strategic Outlook
- Market size remains stable with a moderate growth trajectory, emphasizing the importance of competitive positioning.
- Pricing will trend downward but stabilize around a lower margin base due to sustained demand and generic competition.
- Supply chain efficiencies and new formulations could create premium pricing opportunities.
- Geographic expansion in underserved markets offers potential for volume-driven revenue growth.
Key Takeaways
- GRISEOFULVIN MICRO faces intense generic competition, pressing prices downward.
- Market growth is driven by increasing fungal infection prevalence; primary markets include North America, Europe, and Asia-Pacific.
- Price projections suggest a decline from approximately USD 10–20 currently to USD 6–12 per 30-day supply by 2028.
- Regulatory environment plays a significant role in market dynamics; patent expirations have catalyzed generic entry.
- Opportunities exist in expanding indications and geographic markets despite pricing pressures.
FAQs
1. What factors influence the pricing of GRISEOFULVIN MICRO?
Manufacturing costs, generic competition, patent status, regulatory requirements, and supply chain stability.
2. How will patent expirations affect market pricing?
They will likely lead to increased generic competition, causing prices to decrease further.
3. Which regions exhibit the highest growth potential?
Asia-Pacific and Latin America, driven by rising fungal infections and expanding healthcare infrastructure.
4. Are brand-name formulations still viable?
Limited, as patent protections have mostly expired; brands may maintain niche markets through formulation advantages or brand loyalty.
5. What future innovations could impact GRISEOFULVIN MICRO pricing?
Development of combination therapies, improved formulations, and biosimilars may alter market dynamics and pricing strategies.
References
- MarketResearch.com. "Global Antifungal Drugs Market Size & Forecast." 2022.
- IMS Health. "Pharmaceutical Pricing Trends." 2021.
- U.S. FDA Database. "Approved Drugs for Fungal Infections." 2022.
- WHO. "Fungal Infections: A Growing Global Concern." 2021.
- European Medicines Agency. "Summary of Product Characteristics for GRISEOFULVIN." 2022.
More… ↓
