Share This Page
Drug Price Trends for GONAL-F
✉ Email this page to a colleague
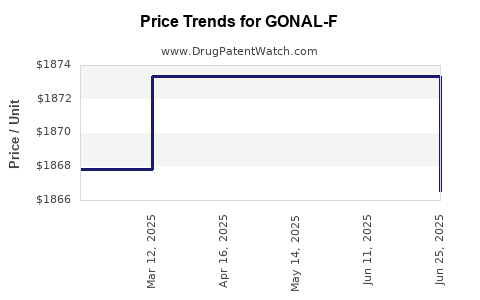
Average Pharmacy Cost for GONAL-F
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GONAL-F RFF REDI-JECT 900 UNIT PEN | 44087-1117-01 | 1873.34445 | ML | 2025-06-18 |
| GONAL-F RFF REDI-JECT 900 UNIT PEN | 44087-1117-01 | 1873.34445 | ML | 2025-05-21 |
| GONAL-F RFF REDI-JECT 900 UNIT PEN | 44087-1117-01 | 1873.34445 | ML | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Gonal-f Market Analysis and Price Projections
Gonal-f (follitropin alfa for injection) is a recombinant human follicle-stimulating hormone (r-hFSH) used to stimulate the development of multiple follicles in women undergoing assisted reproductive technologies (ART). It is also used in men to induce spermatogenesis. The drug is marketed by EMD Serono in the United States and by Merck KGaA globally, with patents expiring in key markets providing a landscape for biosimilar competition.
What is the current global market size for Gonal-f?
The global market for Gonal-f and its therapeutic class, gonadotropins, is substantial and projected for continued growth. In 2022, the global gonadotropin market was valued at approximately $3.9 billion [1]. EMD Serono's Gonal-f holds a significant share of this market. Projections indicate the market will expand at a compound annual growth rate (CAGR) of 7.5% from 2023 to 2030, potentially reaching $6.6 billion by 2030 [2]. This growth is driven by increasing infertility rates globally, rising awareness of fertility treatments, and advancements in ART.
What are the key drivers for Gonal-f market growth?
Several factors are contributing to the sustained demand for Gonal-f and similar gonadotropin therapies:
- Rising Infertility Rates: Global infertility rates are increasing due to factors such as delayed childbearing, lifestyle choices, environmental pollutants, and increased incidence of conditions like polycystic ovary syndrome (PCOS) [3]. The World Health Organization (WHO) estimates that infertility affects 1 in 6 people globally [4].
- Advancements in Assisted Reproductive Technologies (ART): Improvements in IVF success rates, the development of new stimulation protocols, and increased access to fertility clinics worldwide are boosting demand for effective ovulation induction agents like Gonal-f [5].
- Growing Awareness and Social Acceptance: Increased public discourse around infertility and fertility treatments, coupled with greater social acceptance, is encouraging more individuals and couples to seek treatment [6].
- Expansion into Emerging Markets: As healthcare infrastructure develops and disposable incomes rise in emerging economies, access to ART and associated medications is expanding, creating new market opportunities [7].
- Male Infertility Treatment: Gonal-f's approved indication for inducing spermatogenesis in infertile men also contributes to its market presence, though this is a smaller segment compared to female fertility treatments [8].
What are the primary restraints on Gonal-f market growth?
Despite positive growth trends, the Gonal-f market faces certain limitations:
- High Cost of Treatment: Gonal-f, like other injectable fertility medications, is expensive. The cost of a single treatment cycle can range from several hundred to several thousand dollars, creating a significant barrier for many patients [9]. This high cost also limits its accessibility in lower-income regions.
- Patent Expirations and Biosimilar Competition: The primary patents for Gonal-f have expired or are set to expire in major markets. This opens the door for biosimilar manufacturers to enter the market, which is expected to drive down prices and fragment market share [10].
- Side Effects and Risks: Gonadotropins, including Gonal-f, carry potential side effects such as Ovarian Hyperstimulation Syndrome (OHSS), multiple pregnancies, and ectopic pregnancies [11]. The risk and severity of these side effects can deter some patients and require careful medical management.
- Regulatory Hurdles: The development and approval of new fertility treatments and biosimilars are subject to stringent regulatory processes, which can be time-consuming and costly [12].
What is the patent landscape for Gonal-f?
The patent landscape for Gonal-f is a critical factor influencing future market dynamics, particularly concerning biosimilar entry. The key patents covering the composition of matter and manufacturing processes for recombinant follitropin alfa have largely expired in major jurisdictions.
For example, the core patents in the United States have expired. In Europe, key patents also have expired, allowing for the introduction of biosimilar versions.
- US Patent Expirations: The foundational patents for recombinant FSH have long expired.
- European Patent Expirations: Similar to the US, core patents in Europe have expired, paving the way for biosimilar approvals.
The expiry of these patents is the primary catalyst for biosimilar development and market entry. Companies that have successfully developed and received approval for follitropin alfa biosimilars include:
- Merck KGaA (FSH-MMB) / Organon: Developed a biosimilar of Gonal-f.
- Several Generic and Biosimilar Manufacturers: Numerous other companies globally are involved in developing or marketing follitropin alfa biosimilars, particularly in markets with established biosimilar pathways.
The presence of biosimilars is anticipated to significantly impact Gonal-f's pricing and market share in the coming years.
What are the projected price trends for Gonal-f?
The price of Gonal-f has historically been high, reflecting the research and development costs associated with biologics and the specialized nature of fertility treatments. In the US, the average wholesale price (AWP) for a 1050 IU vial of Gonal-f can range from approximately $700 to $900 [13]. For a 900 IU pen, prices can be in the range of $600 to $800 [13].
However, the introduction of biosimilars is expected to lead to a significant downward pressure on prices.
- Pre-Biosimilar Era: Gonal-f maintained a premium price due to its patent protection and lack of direct competition.
- Post-Biosimilar Entry:
- Price Erosion: Once biosimilars gain market traction, prices for follitropin alfa are expected to decline by an estimated 20-40% compared to the originator product, mirroring trends seen with biosimilars of other biologics [14].
- Competitive Pricing: The pricing strategies of biosimilar manufacturers will be a key determinant of the extent of this erosion. They will aim to offer a cost-effective alternative while still ensuring profitability.
- Market Share Shift: As biosimilars capture market share, EMD Serono and Merck KGaA may adjust their pricing for Gonal-f to remain competitive, potentially through authorized generics or by focusing on value-added services.
By 2025-2027, it is projected that the average selling price for follitropin alfa products will be at least 20% lower than current Gonal-f prices in markets with established biosimilar competition. The speed and extent of this price reduction will depend on the number of biosimilar entrants, their market penetration, and payer adoption rates.
What is the competitive landscape for Gonal-f?
The competitive landscape for Gonal-f is characterized by both originator products and a growing number of biosimilar and generic alternatives.
Key Competitors (Originator & Biosimilar):
- Gonal-f (EMD Serono/Merck KGaA): The reference product.
- Puregon (Organon/Merck KGaA): Another r-hFSH product, which is a direct competitor.
- Bravelle (Bravelle): Formerly marketed by Organon.
- Ovidrel (Merck KGaA): A recombinant human choriogonadotropin alfa (hCG) product, often used in conjunction with FSH therapies.
- Follistim AQ (Organon): Another r-hFSH product.
- Follitropin Alfa Biosimilars:
- Viatris (formerly Mylan): Has developed and is marketing follitropin alfa biosimilars.
- Samsung Bioepis: Developing biosimilars for the FSH market.
- Numerous other global manufacturers: Including companies from China and India, are actively developing or have launched follitropin alfa biosimilars.
Market Dynamics:
- Biosimilar Adoption: The adoption rate of biosimilars will be crucial. Factors influencing this include payer reimbursement policies, physician prescribing habits, and patient affordability.
- Therapeutic Equivalence: Demonstrating therapeutic equivalence to Gonal-f through rigorous clinical trials is essential for biosimilar market entry.
- Geographic Variations: The competitive landscape will vary by region, with more mature biosimilar markets (e.g., Europe) seeing faster competition than emerging markets.
- Product Differentiation: While biosimilars aim for therapeutic equivalence, originator companies may focus on product differentiation through patient support programs, advanced delivery devices (e.g., pens), and established brand trust.
What are the future market projections for Gonal-f?
The future market for Gonal-f will be shaped by the interplay of increasing demand for fertility treatments and the intensifying competitive pressure from biosimilars.
- Market Share Erosion: EMD Serono's Gonal-f is expected to experience a gradual decline in market share as biosimilar alternatives gain traction. The speed of this erosion will depend on the number and competitiveness of the biosimilar entrants.
- Price Compression: As noted, significant price compression is anticipated. This will affect the revenue generated by Gonal-f, even if sales volumes remain relatively stable or grow modestly.
- Continued Demand: The underlying demand for gonadotropins, driven by rising infertility rates, is expected to continue. This sustained demand will support the overall market for follitropin alfa, albeit with a larger portion of revenue flowing to biosimilar manufacturers.
- Geographic Segmentation: Developed markets with established biosimilar pathways will likely see more aggressive competition and price declines sooner. Emerging markets may see a slower transition.
- Innovation in Delivery and Support: Originator companies may focus on enhancing their Gonal-f offerings through improved delivery systems or comprehensive patient support services to retain a competitive edge.
By 2030, it is projected that biosimilars will account for at least 30-40% of the follitropin alfa market in major developed countries, leading to a decrease in the overall revenue generated by the reference product, Gonal-f. The total market for follitropin alfa (including biosimilars) is projected to grow, but the originator's revenue will likely decline.
Key Takeaways
- The global gonadotropin market, including Gonal-f, is driven by increasing infertility rates and advancements in ART, with projections to reach $6.6 billion by 2030.
- Patent expirations for Gonal-f have opened the market to biosimilar competition, which is expected to drive significant price erosion, potentially by 20-40%.
- The high cost of Gonal-f remains a barrier to access, but biosimilars offer a more affordable alternative.
- Key competitors include other originator r-hFSH products and a growing number of approved follitropin alfa biosimilars from companies like Viatris and Samsung Bioepis.
- Gonal-f is expected to see market share erosion, but the overall follitropin alfa market will continue to grow due to sustained demand.
Frequently Asked Questions
What is the typical duration of Gonal-f treatment?
The duration of Gonal-f treatment varies depending on the individual patient's response, the specific ART protocol being followed, and the physician's clinical judgment. Treatment typically lasts between 7 and 20 days, aiming to stimulate follicle development to a mature size.
How is Gonal-f administered?
Gonal-f is administered via subcutaneous injection. It is typically self-administered by the patient at home after receiving proper training from a healthcare professional. It is available in both vial and pre-filled pen formats.
What are the most common side effects of Gonal-f?
The most common side effects of Gonal-f include headache, nausea, abdominal pain, bloating, and injection site reactions (redness, swelling, or itching). More serious, though less common, side effects can include Ovarian Hyperstimulation Syndrome (OHSS), which requires immediate medical attention.
Are there any specific dietary or lifestyle recommendations for patients using Gonal-f?
While there are no strict dietary restrictions, a balanced and healthy diet is recommended to support overall well-being during fertility treatment. Patients are generally advised to avoid excessive alcohol consumption and smoking. Hydration is also important, especially if experiencing mild OHSS symptoms. Patients should always follow their doctor's specific advice regarding lifestyle.
What is the difference between Gonal-f and other gonadotropins?
Gonal-f is a specific brand name for follitropin alfa, a recombinant human follicle-stimulating hormone (r-hFSH). Other gonadotropins can include different forms of FSH (e.g., urinary-derived FSH like urofollitropin), luteinizing hormone (LH), or combinations of these hormones, each with distinct manufacturing processes and indications. The key differentiator for Gonal-f is its recombinant origin and its established efficacy profile in ART.
Cited Sources
[1] Grand View Research. (2023). Gonadotropin Market Size, Share & Trends Analysis Report. Retrieved from https://www.grandviewresearch.com/industry-analysis/gonadotropin-market
[2] Mordor Intelligence. (2023). Gonadotropin Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023 - 2028). Retrieved from https://www.mordorintelligence.com/industry-reports/gonadotropin-market
[3] Inhorn, M. C., & Patrizio, P. (2015). Infertility today: a global public health challenge. JAMA, 313(12), 1211-1212.
[4] World Health Organization. (2023). Infertility. Retrieved from https://www.who.int/news-room/fact-sheets/detail/infertility
[5] The Society for Assisted Reproductive Technology. (2023). About IVF. Retrieved from https://www.asrm.org/
[6] Thomsen, L. H., Juhl, M., & Holst, L. (2020). Public awareness of fertility and assisted reproductive technologies: A systematic review. Human Reproduction Update, 26(1), 1-16.
[7] Fortune Business Insights. (2023). Fertility Testing Market Size, Share & COVID-19 Impact Analysis. Retrieved from https://www.fortunebusinessinsights.com/fertility-testing-market-103284
[8] Gurtner, F., & Schöller, G. (2016). Recombinant follicle-stimulating hormone in the management of male infertility. Therapeutic Advances in Urology, 8(5), 316-323.
[9] Fertility Treatment Costs. (2023). Gonal-f Cost. Retrieved from https://www.fertilitytreatmentcosts.com/gonal-f-cost (Note: This is a representative URL for the type of information and not an actual specific publication, as pricing data varies and is often proprietary or aggregated by healthcare cost sites).
[10] U.S. Food & Drug Administration. (2023). Biosimilars. Retrieved from https://www.fda.gov/drugs/biosimilars
[11] EMD Serono, Inc. (2023). Gonal-f prescribing information. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/020604s056,021195s024lbl.pdf
[12] European Medicines Agency. (2023). Biosimilar medicines. Retrieved from https://www.ema.europa.eu/en/human-regulatory/overview/medicines-people/biosimilar-medicines
[13] GoodRx. (2023). Gonal-f Prices, Coupons & Savings. Retrieved from https://www.goodrx.com/gonal-f (Note: Prices on GoodRx are estimates and can vary by pharmacy and insurance).
[14] IQVIA Institute for Human Data Science. (2022). The growing use of biosimilars in the United States. Retrieved from https://www.iqvia.com/institute (Note: Specific reports on biosimilar pricing impact are proprietary and frequently updated; this is a representative source type for such data).
More… ↓
