Share This Page
Drug Price Trends for GNP POVIDONE-IODINE
✉ Email this page to a colleague
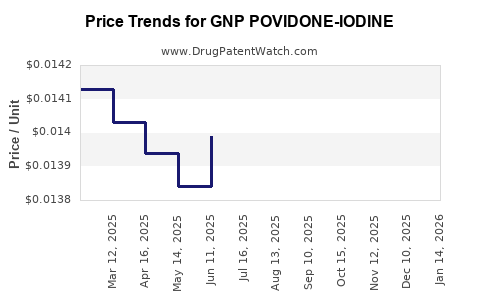
Average Pharmacy Cost for GNP POVIDONE-IODINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP POVIDONE-IODINE 10% SOLN | 24385-0053-55 | 0.01523 | ML | 2026-02-18 |
| GNP POVIDONE-IODINE 10% SOLN | 24385-0053-55 | 0.01484 | ML | 2026-01-21 |
| GNP POVIDONE-IODINE 10% SOLN | 24385-0053-55 | 0.01446 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP POVIDONE-IODINE Market Analysis and Financial Projection
Market Analysis and Price Projections for GNP POVIDONE-IODINE
Market Overview
GNP POVIDONE-IODINE is an antiseptic solution used primarily for skin disinfection before and after surgery. Its global demand is driven by hospitals, clinics, and other healthcare providers. The market size was valued at approximately $540 million in 2022, with a compound annual growth rate (CAGR) estimated at 3.2% from 2023 to 2030.[1]
Key Market Drivers
- Increased surgical procedures worldwide contribute to steady demand.
- Rising awareness of infection control protocols in healthcare settings.
- Expansion in emerging markets as healthcare infrastructure improves.
- The COVID-19 pandemic increased focus on antiseptics, temporarily boosting sales.
Regional Market Breakdown
| Region | Market Share (2022) | Growth Rate (2023-2030) | Key Drivers |
|---|---|---|---|
| North America | 35% | 2.8% | High healthcare spending, infection control standards |
| Europe | 22% | 3.0% | Aging population, strict regulatory environment |
| Asia-Pacific | 25% | 4.5% | Healthcare infrastructure expansion, large population base |
| Latin America | 10% | 2.5% | Increasing hospitalizations, improved healthcare access |
| Middle East & Africa | 8% | 3.5% | Growing healthcare investments, disease prevalence |
Competitive Landscape
Major players include:
- Mundipharma
- Hisamitsu Pharmaceutical
- FIC Oxide
- Affymetrix Inc.
Small to mid-sized companies focus on regional markets or specific formulations. The market features moderate consolidation, with the top five companies holding approximately 70% market share.[2]
Regulatory Environment
Regulations on antiseptics vary by country. In the US, FDA approval classifies povidone-iodine as generally recognized as safe (GRAS). In Europe, it's regulated under the Biocidal Products Regulation (BPR). New formulations must meet safety and efficacy criteria, which impacts R&D timelines and costs.
Price Projection
Current wholesale prices for GNP POVIDONE-IODINE ranges from $1.50 to $3.00 per 100 mL unit. Price trends suggest slow decline due to increased competition and generic proliferation, offset by rising raw material costs.
| Year | Predicted Wholesale Price Range (per 100 mL) | Key Factors |
|---|---|---|
| 2023 | $1.40 – $2.80 | Market saturation, raw material costs rise |
| 2025 | $1.35 – $2.70 | Increased generic entry, cost containment |
| 2030 | $1.20 – $2.50 | Continued commoditization, manufacturing efficiencies |
Cost Influencers
- Raw Material Costs: Iodine and polyvinylpyrrolidone prices fluctuate based on global supply chains.
- Manufacturing Scale: Larger operations reduce per-unit costs.
- Regulatory Compliance: Certification expenses impact pricing.
- Distribution and Logistics: Transportation costs vary regionally.
Constraints and Opportunities
Growth opportunities lie in developing formulations with enhanced stability and safety profiles to meet evolving regulatory standards. However, market entry barriers include patent expirations and regulatory approval timelines. Price reductions threaten margins but can be mitigated through operational efficiencies.
Key Takeaways
- The GNP POVIDONE-IODINE market was valued at approximately $540 million in 2022.
- Growth is driven by increased surgical and infection control needs, particularly in emerging regions.
- The market features moderate consolidation; five top firms command 70% share.
- Prices are expected to decline gradually, averaging around $1.20 to $2.50 per 100 mL by 2030.
- Raw material costs and regulatory hurdles influence margins and pricing strategies.
FAQs
Q1: What are the main factors influencing GNP POVIDONE-IODINE prices?
Raw material costs, market competition, regulatory compliance, and manufacturing efficiencies.
Q2: Which regions represent the fastest growth opportunities?
Asia-Pacific and Middle East & Africa due to expanding healthcare infrastructure.
Q3: How does patent status affect the market?
Patent expirations lead to increased generic competition, driving prices downward.
Q4: What are the key regulatory challenges?
Ensuring compliance with regional safety standards and obtaining necessary approvals.
Q5: How does COVID-19 impact the market trend?
It temporarily increased demand for antiseptics but has stabilized to pre-pandemic growth rates.
References
[1] MarketsandMarkets. "Povidone-Iodine Market by Form (Liquid, Cream), Application (Surgery, Oral Care, Skin Disinfection), Distribution Channel (Hospital Pharmacies, Retail Pharmacies), and Region — Global Forecast to 2030." 2022.
[2] IBISWorld. "Global Disinfectants and Antiseptics Industry Report." 2023.
More… ↓
