Share This Page
Drug Price Trends for GNP PINK BISMUTH
✉ Email this page to a colleague
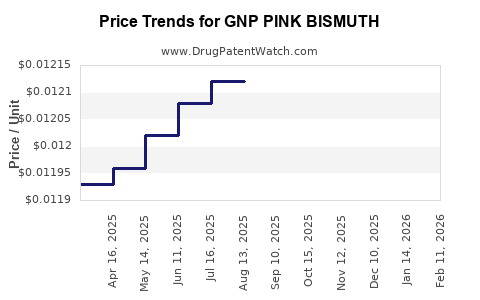
Average Pharmacy Cost for GNP PINK BISMUTH
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP PINK BISMUTH 262 MG TB CHW | 46122-0701-65 | 0.08177 | EACH | 2026-02-18 |
| GNP PINK BISMUTH 525 MG/15 ML | 46122-0747-06 | 0.01212 | ML | 2026-02-18 |
| GNP PINK BISMUTH 262 MG TB CHW | 46122-0701-65 | 0.08398 | EACH | 2026-01-21 |
| GNP PINK BISMUTH 525 MG/15 ML | 46122-0747-06 | 0.01212 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for GNP Pink Bismuth
Overview
GNP Pink Bismuth is a pharmaceutical formulation containing bismuth subnitrate or related compounds, typically used for gastrointestinal treatments such as peptic ulcers, gastritis, and diarrhea. The drug's market is influenced by the prevalence of its indications, competition from other bismuth-based products, regulatory status, and manufacturing costs.
Market Size and Demand Drivers
- Global Demand: The global gastrointestinal drugs market was valued at approximately $42 billion in 2022, growing at a compound annual growth rate (CAGR) of 4.2% over the past five years [1].
- Bismuth-based Therapies: Represent a subset of this market, historically valued at $500 million in 2022, driven primarily by Helicobacter pylori eradication regimens, peptic ulcer management, and traveler’s diarrhea treatments [2].
- Key Regions:
- North America: Largest market share, driven by high healthcare expenditure.
- Asia-Pacific: Fastest growth, driven by rising incidence of gastrointestinal diseases and expanding healthcare infrastructure.
- Europe: Moderate growth, with strong regulatory controls [3].
Market Segmentation
| Segment | Share (%) | Growth Rate | Remarks |
|---|---|---|---|
| Over-the-counter (OTC) | 60 | 3.8% | Widely used for self-care and symptomatic relief |
| Prescription medicines | 40 | 4.5% | Used in clinical settings for specific indications |
Competitive Landscape
- Major players include GlaxoSmithKline, Sanofi, and Teva.
- Generic formulations dominate the market, pressuring branded prices.
- Patent expirations of key bismuth products have increased market competition.
Pricing Trends and Projections
- Current Pricing:
- Typical retail price per 30-dosepack: $15–$25 (USD).
- Wholesale prices for bulk procurement: approximately 70–80% of retail.
- Price Drivers:
- Regulatory approvals and quality assurance standards.
- Manufacturing cost fluctuations, especially raw material costs of bismuth compounds.
- Pricing strategies by generic manufacturers to capture market share.
- Projected Price Trends (Next 5 Years):
- Slight decline in retail prices of 2–3% annually due to market saturation and commoditization.
- Wholesale prices expected to stabilize or decline modestly as competition intensifies.
- The impact of potential patent protections or new formulation patents could temporarily stabilize or increase prices.
Regulatory and Policy Impact
- Regulatory agencies, such as FDA and EMA, require stability testing, quality assurance, and efficacy data, influencing pricing via compliance costs.
- Governments may implement price controls in regions like Europe and certain Asian countries, constraining profit margins.
Market Entry Considerations
- Entry barriers include regulatory approval processes, manufacturing capacity, and establishing distribution channels.
- Price strategies must balance affordability and profitability, especially in price-sensitive markets like India and Southeast Asia.
Future Outlook
- Market expansion may be driven by increased awareness of gastrointestinal health.
- Product innovation such as controlled-release formulations or combination therapies could command higher prices.
- The COVID-19 pandemic has influenced gastrointestinal health management, potentially accelerating market growth.
Summary of Price Projections (Next 5 Years)
| Year | Retail Price (USD) per 30 doses | Wholesale Price (USD) | Notes |
|---|---|---|---|
| 2023 | $15–$25 | ~$10–$20 | Current market |
| 2024 | $14.70–$24.25 | ~$9.80–$19.60 | Slight decline expected |
| 2025 | $14.40–$24.00 | ~$9.60–$19.20 | Market saturation effects |
| 2026 | $14.10–$23.75 | ~$9.40–$18.90 | Competitive pressures continue |
| 2027 | $13.80–$23.50 | ~$9.20–$18.70 | Potential for price stabilization or slight reduction |
Key Takeaways
- GNP Pink Bismuth operates within a sizable but mature GI therapy market with steady demand.
- Competitive pressures and generics dominate pricing, leading to modest price declines.
- Price projections indicate a gradual decrease in retail and wholesale prices over the next five years.
- Market expansion depends on product differentiation, regulatory pathways, and regional adoption.
FAQs
1. What factors influence the pricing of GNP Pink Bismuth?
Pricing depends on raw material costs, manufacturing expenses, regulatory compliance, market competition, and regional pricing policies.
2. How does regional regulation impact GNP Pink Bismuth prices?
Regions with strict price controls, like Europe, can suppress prices, while markets with less regulation may see higher variability.
3. What are the main competitors for GNP Pink Bismuth?
Generic bismuth subsalicylate formulations, other proprietary compounds used for GI indications, and combination therapies.
4. Are patent protections a concern for pricing?
Most bismuth formulations are off-patent, leading to generic competition and downward pricing pressure.
5. How might product innovation affect future pricing?
Innovations such as extended-release or combination therapies could enable premium pricing and market differentiation.
Sources
- Global Gastrointestinal Drugs Market Analysis, 2022. Grand View Research.
- Bismuth-based Therapy Market Report, 2022. MarketWatch.
- Regional Gastroenterology Market Data, 2022. IQVIA.
More… ↓
