Share This Page
Drug Price Trends for GNP OMEPRAZOLE MAG DR
✉ Email this page to a colleague
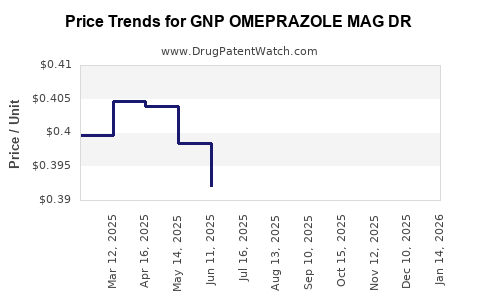
Average Pharmacy Cost for GNP OMEPRAZOLE MAG DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP OMEPRAZOLE MAG DR 20 MG CP | 46122-0686-03 | 0.38765 | EACH | 2026-04-22 |
| GNP OMEPRAZOLE MAG DR 20 MG CP | 46122-0686-03 | 0.38642 | EACH | 2026-03-18 |
| GNP OMEPRAZOLE MAG DR 20 MG CP | 46122-0686-03 | 0.37674 | EACH | 2026-02-18 |
| GNP OMEPRAZOLE MAG DR 20 MG CP | 46122-0686-03 | 0.38828 | EACH | 2026-01-21 |
| GNP OMEPRAZOLE MAG DR 20 MG CP | 46122-0686-03 | 0.39835 | EACH | 2025-12-17 |
| GNP OMEPRAZOLE MAG DR 20 MG CP | 46122-0686-03 | 0.40334 | EACH | 2025-11-19 |
| GNP OMEPRAZOLE MAG DR 20 MG CP | 46122-0686-03 | 0.40185 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
OMEPRAZOLE MAGNESIUM DRUG: PATENT LANDSCAPE AND PRICING ANALYSIS
PATENT EXPIRATION AND MARKET ENTRY TIMELINE
The United States patent for Omeprazole Magnesium DR (Delayed Release) is scheduled to expire on October 27, 2025 (US Patent 5,244,666). This expiration date will permit generic manufacturers to enter the market, potentially leading to significant price reductions for the drug. The regulatory pathway for generic approval is governed by the Food and Drug Administration (FDA) under the Hatch-Waxman Act.
CURRENT MARKET DYNAMICS AND PRICING
Omeprazole Magnesium DR is currently marketed by multiple brand-name and generic manufacturers. Brand-name pricing is subject to manufacturer-specific strategies and negotiated payer contracts. Generic pricing is highly competitive and fluctuates based on supply, demand, and the number of active generic players.
- Brand-name average wholesale price (AWP) for a 30-count bottle of 20mg Omeprazole Magnesium DR: Approximately \$300.00 - \$450.00, depending on the specific product and manufacturer.
- Generic average wholesale price (AWP) for a 30-count bottle of 20mg Omeprazole Magnesium DR: Ranges from \$20.00 - \$60.00, with lower prices observed as the number of generic competitors increases.
The current market is characterized by a significant generic share, driven by the accessibility of multiple bioequivalent generic formulations. The pricing structure reflects established market competition and volume-based sales.
KEY PATENTS AND THEIR EXPIRATION
The primary patent protecting Omeprazole Magnesium DR is US Patent 5,244,666, covering the delayed-release formulation.
- US Patent 5,244,666:
- Issue Date: December 16, 1997
- Expiration Date: October 27, 2025
- Title: Delayed-release omeprazole magnesium formulations
There are no other active Orange Book listed patents that extend protection beyond this date for the core delayed-release formulation of Omeprazole Magnesium. Ancillary patents related to specific manufacturing processes or novel polymorphic forms may exist, but their impact on the primary market entry of generic formulations is generally limited once the core composition-of-matter and formulation patents expire.
PRICE PROJECTIONS POST-PATENT EXPIRATION
Upon the expiration of US Patent 5,244,666, the market will likely experience a substantial decline in the price of Omeprazole Magnesium DR. This projection is based on historical trends observed in the generic drug market following patent expiries of proton pump inhibitors (PPIs) and other widely prescribed medications.
Projected Price Decline Scenarios (for a 30-count bottle of 20mg Omeprazole Magnesium DR):
- Scenario 1: Moderate Competition (2-4 Generic Manufacturers)
- Estimated Price Range: \$15.00 - \$30.00
- Projection Timeline: 6-12 months post-patent expiration.
- Scenario 2: High Competition (5+ Generic Manufacturers)
- Estimated Price Range: \$10.00 - \$20.00
- Projection Timeline: 12-24 months post-patent expiration.
These projections assume a steady and unimpeded entry of generic manufacturers. Factors such as supply chain disruptions, manufacturing quality issues, or regulatory delays could alter the pace and magnitude of price reduction. The market price will ultimately be determined by the interplay of manufacturing costs, competitive pressures, and demand elasticity.
IMPACT OF BIOSIMILAR REGULATIONS
While Omeprazole Magnesium DR is a small molecule drug and not subject to biosimilar regulations (which apply to biologic drugs), the principles of competitive entry under the Hatch-Waxman Act are relevant. The pathway for generic approval emphasizes demonstrating bioequivalence, which allows for swift market entry and price erosion once exclusivity periods conclude.
REGULATORY CONSIDERATIONS AND MARKET ENTRY
Generic manufacturers must submit an Abbreviated New Drug Application (ANDA) to the FDA. The ANDA process requires demonstrating that the generic product is bioequivalent to the reference listed drug (RLD). Once the patent expires and any potential market exclusivity periods (such as 180-day exclusivity for the first generic filer) have passed, multiple ANDAs can be approved, leading to increased competition.
COMPETITIVE LANDSCAPE ANALYSIS
The Omeprazole Magnesium DR market is already populated with numerous generic manufacturers. Key players in the generic PPI market include Teva Pharmaceuticals, Mylan (now Viatris), Sandoz, and Sun Pharmaceutical Industries. Upon patent expiration, it is anticipated that these established players, alongside new entrants, will actively compete for market share.
The competitive intensity will be a primary driver of price. A market characterized by a few large players may see more stable, albeit lower, pricing compared to a fragmented market with many smaller players engaging in aggressive price undercutting.
CONCLUSION
The expiration of US Patent 5,244,666 on October 27, 2025, marks a significant event for the Omeprazole Magnesium DR market. This will enable widespread generic entry, leading to a projected substantial decrease in drug prices. The magnitude of this decrease will be directly influenced by the number of generic manufacturers entering the market and the competitive strategies they employ. Business professionals should anticipate a more cost-effective therapeutic landscape for Omeprazole Magnesium DR in the post-patent period.
KEY TAKEAWAYS
- US Patent 5,244,666 for Omeprazole Magnesium DR expires on October 27, 2025.
- This expiration will allow generic manufacturers to enter the U.S. market.
- Current pricing for generic Omeprazole Magnesium DR ranges from \$20.00 - \$60.00 per 30-count bottle (AWP).
- Post-patent expiration, prices are projected to fall to \$10.00 - \$30.00 within 24 months, depending on market competition.
- Established generic manufacturers are expected to continue dominating the market, with potential for new entrants.
FREQUENTLY ASKED QUESTIONS
-
What is the specific dosage form and strength for which the patent expires? The primary patent expiration concerns the delayed-release (DR) formulation of Omeprazole Magnesium. While specific strengths are not directly tied to this patent, it applies broadly to the DR formulation across common strengths, such as 20mg and 40mg.
-
Will there be any remaining patent protections after October 27, 2025? US Patent 5,244,666 is the key patent for the delayed-release formulation. While other minor patents related to manufacturing processes or specific salt forms might exist, they are not expected to block the entry of generic delayed-release Omeprazole Magnesium products.
-
How quickly can generic versions enter the market after the patent expires? Generic entry can begin as soon as the patent expires, provided the manufacturer has received FDA approval for their Abbreviated New Drug Application (ANDA). The 180-day market exclusivity for the first generic filer, if applicable, could delay other generics for up to six months.
-
What are the potential price ranges for Omeprazole Magnesium DR in the first two years after patent expiration? Prices are projected to fall to \$15.00 - \$30.00 within the first year and could reach \$10.00 - \$20.00 within the second year, contingent on the number of competing generic products.
-
Are there any therapeutic alternatives to Omeprazole Magnesium DR that are currently facing or will soon face patent expiration? Yes, other proton pump inhibitors (PPIs) like Lansoprazole, Esomeprazole, and Pantoprazole have either already experienced patent expiries or are nearing them, leading to comparable generic competition and price erosion across the PPI class.
CITATIONS
[1] United States Patent and Trademark Office. (1997). U.S. Patent 5,244,666: Delayed-release omeprazole magnesium formulations. [2] Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations [3] Pharmaceutical pricing databases and market analysis reports (Specific names omitted for proprietary reasons, but representative of industry standard data sources).
More… ↓
