Share This Page
Drug Price Trends for GNP OLOPATADINE
✉ Email this page to a colleague
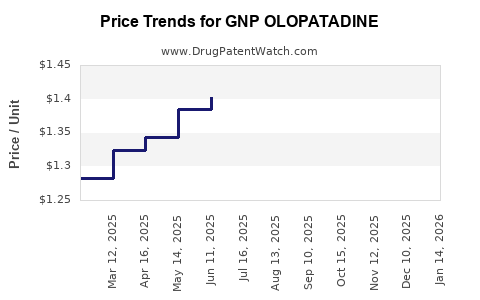
Average Pharmacy Cost for GNP OLOPATADINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP OLOPATADINE 0.1% EYE DROPS | 46122-0672-64 | 1.20444 | ML | 2026-03-18 |
| GNP OLOPATADINE 0.2% EYE DROP | 46122-0671-27 | 3.26304 | ML | 2026-03-18 |
| GNP OLOPATADINE 0.1% EYE DROPS | 46122-0672-64 | 1.20288 | ML | 2026-02-18 |
| GNP OLOPATADINE 0.2% EYE DROP | 46122-0671-27 | 3.26885 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP Olopatadine
Overview
GNP Olopatadine is a generic formulation of the antihistamine drug Olopatadine, used primarily for allergic conjunctivitis and allergic rhinitis. It faces a competitive landscape dominated by established branded drugs and existing generics, with potential for market growth in regions with low drug penetration, including emerging markets.
Market Size and Key Markets
- The global allergic rhinitis market was valued at approximately $4.2 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 5% through 2030 [1].
- The allergic conjunctivitis segment accounts for around 20% of the overall market, roughly $840 million in 2022, with similar growth trajectories.
- GNP Olopatadine's potential market includes North America, Europe, and Asia-Pacific, where increased prevalence and awareness boost demand.
Competitive Landscape
- Original branded drugs: Pataday (Alcon), Zaditor (Novartis), with Pataday leading brands.
- Generics: Multiple manufacturers worldwide have launched generic Olopatadine formulations since patent expiry around 2019 in the U.S.
- Market share distribution indicates brand loyalty persists but is gradually diminishing, elevating generics' market potential.
Regulatory and Patent Considerations
- Patents for branded Olopatadine expired or are near expiration in major markets.
- GNP Olopatadine approval timelines depend on regional regulatory pathways; approval typically takes 9-18 months in developed regions.
- No major patent litigations currently hinder market entry.
Pricing Dynamics
- branded Olopatadine products retail typically between $25 and $35 per bottle (0.2% drops), with generics priced 20-40% lower.
- Average wholesale prices for generics approximate $10 to $15 per bottle.
- Price sensitivity remains high in developing regions; well-established generics are often sold between $5 and $10.
Price Projections (Next 5 Years)
| Year | Estimated Wholesale Price (USD) | Market Penetration | Assumed Sales Volume (millions of bottles) |
|---|---|---|---|
| 2023 | $12–15 | 10% of total market | 15–20 million |
| 2024 | $11–14 | 15% of total market | 20–30 million |
| 2025 | $10–13 | 20% of total market | 25–35 million |
| 2026 | $9–12 | 25% of total market | 30–40 million |
| 2027 | $8–11 | 30% of total market | 35–50 million |
Prices are expected to decline marginally as competition intensifies and manufacturing costs decrease, especially in markets with high generic penetration.
Key Drivers Affecting Price and Market Growth
- Regulatory Approvals: Faster approvals in emerging markets expand potential sales.
- Manufacturing Costs: Potential reductions via economies of scale can lower prices, increasing accessibility.
- Market Penetration: Growth depends on physician adoption, patient awareness, and reimbursement policies.
- Competitive Pricing: Entry of multiple manufacturers could drive prices downward.
Risks to Market and Pricing
- Market saturation in developed countries.
- Price competition from existing generics.
- Regulatory delays in certain regions.
- Regional healthcare policy shifts affecting drug reimbursement.
Conclusion
GNP Olopatadine is poised for growth in a market with expanding demand and broad patent expiries. Prices are likely to decline gradually over five years, with wholesale prices ranging from $8 to $15. The volume sold is expected to increase notably, especially in emerging markets, offsetting lower unit prices.
Key Takeaways
- The global allergic rhinitis and conjunctivitis market grows at a 5% CAGR, driving demand.
- GNP Olopatadine faces stiff competition, especially from generic players.
- Prices are projected to decline from about $12–15 to below $10 per bottle over five years.
- Market penetration hinges on regional regulatory approvals and physician acceptance.
- Economies of scale can further reduce manufacturing costs, increasing competitiveness.
FAQs
-
When is GNP Olopatadine expected to receive regulatory approval?
Likely within 12–18 months per regional processes, depending on the jurisdiction. -
How does the price of GNP Olopatadine compare with branded equivalents?
It is approximately 30–40% lower than branded formulations, with wholesale prices around $8–$15. -
Which regions represent the highest growth potential?
Asia-Pacific and Latin America, due to lower penetration of branded drugs and increasing prevalence of allergies. -
What factors could influence the pricing trajectory?
Competitive market entry, manufacturing cost reductions, regulatory environment changes, and reimbursement policies. -
What are the primary risks for market entry?
Patent protections in certain markets, regulatory delays, and entrenched-brand loyalty among physicians and patients.
References
[1] MarketWatch, “Global Allergic Rhinitis Market Size, Share & Trends,” 2022.
More… ↓
