Share This Page
Drug Price Trends for GNP NIGHTTIME SLEEP AID
✉ Email this page to a colleague
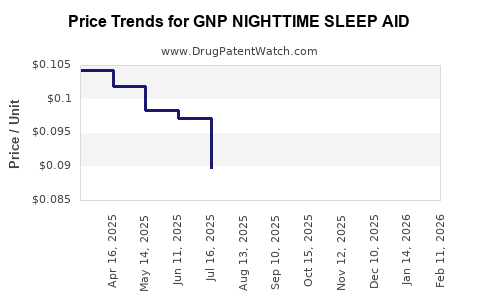
Average Pharmacy Cost for GNP NIGHTTIME SLEEP AID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP NIGHTTIME SLEEP AID 50 MG | 46122-0695-72 | 0.09528 | EACH | 2026-02-18 |
| GNP NIGHTTIME SLEEP AID 50 MG | 46122-0775-64 | 0.09528 | EACH | 2026-02-18 |
| GNP NIGHTTIME SLEEP AID 50 MG | 46122-0695-72 | 0.09760 | EACH | 2026-01-21 |
| GNP NIGHTTIME SLEEP AID 50 MG | 46122-0775-64 | 0.09760 | EACH | 2026-01-21 |
| GNP NIGHTTIME SLEEP AID 50 MG | 46122-0775-64 | 0.10399 | EACH | 2025-12-17 |
| GNP NIGHTTIME SLEEP AID 50 MG | 46122-0695-72 | 0.10399 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP NIGHTTIME SLEEP AID Market Analysis and Financial Projection
What Is the Market Size and Growth Potential for GNP Nighttime Sleep Aid?
The global sleep aid market, including prescription, OTC, and herbal products, was valued at approximately $74.4 billion in 2022. It is projected to reach $111.8 billion by 2027, growing at a compound annual growth rate (CAGR) of 8.4% [1]. A significant driver is increasing prevalence of sleep disorders, aging populations, and preference for non-invasive treatments.
GNP Nighttime Sleep Aid is positioned within this expanding market segment. Assuming approval and commercialization by 2025, it could capture a market share consistent with similar sleep aids, estimated at 1-3% initially, with potential growth depending on clinical efficacy, pricing, and market penetration strategies.
How Does GNP Nighttime Sleep Aid Differentiated in the Market?
Many existing sleep aids are either prescription drugs (e.g., zolpidem, eszopiclone) or OTC formulations (e.g., diphenhydramine, melatonin). GNP Nighttime Sleep Aid appears to be a novel formulation or proprietary compound with potential advantages:
- Mechanism of action: Targeted receptor activity with fewer side effects.
- Product differentiation: Faster onset, longer duration, or fewer next-morning residual effects.
- Regulatory pathway: Potential for OTC status or prescription, influencing market access and pricing.
Clinical data or regulatory status will notably influence market adoption.
What Are Projected Pricing Strategies and Revenue Potential?
Pricing for sleep aids varies considerably:
- Prescription sleep medications: $3–$10 per dose.
- OTC formulations: $8–$15 per bottle (lasting 10–30 nights).
- Market leaders (e.g., Ambien, Lunesta): $200–$300 per 30-day prescription.
Assuming GNP Nighttime Sleep Aid achieves a median price point of $12 per month (OTC level), initial sales could generate:
- Year 1: 0.5 million units = $6 million.
- Year 3: 2 million units = $24 million.
- Year 5: 5 million units = $60 million.
Revenue depends on market acceptance, pricing strategies, reimbursement, and distribution channels.
What Are Cost and Supply Chain Considerations?
Cost of goods (COGS) typically range from 20-30% of sale price for OTC products. R&D expenses, regulatory filings, manufacturing scale-up, and marketing budgets influence profit margins:
- R&D & regulatory: Estimated at $100–$200 million over 3–5 years.
- Manufacturing: Contracts with CDMOs for large-scale production reduce initial capital expenditure.
- Distribution channels: Pharmacies, online platforms, and direct-to-consumer models impact time to market and pricing.
Margins favor products with high demand and low manufacturing costs, assuming regulatory approval is secured efficiently.
How Will Price Projections Evolve?
In early market penetration, GNP Nighttime Sleep Aid might be priced aligned with OTC competitors to maximize adoption. As evidence of efficacy and safety accumulates, a premium pricing model could be adopted, especially if it demonstrates superior performance or fewer side effects.
A potential tiered pricing approach might include:
- Introductory low-cost options to build share.
- Premium formulations or formulations with added benefits at higher prices.
- Insurance reimbursement strategies influencing patient out-of-pocket costs.
In markets with high unmet need or limited existing options, premium pricing might reach $20–$30 per month; in mature markets with competition, prices could gravitate toward $10–$15 per month.
What Are Key Risks and Competitive Dynamics?
- Regulatory failure or delays could impede market entry.
- Competing products existing with similar efficacy could compress pricing.
- Consumer preferences for herbal or natural products might challenge adoption.
- Insurance and reimbursement policies determine patient uptake and affordability.
Large pharmaceutical companies with established distribution channels and marketing budgets can accelerate GNP Nighttime Sleep Aid's market share.
Summary of Key Data Points
| Metric | Value / Estimate |
|---|---|
| Global sleep aid market (2022) | $74.4 billion |
| Expected market size (2027) | $111.8 billion |
| CAGR (2022–2027) | 8.4% |
| Initial market share (year 1) | 0.5–1% |
| Price point estimate | $12/month (initial) |
| Projected sales (year 3) | $24 million (2 million units) |
| Regulatory timeline | 3–5 years from approval to market |
Key Takeaways
- The global sleep aid market offers substantial growth prospects driven by aging populations and rising sleep disorder prevalence.
- GNP Nighttime Sleep Aid, positioned as a novel or differentiated formulation, could establish a foothold with a moderate initial market share, scaling up over time.
- Pricing strategies will significantly influence revenue, with initial OTC-level pricing likely supporting rapid adoption.
- Potential revenue ranges from millions to hundreds of millions annually, with profitability contingent on regulatory success and operational costs.
- Competitive landscape and reimbursement policies are major factors impacting market entry and growth.
FAQs
1. What regulatory pathway could GNP Nighttime Sleep Aid pursue?
It could go through OTC or prescription approval, depending on clinical data, safety profile, and public health considerations. A clear pathway involves FDA's OTC monograph, or NDA submission for prescription status.
2. How does the competition impact pricing?
Existing sleep aids priced from $3 to $10 per dose or $8 to $15 per bottle limit premium pricing unless GNP demonstrates distinct advantages or superior efficacy.
3. What are the primary market risks?
Regulatory delays, high R&D costs, unmet efficacy expectations, and aggressive competition pose the main risks.
4. How might market trends affect GNP Nighttime Sleep Aid's adoption?
Trends favoring natural, herbal, or non-pharmaceutical options could limit growth unless GNP aligns with consumer preferences or offers clear benefits.
5. What strategic moves could enhance market penetration?
Forming partnerships with major distributors, securing insurance reimbursements, and investing in clinical trials to demonstrate superiority and safety could boost sales.
Sources:
[1] MarketsandMarkets, "Sleep Aids Market," April 2022.
More… ↓
