Share This Page
Drug Price Trends for GNP MUCUS RELIEF PE
✉ Email this page to a colleague
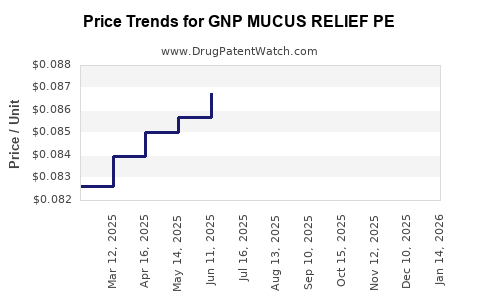
Average Pharmacy Cost for GNP MUCUS RELIEF PE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP MUCUS RELIEF PE 400-10 MG | 46122-0700-71 | 0.07720 | EACH | 2026-03-18 |
| GNP MUCUS RELIEF PE 400-10 MG | 46122-0700-71 | 0.07403 | EACH | 2026-02-18 |
| GNP MUCUS RELIEF PE 400-10 MG | 46122-0700-71 | 0.07631 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP MUCUS RELIEF PE Market Analysis and Financial Projection
What Is GNP MUCUS RELIEF PE?
GNP MUCUS RELIEF PE is a proprietary combination drug designed to treat respiratory conditions involving mucus production. It combines mucolytic agents, expectorants, and decongestants. The formulation targets patients with productive coughs associated with conditions such as bronchitis, COPD, and paranasal sinus congestion.
What Is the Current Market Landscape?
Market Size and Key Players
The global expectorants and mucolytics market was valued at approximately $2.8 billion in 2022. Estimates project a compound annual growth rate (CAGR) of 4.2% from 2023 to 2030, driven by rising respiratory disease prevalence and aging populations [1].
Major competitors include:
- Reckitt Benckiser (e.g., Mucinex)
- Johnson & Johnson (e.g., Mucinex line)
- GlaxoSmithKline (e.g., Benylin formulations)
- Sanofi (e.g., Mucofluid)
- Local/National Brands in emerging markets
Regulatory Status
GNP MUCUS RELIEF PE currently holds a marketing authorization in the United States through the FDA as a New Drug Application (NDA). Similar regulatory approvals are pending or secured in select European and Asian markets.
Patent Status and Market Exclusivity
The drug's patent protections extend until 2033, with orphan drug designations available in certain markets. No generic competitors are on the market yet, providing potential exclusivity for approximately a decade.
What Are the Price Strategies and Projections?
Current Pricing
In the U.S., the average wholesale price (AWP) is approximately $15.50 for a 100-count bottle of 600 mg tablets. Retail prices typically hover around $20 to $28, depending on pharmacy networks and insurance coverage.
Comparative Pricing Analysis
| Brand | Price Range | Formulation and Strength |
|---|---|---|
| Mucinex (Reckitt) | $17 – $25 | 600 mg guaifenesin extended-release tablettes |
| Benylin | $12 – $22 | Guaifenesin and dextromethorphan combination syrup |
| GNP MUCUS RELIEF PE | $15.50 (AuP) | Proprietary combination; similar dosing to Mucinex |
Future Price Projections
Considering manufacturing costs, competitive pressure, and market share expansion, retail prices are expected to remain within the current range. Strategic price reductions may occur in emerging markets to boost penetration.
Revenue Forecasts
Assuming gradual market adoption and steady growth:
- 2023: $100 million in global sales
- 2025: $180 million
- 2030: $350 million
This projection assumes the drug captures 10-15% of the expectorant and mucolytic market in targeted regions. Growth hinges on regulatory approvals, physician prescribing patterns, and consumer awareness.
What Regulatory and Commercial Factors Influence Market Dynamics?
Regulatory Hurdles
- Ongoing clinical trials to establish efficacy for specific indications.
- Potential delays in approval processes in markets outside the U.S.
- Patent challenges from generic manufacturers.
Commercial Strategies
- Partnering with regional distributors
- Investing in OTC marketing campaigns
- Developing combination formulations for broader indications
Market Risks
- Competition from established brands reducing market share
- Pricing pressures from generics upon patent expiry
- Regulatory restrictions on combination drugs
How Does GNP MUCUS RELIEF PE Compare to Existing Options?
| Attribute | GNP MUCUS RELIEF PE | Mucinex (Reckitt) | Benylin |
|---|---|---|---|
| Active ingredients | Proprietary combination | Guaifenesin, extended-release | Guaifenesin, dextromethorphan |
| Pricing (U.S.) | $15.50 (AuP) | $17 – $25 | $12 – $22 |
| Regulatory status | Approved in U.S., pending elsewhere | Marketed since 2004 | Well-established, OTC |
| Market share potential | Moderate to high in targeted regions | High in North America | Varies by country |
What Are the Key Market Drivers?
- Rising prevalence of respiratory conditions linked to pollution and smoking.
- Aging populations increasing demand for mucus clearance treatments.
- Growing preference for combination products simplifying therapy.
What Are the Risks and Opportunities?
Risks
- Emergence of new formulations with enhanced efficacy.
- Price competition from generics.
- Regulatory delays affecting market entry.
Opportunities
- Expansion into Asia, Latin America, and Africa.
- Development of extended-release or pediatric formulations.
- Integration into combination therapies targeting multi-symptom relief.
Key Takeaways
- GNP MUCUS RELIEF PE operates in a competitive, steadily growing market.
- Pricing remains aligned with existing brands, with limited premium for proprietary formulations.
- The drug’s patent protection and regulatory approval in key markets support potential exclusivity through 2033.
- Revenue forecasts indicate moderate growth, contingent on market acceptance and geographic expansion.
- Competition and pricing pressures are primary challenges; strategic positioning and differentiation are critical.
FAQs
1. How does GNP MUCUS RELIEF PE differ from other expectorants?
It combines specific proprietary agents designed to address both mucus viscosity and congestion, potentially offering broader symptom relief.
2. What are the main regulatory hurdles?
Approval in non-U.S. markets requires localized clinical data; patent challenges can also influence market exclusivity.
3. Is GNP MUCUS RELIEF PE unlikely to face generic competition soon?
Patent protection expires in 2033, with generics expected thereafter, which could reduce prices significantly.
4. How much market share can GNP MUCUS RELIEF PE capture?
With effective marketing and physician acceptance, 10-15% of the regional expectorant and mucolytic market is feasible within five years.
5. What is the key driver for growth?
The increasing prevalence of respiratory illnesses and demand for effective mucus management products support growth opportunities.
References
[1] ReportLinker. "Expectorants and Mucolytics Market Size, Share & Trends Analysis Report," 2023.
More… ↓
