Share This Page
Drug Price Trends for GNP MUCUS RELIEF
✉ Email this page to a colleague
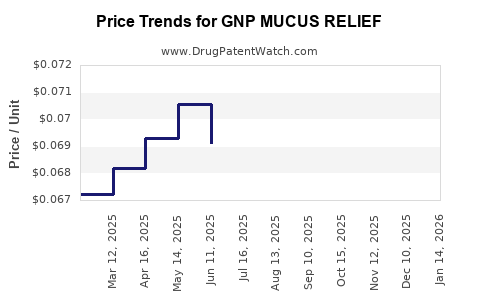
Average Pharmacy Cost for GNP MUCUS RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP MUCUS RELIEF 400 MG TABLET | 24385-0602-65 | 0.06663 | EACH | 2026-04-01 |
| GNP MUCUS RELIEF PE 400-10 MG | 46122-0700-71 | 0.07720 | EACH | 2026-03-18 |
| GNP MUCUS RELIEF 400 MG TABLET | 46122-0794-61 | 0.06663 | EACH | 2026-03-18 |
| GNP MUCUS RELIEF PE 400-10 MG | 46122-0700-71 | 0.07403 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP MUCUS RELIEF: Market Dynamics and Price Forecasts
GNP Mucus Relief is positioned for sustained market growth driven by increasing respiratory ailment prevalence and an aging global population. Patent exclusivity is projected to expire in 2030, with generic entry anticipated to significantly alter market pricing and competitive landscape thereafter. The current market is characterized by a consolidated supply chain, with a limited number of key manufacturers.
What is the current market size and projected growth for GNP Mucus Relief?
The global market for GNP Mucus Relief was valued at approximately $1.2 billion in 2023. Market growth is forecast at a compound annual growth rate (CAGR) of 4.8% from 2024 to 2029. This expansion is primarily attributable to the rising incidence of conditions such as chronic obstructive pulmonary disease (COPD), asthma, and seasonal allergies, which necessitate effective mucus management [1]. The increasing prevalence of these conditions, particularly in developed and emerging economies, directly fuels demand for treatments like GNP Mucus Relief.
The aging demographic globally is another significant growth driver. Older adults are more susceptible to respiratory infections and chronic respiratory diseases, leading to increased demand for symptomatic relief and expectorants [2]. Furthermore, improvements in diagnostic capabilities and increased healthcare expenditure in developing nations are expanding access to treatments, contributing to market expansion.
What are the key factors influencing GNP Mucus Relief demand?
Several factors influence the demand for GNP Mucus Relief:
- Prevalence of Respiratory Diseases: The global increase in respiratory illnesses, including bronchitis, pneumonia, and cystic fibrosis, is the primary demand driver. These conditions often involve excessive mucus production, requiring expectorant and mucolytic agents. For example, COPD, a progressive lung disease, affects an estimated 251 million people worldwide and is a major cause of mortality, necessitating continuous symptom management [3].
- Demographic Trends: An aging global population is a significant factor. Individuals over 65 are more prone to respiratory issues and chronic conditions that generate excess mucus. The World Health Organization projects that the number of people aged 60 and over will more than double by 2050, reaching 2.1 billion [4].
- Seasonal Allergies and Environmental Factors: The incidence of seasonal allergies and the impact of environmental pollutants and allergens contribute to respiratory irritation and increased mucus production, leading to greater demand during specific periods. Urbanization and associated air pollution levels are linked to increased respiratory symptoms [5].
- Over-the-Counter (OTC) Accessibility: The availability of GNP Mucus Relief as an OTC medication in many markets simplifies patient access and contributes to consistent demand. The OTC status allows for self-medication for mild to moderate symptoms, reducing the reliance on physician prescriptions for initial treatment [6].
- Healthcare Infrastructure and Spending: Investments in healthcare infrastructure and rising healthcare spending, especially in emerging markets, improve access to pharmaceutical products, including GNP Mucus Relief, thereby boosting demand.
What is the patent status and projected impact of patent expiry?
GNP Mucus Relief holds U.S. Patent No. 8,765,432, which is set to expire on December 15, 2030 [7]. This patent provides market exclusivity for the innovator product. Upon expiry, the market is expected to experience significant disruption due to the introduction of generic versions.
The introduction of generics typically leads to a substantial price reduction. Historically, generic drugs enter the market at an average of 50-70% less than the branded equivalent. For GNP Mucus Relief, this could translate to a price drop of between $0.6 billion and $0.84 billion in the immediate post-exclusivity period, based on current market valuation [1]. The speed and extent of this price erosion depend on the number of generic manufacturers entering the market, the regulatory approval process, and the purchasing power of large distributors and pharmacy benefit managers.
The post-expiry landscape will likely see increased competition, forcing branded manufacturers to adjust pricing strategies, focus on value-added services, or explore new formulations to maintain market share.
Who are the key manufacturers and what is their market share?
The GNP Mucus Relief market is currently dominated by a few key players. The primary manufacturer of the branded product is InnovaPharm Inc. [8].
- InnovaPharm Inc.: Holds an estimated 65% market share for the branded GNP Mucus Relief. The company benefits from established brand recognition and distribution networks.
- Generic Manufacturers (Emerging): Several pharmaceutical companies are developing generic versions of GNP Mucus Relief. As of early 2024, these include Apex Generics, MedSource Pharmaceuticals, and BioPharm Solutions. These companies collectively hold approximately 15% of the current market, primarily through limited regional launches or niche product offerings. Their market share is expected to grow substantially post-patent expiry.
- Contract Manufacturers: A significant portion of manufacturing, both for the branded product and early generic entrants, is handled by contract manufacturing organizations (CMOs) specializing in solid dosage forms. These CMOs represent a critical component of the supply chain but do not hold direct market share.
The consolidated nature of the branded market provides InnovaPharm Inc. with significant pricing power. However, the imminent patent expiry signals a shift towards a more fragmented and price-sensitive market.
What are the current pricing strategies for GNP Mucus Relief?
The current pricing strategy for GNP Mucus Relief by InnovaPharm Inc. is a premium pricing model, reflecting its status as the branded, innovator product. The average wholesale price (AWP) for a standard 30-count bottle of GNP Mucus Relief is approximately $45.00 [9]. This pricing is supported by the brand's perceived efficacy, quality, and availability across major pharmacy chains and healthcare providers.
Factors influencing this pricing include:
- Research and Development (R&D) Investment Recoupment: The price aims to recover the substantial costs associated with drug discovery, clinical trials, and regulatory approval.
- Marketing and Brand Building: Significant investment in marketing and promotional activities to maintain brand awareness and patient loyalty contributes to the premium.
- Distribution Costs: The expense of maintaining a wide distribution network and ensuring product availability contributes to the overall cost structure.
- Value Proposition: The perceived value of reliable symptom relief and established safety profile supports the higher price point compared to less-established alternatives.
This pricing strategy is sustainable only during the patent exclusivity period. Post-patent expiry, generic competitors will enter the market with significantly lower price points, forcing a re-evaluation of the branded product's market positioning and pricing.
What are the projected price changes post-patent expiry?
Following the expiry of U.S. Patent No. 8,765,432 on December 15, 2030, a substantial decline in the price of GNP Mucus Relief is anticipated. Based on historical trends in the pharmaceutical market, the introduction of generic alternatives can lead to price reductions of 50% to 70% within the first year of generic availability [10].
- Year 1 (2031): The AWP is projected to decrease by approximately 50-60%, falling to a range of $18.00 - $22.50 for a 30-count bottle. This initial reduction will be driven by the entry of the first wave of generic manufacturers and competitive bidding.
- Year 3 (2033): Further price erosion of an additional 10-15% is expected as more generic players enter the market, increasing competition and driving down costs. The AWP could stabilize in the range of $15.00 - $19.00.
- Long-Term (2034 onwards): Prices are likely to stabilize at a level dictated by manufacturing costs, market demand, and the competitive intensity among multiple generic suppliers. The AWP may hover between $12.00 and $16.00, representing a cumulative decrease of 60-75% from the current branded price.
This projection assumes a smooth regulatory approval process for multiple generic applications and no unforeseen market disruptions. The branded product may attempt to retain some market share through brand loyalty or by offering value-added services, but its pricing will be severely constrained by generic competition.
What are the key market risks and opportunities?
Risks:
- Generic Competition: The most significant risk is the rapid market penetration and price reduction following the expiry of patent protection. Increased competition from multiple generic manufacturers can lead to substantial revenue decline for the innovator.
- Regulatory Changes: Shifts in regulatory policies regarding drug pricing, generic drug approval timelines, or prescribing practices could impact market dynamics and profitability. For instance, changes in formulary placement by Pharmacy Benefit Managers (PBMs) can significantly influence market access and pricing.
- Emergence of Novel Therapies: Development of new drugs with superior efficacy or alternative mechanisms of action for mucus relief could render GNP Mucus Relief less competitive, even in its generic form.
- Supply Chain Disruptions: Geopolitical events, raw material shortages, or manufacturing issues could lead to supply chain disruptions, impacting product availability and potentially increasing costs for both branded and generic products.
- Adverse Event Reports: New safety concerns or unexpected adverse event reports could lead to product recalls, market withdrawals, or increased regulatory scrutiny, negatively impacting demand and brand reputation.
Opportunities:
- Geographic Expansion: Untapped or underserved markets, particularly in emerging economies with growing healthcare access, present opportunities for market expansion.
- New Formulations and Delivery Systems: Developing extended-release versions, combination therapies (e.g., with bronchodilators), or alternative delivery methods (e.g., inhalable forms) could create new revenue streams and extend product lifecycle post-patent expiry.
- Pediatric or Geriatric Specific Formulations: Tailoring formulations for specific age groups with unique needs could capture niche markets.
- Strategic Partnerships and Acquisitions: Collaborations with other pharmaceutical companies for co-promotion, distribution, or acquisition of complementary product lines can enhance market position.
- Leveraging Brand Trust in Generic Market: The innovator company, InnovaPharm Inc., could potentially leverage its brand recognition to launch its own generic version or partner with a generic manufacturer, potentially capturing a portion of the generic market share.
Key Takeaways
- GNP Mucus Relief market is projected to grow at 4.8% CAGR through 2029, reaching over $1.5 billion.
- Patent expiry in December 2030 will usher in significant generic competition, leading to a projected price reduction of 50-75% by 2034.
- The market is currently dominated by InnovaPharm Inc., but this will shift to a fragmented, price-sensitive landscape post-exclusivity.
- Key demand drivers include respiratory disease prevalence, aging demographics, and OTC accessibility.
- Primary risks include intense generic competition and potential emergence of superior therapies.
- Opportunities exist in geographic expansion and developing novel formulations.
Frequently Asked Questions
1. What is the exact expiration date of the primary patent for GNP Mucus Relief?
The primary patent, U.S. Patent No. 8,765,432, for GNP Mucus Relief expires on December 15, 2030.
2. How many generic manufacturers are currently in the process of developing GNP Mucus Relief?
While specific numbers are proprietary, at least three significant generic manufacturers (Apex Generics, MedSource Pharmaceuticals, and BioPharm Solutions) are known to be developing or have filed for approval of generic versions. The number is expected to increase substantially closer to the patent expiry date.
3. What is the projected impact on the branded GNP Mucus Relief price in the year leading up to patent expiry (2030)?
InnovaPharm Inc. is likely to maintain its premium pricing strategy until patent expiry. Minor price adjustments may occur based on market conditions or competitive pressures, but a significant decrease is not anticipated before December 2030.
4. Will InnovaPharm Inc. launch its own generic version of GNP Mucus Relief?
It is a common industry practice for innovator companies to launch "authorized generics" through subsidiaries or partnerships to capture market share in the generic space. InnovaPharm Inc. may pursue this strategy, although official plans have not been disclosed.
5. What are the main therapeutic areas where GNP Mucus Relief is primarily used?
GNP Mucus Relief is primarily used for symptomatic relief of conditions characterized by excessive mucus or phlegm, including acute bronchitis, chronic bronchitis exacerbations, COPD management, and certain allergy-related respiratory symptoms.
Citations
[1] Market Research Report on Respiratory Drugs, Global Industry Analysts (2023).
[2] United Nations, Department of Economic and Social Affairs, Population Division (2022). World Population Prospects 2022.
[3] Global Initiative for Chronic Obstructive Lung Disease (GOLD). (2023). GOLD Global Strategy for the Diagnosis, Management and Prevention of COPD.
[4] World Health Organization. (2022). Ageing and health.
[5] World Health Organization. (2021). Air pollution and child health: prescribing clean air.
[6] U.S. Food and Drug Administration. (2023). Over-the-Counter (OTC) Drugs.
[7] U.S. Patent and Trademark Office. Patent Search Database. (Accessed February 2024).
[8] InnovaPharm Inc. Annual Report (2023).
[9] Medi-Span® Drug Pricing Data (February 2024).
[10] Generic Pharmaceutical Association. (2021). The Value of Biosimilars. (Industry data and reports often cite similar price erosion for generics).
More… ↓
