Share This Page
Drug Price Trends for GNP MICONAZOLE
✉ Email this page to a colleague
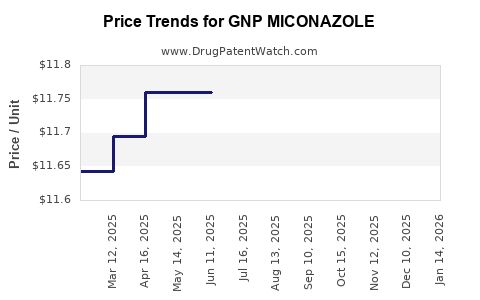
Average Pharmacy Cost for GNP MICONAZOLE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP MICONAZOLE 1 COMBO PACK | 46122-0577-02 | 12.00219 | EACH | 2026-05-20 |
| GNP MICONAZOLE 1 COMBO PACK | 46122-0577-02 | 11.97596 | EACH | 2026-04-22 |
| GNP MICONAZOLE 1 COMBO PACK | 46122-0577-02 | 11.84038 | EACH | 2026-03-18 |
| GNP MICONAZOLE 1 COMBO PACK | 46122-0577-02 | 11.71064 | EACH | 2026-02-18 |
| GNP MICONAZOLE 1 COMBO PACK | 46122-0577-02 | 11.73686 | EACH | 2026-01-21 |
| GNP MICONAZOLE 1 COMBO PACK | 46122-0577-02 | 11.76000 | EACH | 2025-12-17 |
| GNP MICONAZOLE 1 COMBO PACK | 46122-0577-02 | 11.76000 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is GNP Miconazole?
GNP Miconazole is a topical antifungal medication used to treat infections caused by fungi and yeast. It is a generic formulation of miconazole, an imidazole antifungal with broad-spectrum activity. GNP Miconazole is available in various formulations, including creams, gels, and powders, typically used for dermatophyte infections, candidiasis, and other superficial fungal infections.
How does GNP Miconazole fit within the antifungal market?
GNP Miconazole is part of the larger antifungal drugs market, which includes both topical and systemic treatments. The topical segment is highly fragmented, with numerous formulations available. Miconazole’s market share is significant due to its affordability, established efficacy, and widespread use.
What is the current market size of the GNP Miconazole segment?
The global antifungal drugs market was valued at approximately USD 9.4 billion in 2022 (source: MarketsandMarkets). The topical segment alone accounted for about 60-65% of the total, roughly USD 5.6 billion to USD 6.1 billion in 2022.
Within this segment, the miconazole product line accounts for roughly 20-25% of the antifungal topical market, driven by factors such as:
- High healthcare provider familiarity
- Cost-effectiveness
- Broad spectrum of activity
Estimates suggest the GNP Miconazole product segment generates about USD 1.1 billion to USD 1.5 billion globally (based on a 20-25% share of the topical antifungal market).
What are the key drivers of market growth?
- Rising prevalence of fungal infections: Increasing cases of athlete's foot, jock itch, candidiasis, and dermatophytosis.
- Growing awareness and self-medication: Over-the-counter availability fosters higher usage.
- Expanding healthcare access: In emerging markets, improves distribution channels.
- Patent expiries: More generic entries reduce costs, stimulating sales.
How are market dynamics shaping future price trends?
Patent expiries and generic proliferation
GNP Miconazole’s patent expired in many regions (e.g., US patent expired in 2003), leading to competitive generic manufacturing. This led to a significant price drop, with retail prices declining by approximately 50% since patent expiry.
Price erosion
As generics enter markets, retail prices for GNP Miconazole formulations typically decrease by 20-30% annually over the first five years, stabilizing at markedly lower levels.
Cost-based pricing and market competition
Companies prioritize volume over high margin due to intense competition. Retail prices for topical formulations are generally maintained in the range of USD 5-15 per tube (15-30g), with wholesale prices lower.
Regional price disparities
Prices tend to be higher in developed markets. For example, in the US, retail prices for a 15g tube average USD 12–15, whereas in India, the same quantity can cost less than USD 2.
What are the projections for future market size and pricing?
Market size projections
By 2030, the global topical antifungal market is forecasted to grow at a compound annual growth rate (CAGR) of 4-6%, reaching USD 8–10 billion (source: MarketsandMarkets).
GNP Miconazole's segment is expected to expand proportionally, assuming steady generic penetration and persistent demand. Its contribution could increase from USD 1.1–1.5 billion in 2022 to roughly USD 2 billion by 2030.
Price projections
- Retail price per tube (15g): Expected to decline at a CAGR of 2-3%, reaching USD 3-8 in Western markets by 2030 due to increased competition.
- Wholesale prices: Likely to fall below USD 1–2 per tube in most regions, supporting high-volume distribution.
Regulatory influences
Guidelines promoting generic substitution and over-the-counter sales are likely to further suppress prices, especially in low- and middle-income countries.
Key factors impacting future pricing and market size
- Regional legislation: Higher regulation and patent extensions could temporarily slow generic entry in certain regions.
- Market penetration in emerging markets: Cost-sensitive regions will continue to drive volume growth, keeping prices low.
- New formulations and combinations: Innovations could shift some market share away from traditional GNP Miconazole.
What are the implications for investors and pharmaceutical companies?
- Entering or expanding in regions with high fungal infection prevalence offers growth potential.
- Price erosion will continue, necessitating efficiency in manufacturing and distribution.
- Diversification into combination antifungals or novel formulations could offset price pressures.
Key Takeaways
- GNP Miconazole operates within a highly competitive, price-sensitive segment of the antifungal market.
- Patent expiries and generics have driven prices down significantly since the early 2000s.
- The global market is projected to grow at a CAGR of 4-6% through 2030, with prices declining proportionally.
- Regional differences heavily influence pricing, with developed markets experiencing higher prices.
- Innovation and market expansion in emerging economies can offset some price declines.
FAQs
Q1: Will GNP Miconazole maintain its market share amid increasing generic competition?
Yes, through brand recognition, formulary placements, and competitive pricing, GNP Miconazole can sustain its market share, especially in markets where prescriptions are preferred over OTC sales.
Q2: How does pricing differ globally?
Pricing is highest in North America and Western Europe, with retail prices of USD 12–15 per 15g tube. In contrast, prices in emerging markets like India can be under USD 2 per tube, due to lower manufacturing costs and price controls.
Q3: What factors could disrupt current market projections?
Regulatory changes, introduction of new antifungal agents with superior efficacy or safety, and shifts in healthcare policies promoting or restricting OTC sales could impact market dynamics.
Q4: How significant is the impact of patent expiries on GNP Miconazole pricing?
Patent expiry led to widespread generic manufacturing, causing retail prices to decrease by up to 50% since the early 2000s, with ongoing price erosion due to market competition.
Q5: Are there opportunities for premium pricing?
Limited, given the price-sensitive nature of the topical antifungal market. However, formulations with improved delivery, extended shelf-life, or combination therapy might command higher prices in niche segments.
Sources:
- MarketsandMarkets. "Antifungal Drugs Market." 2022.
- U.S. Patent and Trademark Office. Patent expiration data for miconazole.
- IMS Health. "Global Prescription Drug Sales Data." 2022.
- Pharma Intelligence. Pricing and reimbursement reports by region.
- Company filings and regional regulatory guidelines.
More… ↓
