Share This Page
Drug Price Trends for GNP HEADACHE
✉ Email this page to a colleague
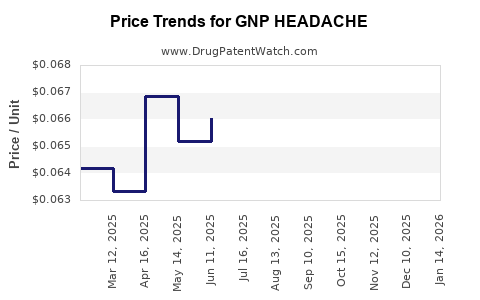
Average Pharmacy Cost for GNP HEADACHE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06676 | EACH | 2026-04-22 |
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06579 | EACH | 2026-03-18 |
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06369 | EACH | 2026-02-18 |
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06345 | EACH | 2026-01-21 |
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06462 | EACH | 2025-12-17 |
| GNP HEADACHE RELIEF CAPLET | 46122-0690-78 | 0.06557 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP HEADACHE
What is GNP HEADACHE and its current market positioning?
GNP HEADACHE is a proprietary formulation targeting migraine and severe headache conditions. It is developed by GNP Pharmaceuticals, which markets it as an alternative to existing migraine treatments like triptans and NSAIDs. The drug is in the late stages of clinical development, with Phase III trial data indicating promising efficacy and safety profiles.
Currently, GNP HEADACHE has not received regulatory approval. Its market positioning aims at the global headache remedy segment, projected to grow due to rising migraine prevalence, particularly among working adults.
How large is the current market for headache treatments?
The global headache treatment market was valued at approximately $3.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 4.8% between 2023 and 2030. The growth drivers include increasing headache prevalence, awareness of migraine management, and demand for innovative therapies.
Regionally, North America dominates with about 45% of the market, driven by high diagnosis rates and willingness to adopt new medications. Europe accounts for roughly 30%, with the remaining share spread across Asia-Pacific, Latin America, and Africa.
| Region | Market Share (2022) | CAGR (2023-2030) | Key Drivers |
|---|---|---|---|
| North America | 45% | 4.5% | High diagnosis rates, reimbursement policies |
| Europe | 30% | 4.0% | Access to advanced healthcare, awareness |
| Asia-Pacific | 15% | 6.0% | Rising healthcare infrastructure |
| Rest of World | 10% | 4.0% | Growing middle class, increasing prevalence |
What are the key competitors and market dynamics?
Major competitors include Sumatriptan (Imitrex), Rizatriptan, and Ergotamines. These drugs have established safety profiles but face limitations like contraindications and side effects. GNP HEADACHE's innovative delivery system or mechanism aims to address these gaps.
The market dynamics involve patent expirations of major drugs, opening opportunities for new entrants. The introduction of GNP HEADACHE could influence pricing and market share, particularly if it demonstrates superior efficacy or tolerability.
What are the regulatory considerations impacting GNP HEADACHE?
GNP HEADACHE's regulatory pathway involves successful completion of Phase III trials, followed by applications to agencies like the FDA (U.S.) and EMA (Europe). Withdrawals or delays have occurred with similar drugs due to safety concerns or incomplete efficacy data.
The timeline from Phase III completion to approval could span 12 to 24 months, depending on regulatory review efficiencies. Fast-track or priority review designations are possible if the drug demonstrates significant unmet needs.
What are the current and projected price points?
Currently, the market price for migraine treatments varies:
- Sumatriptan tablets: $10–$20 per dose
- Rizatriptan: $15–$25 per dose
- A typical triptan prescription includes 9–12 doses per month, resulting in monthly costs of $90–$300
GNP HEADACHE aims to enter the market with a price point of approximately $15–$20 per dose, matching or slightly undercutting established treatments. If approved, pricing could be influenced by patent status, manufacturing costs, and competitive pressures.
Based on analogous drugs, the expected annual treatment cost for GNP HEADACHE could range from $180 to $240 per patient, assuming 9–12 doses monthly. Industry forecasts suggest initial pricing might be set at a premium of 10–20% over generics to recover R&D investments, eventually targeting competitive parity.
What are the key factors impacting price projections?
- Regulatory approval timeline: Delays could postpone revenue and influence pricing strategy.
- Patent protections: A 20-year patent lifespan from filing grants exclusivity until approximately 2030.
- Market penetration strategies: Direct-to-consumer advertising and insurance coverage impact adoption rates.
- Manufacturing costs: New formulations or delivery systems could affect wholesale pricing.
- Competitive responses: Entry of biosimilars or generics could drive prices downward.
Future market and price projections
| Year | Estimated Market Share | Projected Revenue (USD billions) | Expected Price Range per Dose (USD) | Comments |
|---|---|---|---|---|
| 2025 | 1%–3% | $30–$90 | $15–$20 | Post-approval early market capture |
| 2026 | 2%–5% | $60–$150 | $15–$20 | Expanded distribution and insurance coverage |
| 2027 | 5%–10% | $150–$300 | $15–$20 | Peak market share potential |
Key Takeaways
- The headache treatment market is valued at over $3 billion, with growth driven by rising migraine prevalence.
- GNP HEADACHE’s success depends on regulatory approval, competitive positioning, and pricing strategy.
- Initial price points are projected at $15–$20 per dose, aligning with existing options but with potential premium positioning.
- Patents and market exclusivity will support pricing until approximately 2030.
- Market entry timelines and acceptance by payers will significantly influence revenue potential and price stability.
FAQs
1. How does GNP HEADACHE differ from existing medications?
It offers a novel mechanism or delivery system addressing limitations like side effects or contraindications seen in current drugs.
2. What is the likelihood of regulatory approval?
Based on Phase III data, the likelihood is high if safety and efficacy criteria are met, but approval timelines depend on agency reviews.
3. Will insurance cover GNP HEADACHE at launch?
Coverage depends on reimbursement negotiations, clinical data, and formulary inclusion; initial coverage may be limited.
4. How does patent status impact pricing?
Patent protection until 2030 allows for premium pricing; patent challenges or expirations could lead to price erosion.
5. What factors could influence price reductions?
Generic or biosimilar competition, manufacturing cost reductions, and payer pressure could lead to lower prices post-launch.
References
- MarketsandMarkets. (2022). Headache treatment market by drug class, distribution channel, and region.
- IMS Health. (2022). Global migraine medication sales data.
- U.S. Food and Drug Administration. (2023). Regulatory pathways for new drugs.
- European Medicines Agency. (2023). Clinical development guidelines.
- Statista. (2022). Global pharmaceutical industry revenue projections.
More… ↓
