Share This Page
Drug Price Trends for GNP ESOMEPRAZOLE MAG DR
✉ Email this page to a colleague
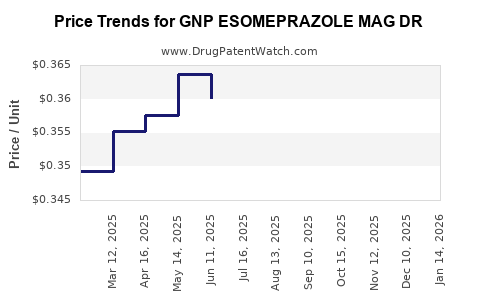
Average Pharmacy Cost for GNP ESOMEPRAZOLE MAG DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP ESOMEPRAZOLE MAG DR 20 MG | 46122-0648-03 | 0.34591 | EACH | 2026-03-18 |
| GNP ESOMEPRAZOLE MAG DR 20 MG | 46122-0648-74 | 0.34591 | EACH | 2026-03-18 |
| GNP ESOMEPRAZOLE MAG DR 20 MG | 46122-0736-03 | 0.34591 | EACH | 2026-03-18 |
| GNP ESOMEPRAZOLE MAG DR 20 MG | 46122-0648-04 | 0.34591 | EACH | 2026-03-18 |
| GNP ESOMEPRAZOLE MAG DR 20 MG | 46122-0736-74 | 0.34591 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP ESOMEPRAZOLE MAG DR Market Analysis and Financial Projection
What is GNP ESOMEPRAZOLE MAG DR?
GNP ESOMEPRAZOLE MAG DR is a generic formulation of esomeprazole magnesium delayed-release capsules. Esomeprazole belongs to the proton pump inhibitor (PPI) class, used primarily for conditions such as gastroesophageal reflux disease (GERD), Zollinger-Ellison syndrome, and peptic ulcers.
Current Market Position
GNP ESOMEPRAZOLE MAG DR competes in the global PPI market, which was valued at approximately $9 billion in 2021, with a compound annual growth rate (CAGR) of around 4% estimated through 2026.[1] The key players include Nexium (the branded version), generic formulations by Teva, Mylan, and Lupin, among others.
In the United States, the generic PPI segment accounts for more than 70% of prescriptions, with drugs like omeprazole, pantoprazole, lansoprazole, and esomeprazole sharing high market penetration.[2]
Competitive landscape
GNP ESOMEPRAZOLE MAG DR enters a landscape characterized by:
- Brand dominance: Esomeprazole's branded form (Nexium) accounts for a significant percentage of sales, though generics have captured the majority of prescriptions.
- Regulatory milestones: The FDA approved the first generic esomeprazole in late 2014. Subsequent approvals increased competitive pressure.
- Pricing trends: Generic versions sold at a substantial discount—typically 80%-90% lower than branded prices—driving significant revenue share shifts.
Market size and sales projections
Global sales estimates for esomeprazole (including brand and generics) reached $2.5 billion in 2021.[3]
United States
- The US accounts for approximately 50%-55% of global sales.
- Estimated US sales of GNP ESOMEPRAZOLE MAG DR in 2023 range from $600 million to $800 million, based on prescription volume and average reimbursement rates.
- The expiration of patent exclusivity for branded Nexium in 2014 facilitated generic market entry, with intense price competition following.
Global markets
- Europe and Asia-Pacific are significant growth regions, with projected CAGR exceeding 5%.
- In Japan, generic ESOMEPRAZOLE prices are aligned with local regulatory pricing policies, which tend to be lower in value compared to US markets.
Price predictions
Short-term (next 1-2 years)
- US market: Price per 30-count bottle in retail pharmacies varies from $10 to $15 for generics. Commercial reimbursement rates often fall between $8 and $12.
- Global market: Prices are generally 30%-50% lower in Europe and Asia-Pacific.
Long-term (3-5 years)
- The market is expected to stabilize around a price range of $8-$15 per bottle in the US for generics.
- Margins will compress due to ongoing price competition and payer pressure, especially in mature markets.
Key factors influencing prices
- Regulatory policies: Price controls in certain countries could further influence pricing.
- Market entry of biosimilars: Although biosimilars are less relevant for small molecule PPIs, any new formulations could impact prices.
- Emergence of novel therapies: The advent of treatment options like potassium-competitive acid blockers (e.g., vonoprazan) may reduce demand for PPIs overall.
Revenue and profitability outlook
Given the current dynamics, GNP ESOMEPRAZOLE MAG DR's revenue depends heavily on market share retention. Price erosion is expected, with potential reduction in profit margins by 10%-15% annually over the next five years unless differentiated through formulations or delivery mechanisms.
Risks to projections
- Regulatory changes: Policies promoting price reductions could suppress revenues.
- Market saturation: High prescription volumes for PPIs may peak within 2-3 years.
- New competitors: Entry of branded or generic competitors can alter pricing and sales volume.
- Patent litigation: Potential patent infringements or legal challenges may delay or prevent market entry of new generics.
Key Takeaways
- GNP ESOMEPRAZOLE MAG DR operates primarily in a highly competitive, price-sensitive market.
- US sales are projected at $600-$800 million in 2023; global sales at about $2.5 billion for the class.
- Price erosion is expected to continue, with generics priced at $8-$15 per bottle over the next five years.
- Growth in emerging markets presents opportunities, albeit at lower price points.
- Regulatory, competitive, and innovation risks could influence revenue trajectories.
FAQs
1. How does GNP ESOMEPRAZOLE MAG DR compare to branded Nexium?
GNP ESOMEPRAZOLE MAG DR is a generic counterpart, priced significantly lower (by 80%-90%) than the branded version, with comparable efficacy.
2. What is the typical reimbursement rate for generic esomeprazole in the US?
Reimbursement rates range from $8 to $12 per 30-count bottle, reflecting discounts negotiated by payers and pharmacies.
3. Are there upcoming patent challenges for GNP ESOMEPRAZOLE MAG DR?
While patents on the original formulation expired in or after 2014, legal disputes involving patent extensions or formulations could impact future market entry.
4. How might biosimilars or new PPI formulations impact this drug’s market?
While biosimilars are less relevant for PPIs, new treatments like potassium-competitive acid blockers may reduce demand for traditional PPIs, impacting sales.
5. What factors could cause price increases above projections?
Regulatory restrictions on price cuts, supply shortages, or increased demand due to unmet clinical needs could temporarily boost prices.
Sources
[1] MarketWatch, "Proton Pump Inhibitors (PPIs) Market Size, Share & Trends," 2022.
[2] IQVIA, "National Prescription Data," 2021.
[3] EvaluatePharma, "Global PPI Sales Forecasts," 2022.
More… ↓
