Share This Page
Drug Price Trends for GNP DICLOFENAC SODIUM
✉ Email this page to a colleague
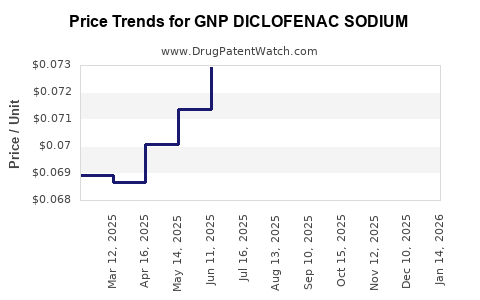
Average Pharmacy Cost for GNP DICLOFENAC SODIUM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP DICLOFENAC SODIUM 1% GEL | 46122-0752-37 | 0.12634 | GM | 2026-03-18 |
| GNP DICLOFENAC SODIUM 1% GEL | 46122-0752-52 | 0.08983 | GM | 2026-03-18 |
| GNP DICLOFENAC SODIUM 1% GEL | 46122-0752-53 | 0.06890 | GM | 2026-03-18 |
| GNP DICLOFENAC SODIUM 1% GEL | 46122-0752-37 | 0.12709 | GM | 2026-02-18 |
| GNP DICLOFENAC SODIUM 1% GEL | 46122-0752-53 | 0.06922 | GM | 2026-02-18 |
| GNP DICLOFENAC SODIUM 1% GEL | 46122-0752-52 | 0.09149 | GM | 2026-02-18 |
| GNP DICLOFENAC SODIUM 1% GEL | 46122-0752-53 | 0.06923 | GM | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Diclofenac Sodium Generics: Market Dynamics and Price Forecast
Diclofenac sodium generics are projected to experience steady market growth driven by increasing prevalence of pain-related conditions and an aging global population. Price erosion will likely continue but at a moderating pace as market competition stabilizes.
What is the Current Market Landscape for Diclofenac Sodium Generics?
The global market for diclofenac sodium generics is characterized by a high degree of competition, with numerous manufacturers vying for market share. Diclofenac sodium is a widely prescribed nonsteroidal anti-inflammatory drug (NSAID) used to treat pain and inflammation associated with conditions such as osteoarthritis, rheumatoid arthritis, and acute musculoskeletal injuries.
Key market drivers include:
- Rising incidence of osteoarthritis and rheumatoid arthritis: The global burden of these chronic inflammatory diseases continues to grow, particularly with aging populations. The World Health Organization (WHO) estimates that osteoarthritis affects approximately 300 million people worldwide, and rheumatoid arthritis affects up to 1% of the global population (1).
- Increased access to healthcare: Expanding healthcare infrastructure and rising disposable incomes in emerging economies are leading to greater patient access to pain management treatments, including generic NSAIDs like diclofenac sodium.
- Cost-effectiveness of generics: As a well-established generic drug, diclofenac sodium offers a significant cost advantage over branded alternatives, making it a preferred choice for both patients and healthcare systems focused on cost containment.
The market is segmented by route of administration, including oral tablets, topical gels, and injectable solutions. Oral formulations represent the largest segment due to their ease of use and widespread availability. Topical formulations are gaining traction for localized pain relief with reduced systemic side effects.
Major players in the diclofenac sodium generics market include Teva Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd., Mylan N.V. (now part of Viatris Inc.), Dr. Reddy's Laboratories Ltd., and Cipla Ltd. These companies leverage their extensive manufacturing capabilities, robust distribution networks, and established regulatory expertise to compete effectively.
What Factors Will Influence Future Price Trends?
Price projections for diclofenac sodium generics indicate a trend of gradual decline, followed by a period of relative stability. Several factors will influence these trends:
- Manufacturing Costs: Raw material costs, particularly for active pharmaceutical ingredients (APIs) and excipients, remain a primary determinant of production costs. Fluctuations in commodity prices and supply chain disruptions can impact these costs. For instance, disruptions in China, a major producer of APIs, have historically led to price volatility.
- Regulatory Landscape: Stringent quality control standards and evolving regulatory requirements from bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) necessitate ongoing investment in compliance. Changes in bioequivalence standards or manufacturing practices can increase compliance costs for manufacturers.
- Competition and Market Saturation: The high number of generic manufacturers creates intense price competition, particularly in mature markets. As more players enter the market or existing players expand production, downward price pressure intensifies. However, as the market matures, the rate of new entrants may slow, leading to a more stable competitive landscape.
- Patent Expirations and Generic Entry: While diclofenac sodium is a mature drug with long-expired primary patents, secondary patents related to specific formulations or delivery methods can still emerge. However, the impact of new patent expirations on the overall diclofenac sodium generics market is minimal given its established generic status. The primary factor influencing price remains the existing competitive landscape.
- Reimbursement Policies: Government reimbursement policies and private payer formularies significantly influence the pricing and market access of generic drugs. Policies favoring cost-effective treatments can bolster demand for diclofenac sodium generics, while tighter reimbursement controls could exert downward price pressure.
- Demand Dynamics: The underlying demand for diclofenac sodium is influenced by the prevalence of pain-related conditions. While this demand is expected to remain strong, shifts in prescribing patterns towards newer or alternative pain management therapies could influence overall volume and, consequently, pricing dynamics.
Projected Price Trajectory:
- Short-Term (1-2 years): Continued moderate price erosion driven by ongoing competition and potential supply-side pressures. Average selling prices (ASPs) may decline by an estimated 2-5% annually.
- Medium-Term (3-5 years): Price stabilization is anticipated as the market reaches a balance between supply and demand, and the rate of new generic entries moderates. Annual price fluctuations are expected to be within 0-2%.
- Long-Term (5+ years): Prices are likely to remain relatively stable, with adjustments primarily driven by inflation, manufacturing cost changes, and incremental shifts in market share among dominant generic players.
What are the Key Opportunities and Challenges for Stakeholders?
Stakeholders in the diclofenac sodium generics market face both significant opportunities and considerable challenges.
Opportunities:
- Emerging Markets: Growing healthcare expenditure and increasing patient populations in Asia-Pacific, Latin America, and Africa present substantial growth opportunities. Expanding distribution networks and establishing local manufacturing partnerships can capitalize on this demand. For example, India's pharmaceutical industry is a major global supplier of generic drugs, including diclofenac sodium, serving these expanding markets.
- Product Differentiation: While the core diclofenac sodium molecule is off-patent, opportunities exist in developing differentiated formulations, such as extended-release tablets, novel topical delivery systems, or combination products with synergistic pain relief agents. These can command premium pricing and capture specific patient segments. For instance, research into topical diclofenac formulations with enhanced skin penetration is ongoing.
- Contract Manufacturing: With established manufacturing expertise and economies of scale, contract manufacturing organizations (CMOs) can secure long-term supply agreements with larger pharmaceutical companies looking to outsource production. This segment is crucial for ensuring consistent supply and competitive pricing across the industry.
- Supply Chain Optimization: Companies that can optimize their supply chains, secure reliable API sources, and achieve cost efficiencies in manufacturing and distribution will gain a competitive advantage. Vertical integration, where feasible, can mitigate risks associated with API procurement.
Challenges:
- Intense Price Competition: The commoditized nature of generic diclofenac sodium generics results in razor-thin profit margins. Sustained price pressure can impact profitability and necessitate a focus on operational efficiency.
- Regulatory Hurdles: Navigating diverse and evolving regulatory requirements across different international markets demands significant investment in compliance and quality assurance. Delays in regulatory approvals can impact market entry timelines.
- Supply Chain Vulnerabilities: Reliance on specific geographic regions for API production or key raw materials exposes manufacturers to risks of shortages, price volatility, and geopolitical instability. Diversifying sourcing strategies is critical.
- Therapeutic Alternatives: The emergence of new pain management therapies, including biologics and novel non-NSAID options, could gradually erode the market share of traditional NSAIDs like diclofenac sodium, albeit slowly given its established efficacy and cost-effectiveness.
- Quality and Counterfeit Concerns: Maintaining high-quality standards is paramount. The risk of substandard or counterfeit products entering the market can damage brand reputation and pose patient safety risks, necessitating robust anti-counterfeiting measures and quality control.
What is the Projected Market Size and Growth Rate?
Estimating the precise market size for diclofenac sodium generics is complex due to data aggregation challenges across numerous regional markets and product variations. However, industry analysis suggests a substantial and growing market.
- Current Market Size: The global market for NSAIDs, of which diclofenac sodium is a significant component, was valued at approximately $15.2 billion in 2022, with generic diclofenac sodium representing a substantial portion of this. Projections indicate the NSAID market is expected to reach approximately $19.8 billion by 2029, growing at a compound annual growth rate (CAGR) of around 3.8% (2). The diclofenac sodium generics segment is estimated to be in the low billions of dollars annually.
- Projected Growth Rate: The diclofenac sodium generics market is expected to grow at a CAGR of approximately 3% to 4% over the next five to seven years. This growth will be primarily driven by the factors outlined earlier, particularly the increasing prevalence of chronic pain conditions and the expanding healthcare access in developing nations. The demand for cost-effective pain management solutions will continue to fuel the growth of generic diclofenac sodium.
Table 1: Diclofenac Sodium Generics Market Growth Drivers
| Driver | Impact on Market Growth |
|---|---|
| Increasing Prevalence of Osteoarthritis | Positive |
| Aging Global Population | Positive |
| Growing Healthcare Expenditure in Emerging Markets | Positive |
| Cost-Effectiveness of Generics | Positive |
| Development of Differentiated Formulations | Moderate Positive |
| Emergence of Therapeutic Alternatives | Moderate Negative |
| Intense Price Competition | Moderating Growth |
Table 2: Projected Price Trends (Average Percentage Change Annually)
| Time Horizon | Oral Formulations | Topical Formulations | Injectable Solutions |
|---|---|---|---|
| Short-Term (1-2 yrs) | -2% to -5% | -1% to -3% | -2% to -4% |
| Medium-Term (3-5 yrs) | 0% to +1% | 0% to +1% | 0% to +1% |
| Long-Term (5+ yrs) | 0% to +2% | 0% to +2% | 0% to +2% |
Note: Projections are based on current market conditions and may be subject to change due to unforeseen economic, regulatory, or competitive shifts.
Key Takeaways
The diclofenac sodium generics market will continue to expand, propelled by chronic pain conditions and global healthcare access. Price declines are projected to decelerate, leading to market stabilization. Stakeholders should focus on emerging markets and product differentiation to capitalize on opportunities, while navigating intense competition and supply chain risks.
Frequently Asked Questions
-
What is the primary competitive advantage for generic diclofenac sodium manufacturers? The primary competitive advantage lies in cost-effective manufacturing and efficient distribution to offer a significantly lower price point compared to originator brands or newer pain relief medications.
-
How do regulatory approvals impact diclofenac sodium generic pricing? While the primary molecule is long off-patent, regulatory approvals for manufacturing sites, specific formulations, or bioequivalence studies are necessary for market entry. Delays or stringent requirements can indirectly affect supply and, consequently, pricing.
-
Are there significant differences in pricing trends between oral and topical diclofenac sodium generics? Topical formulations may experience slightly slower price erosion due to more complex formulation and delivery technology, as well as potentially niche applications, but the overall downward pressure from competition remains.
-
What is the role of API sourcing in diclofenac sodium generic pricing? The cost and availability of Active Pharmaceutical Ingredients (APIs) are critical. Fluctuations in API prices, often influenced by geopolitical factors and manufacturing capacity in key producing countries like China and India, directly impact the cost of goods for generic manufacturers.
-
Will the emergence of biosimil drugs affect the diclofenac sodium generic market? Biosimil drugs are for biologic or highly similar versions of biologic medicines. Diclofenac sodium is a small molecule drug, so biosimilar competition is not applicable. Competition comes from other generic manufacturers of the same small molecule.
Citations
[1] World Health Organization. (n.d.). Osteoarthritis. Retrieved from https://www.who.int/news-room/fact-sheets/detail/osteoarthritis
[2] Grand View Research. (2023). NSAIDs Market Size, Share & Trends Analysis Report By Product (OTC, Prescription), By Type (Diclofenac, Ibuprofen, Naproxen, Celecoxib), By Route of Administration, By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/nsaids-market
More… ↓
