Share This Page
Drug Price Trends for GNP ACETAMINOPHEN
✉ Email this page to a colleague
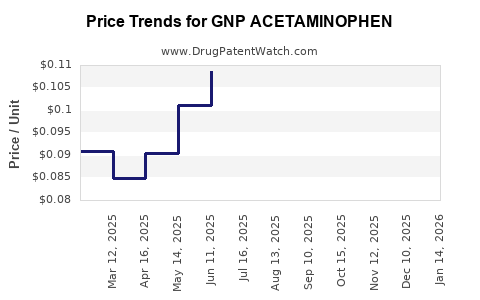
Average Pharmacy Cost for GNP ACETAMINOPHEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP ACETAMINOPHEN-IBUPROFEN 250-125 MG TABLET | 46122-0742-68 | 0.08816 | EACH | 2026-03-18 |
| GNP ACETAMINOPHEN-IBUPROFEN 250-125 MG TABLET | 46122-0742-68 | 0.09000 | EACH | 2026-02-18 |
| GNP ACETAMINOPHEN-IBUPROFEN 250-125 MG TABLET | 46122-0742-68 | 0.09051 | EACH | 2026-01-21 |
| GNP ACETAMINOPHEN-IBUPROFEN 250-125 MG TABLET | 46122-0742-68 | 0.09255 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP ACETAMINOPHEN Market Analysis and Financial Projection
Market Overview for GNP Acetaminophen
GNP Acetaminophen is a brand of acetaminophen, a widely used analgesic and antipyretic. Its global market is driven by high consumption in OTC pain relief products, prescription formulations, and combination drugs. The market size was valued at approximately USD 6.5 billion in 2022, with a compound annual growth rate (CAGR) of about 4.1% projected through 2030.
The drug's main markets include North America, Europe, and Asia-Pacific, with North America's market dominating due to high OTC drug consumption and stringent regulatory approval processes.
Current Market Dynamics
Market Drivers
- Growing awareness about the analgesic properties of acetaminophen.
- Increased preference for OTC pain relief solutions among consumers.
- Expansion of pharmaceutical formulations combining acetaminophen with other compounds.
- Rising prevalence of chronic pain conditions and fever-related illnesses.
Market Challenges
- Regulatory scrutiny over safety, especially concerning liver toxicity associated with overdose.
- Competition from similar analgesics such as NSAIDs and other OTC pain medications.
- Stringent regulations on manufacturing and marketing, particularly in the EU and US.
Market Trends
- Shift towards combination drugs targeting multiple symptoms.
- Development of liquid formulations and pediatric-friendly products.
- Increasing investments in research to improve safety profiles and delivery methods.
Key Manufacturers & Market Share
Top global players include:
| Company | Estimated Market Share (2022) | Notes |
|---|---|---|
| Johnson & Johnson | 22% | Major supplier of Tylenol products |
| Bristol-Myers Squibb | 12% | Offers branded acetaminophen formulations |
| Perrigo Company | 10% | Leading OTC supplier, especially in generics |
| Mallinckrodt Pharmaceuticals | 8% | Focuses on generic formulations |
| Others | 48% | Includes regional brands and private labels |
The market includes both branded and generic acetaminophen products. Generics constitute about 65% of global sales, driven by cost pressures and patent expirations.
Price Projections
Current Price Range
- Brand-name products (e.g., Tylenol): USD 0.10–0.30 per tablet.
- Generic formulations: USD 0.05–0.15 per tablet.
- Bulk API (active pharmaceutical ingredient): USD 3.00–6.00 per kilogram.
Future Price Trends (2023–2030)
- Unit prices for branded products are expected to decline marginally (~2% annually) due to increased generic competition.
- API prices will likely stabilize around USD 4.00 per kg, with minor fluctuations driven by raw material costs and supply chain constraints.
- Market consolidation and patent expiries could further push prices downward, especially for standard formulations.
Factors Affecting Pricing
- Raw material costs, particularly phenol and p-aminophenol.
- Manufacturing costs, influenced by regulatory compliance and quality standards.
- Regulatory actions aimed at curbing overdose risks could impact formulation costs.
- Geographic pricing differences influenced by local regulations and market penetration.
Regulatory Landscape Impact
US FDA classifications list acetaminophen as a drug with serious safety considerations, leading to:
- Emphasis on clear dosing guidelines.
- Restrictions on high-dose products.
- Post-market surveillance, potentially increasing compliance costs.
In Europe, EMA approvals stipulate strict dosage limits and warning labels.
Competitive Landscape and Market Entry
Entering the GNP Acetaminophen market involves regulatory approval across multiple jurisdictions. Generic entrants primarily focus on cost-effective manufacturing and distribution channels. Branded manufacturers rely on marketing, formulation innovations, and patient trust to maintain margins.
Market entry barriers include:
- Extensive regulatory requirements.
- Achieving manufacturing scale.
- Establishing distribution networks.
Key Takeaways
- GNP Acetaminophen faces steady demand globally, with the most significant markets in North America.
- The market has a high share of generics, contributing to downward pricing pressure.
- Prices for API are expected to stabilize or decline marginally through 2030.
- Regulatory scrutiny is shaping product formulations and labeling practices.
- Market consolidation will influence pricing dynamics and competitive strategies.
FAQs
1. What factors are most likely to influence the future price of GNP Acetaminophen?
Raw material costs, regulatory changes, market competition, and manufacturing efficiencies will predominantly impact prices.
2. How does regulatory safety concern impact market projections?
Enhanced safety requirements may increase formulation costs and restrict high-dose formulations, potentially reducing profitability for some manufacturers.
3. Are there new formulations of GNP Acetaminophen under development?
Research focuses on improved delivery mechanisms, pediatric formulations, and combination therapies. These innovations aim to enhance safety and compliance.
4. What is the anticipated growth rate for GNP Acetaminophen through 2030?
The global market is projected to grow at approximately 4.0-4.5% annually.
5. How does the presence of competitors impact pricing?
Increased competition, especially from generics, drives prices downward, limiting profit margins for branded products.
References
[1] MarketWatch, "Acetaminophen Market Size, Share & Trends," 2023.
[2] Grand View Research, "Over-the-Counter Pain Relief Market," 2022.
[3] FDA Regulations, "Acetaminophen Safety and Regulatory Status," 2022.
[4] IQVIA, "Global OTC Market Report," 2022.
[5] Pharma Intelligence, "Generic Drug Pricing and Market Trends," 2023.
More… ↓
