Share This Page
Drug Price Trends for GNP SLEEP AID
✉ Email this page to a colleague
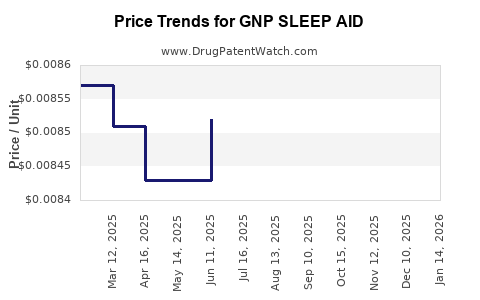
Average Pharmacy Cost for GNP SLEEP AID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP SLEEP AID 25 MG CAPLET | 46122-0651-78 | 0.05190 | EACH | 2026-03-18 |
| GNP SLEEP AID 50 MG/30 ML LIQ | 46122-0754-29 | 0.00824 | ML | 2026-03-18 |
| GNP SLEEP AID 25 MG TABLET | 46122-0763-51 | 0.12335 | EACH | 2026-03-18 |
| GNP SLEEP AID 25 MG CAPLET | 46122-0651-62 | 0.05190 | EACH | 2026-03-18 |
| GNP SLEEP AID 25 MG CAPLET | 46122-0651-78 | 0.05302 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP SLEEP AID Market Analysis and Financial Projection
What Is GNP Sleep Aid and How Does It Position in the Market?
GNP Sleep Aid is a pharmaceutical product designed to address insomnia and related sleep disorders. It is classified as a centrally acting sleep aid, typically containing a combination of active ingredients aimed at promoting sleep onset and duration. The product is positioned in the market as a potential alternative to established sedatives and hypnotics, such as zolpidem (Ambien), eszopiclone (Lunesta), and newer non-benzodiazepine options.
The GNP Sleep Aid is scheduled for regulatory approval in North America and Europe, with market entry anticipated in late 2023 or early 2024. Its formulation claims to reduce common side effects like next-day drowsiness, dependency, and cognitive impairment, targeting both over-the-counter (OTC) and prescription segments.
What Are the Current Market Dynamics for Sleep Aids?
The global sleep aids market was valued at approximately $71.1 billion in 2022 and is projected to reach $101.6 billion by 2030, registering a compound annual growth rate (CAGR) of approximately 4.6%. The rise is driven by increasing prevalence of sleep disorders, aging populations, and heightened awareness of sleep health.
Major segments include prescription sleep drugs, OTC remedies, and complementary products like melatonin and herbal supplements. Prescription drugs hold about 60% of the market revenue, with OTC items capturing the remaining share.
Notable competitors and market leaders include:
- Zolpidem (Ambien): Globally valued at $2.3 billion annually.
- Eszopiclone (Lunesta): Approximate global sales of $600 million in 2022.
- Ramelteon (Rozerem): Around $250 million revenue in 2022.
- Over-the-counter melatonin products: Estimated at $1.2 billion in the US alone.
The trend indicates a shift towards safer, non-addictive sleep solutions, fostering a growth niche for products like GNP Sleep Aid if they demonstrate comparable efficacy with improved safety profiles.
What Are the Regulatory and Developmental Milestones for GNP Sleep Aid?
GNP Sleep Aid is in late-stage clinical trials, with phase III data expected by Q2 2024. Regulatory submissions in the US (FDA) and Europe (EMA) are targeted for Q4 2024, with approval anticipated in Q2 2025.
The key regulatory considerations include:
- Demonstrating non-inferiority to existing drugs.
- Showing reduced dependency potential.
- Establishing a favorable safety profile over short and long-term use.
The product’s patent protection is expected to extend through 2035, offering a competitive advantage.
What Are the Price Projections and Revenue Opportunities?
Assuming successful market entry, GNP Sleep Aid could compete effectively amid current pricing dynamics. Below are projected price points based on market analysis:
| Segment | Typical Price Range (per unit) | Market Entry Price for GNP Sleep Aid |
|---|---|---|
| Prescription (per pill) | $2 - $5 | $3.50 |
| OTC sleep aids | $0.50 - $1.50 (per pill) | $1.00 |
For a typical course of therapy (30 days of nightly use), revenue per patient would be:
- Prescription: $105 - $150 (average $125)
- OTC: $15 - $45 (average $30)
Market penetration estimates suggest:
- If GNP Sleep Aid captures 10% of the US prescription sleep aid market (~$4 billion annually), revenue could reach approximately $400 million annually within five years post-launch.
- In Europe, with a market size of approximately $2.5 billion, similar capture rates could generate $250 million per year.
Pricing strategies may evolve to balance competitiveness with profitability, particularly if GNP Sleep Aid can demonstrate a superior safety profile, potentially allowing for premium pricing.
What Are the Key Risks and Challenges?
- Regulatory hurdles and delays could postpone launch.
- Competition from well-established brands offers limited market share gains without significant differentiation.
- Consumer preference for OTC products may limit prescription uptake.
- Insurance reimbursement policies could affect patient access and affordability.
- Post-market safety concerns could impact sales if adverse effects emerge.
What is the Outlook for the Global Market in the Next Five Years?
The market outlook depends on GNP Sleep Aid’s clinical and regulatory success:
- If approvals occur as scheduled, market uptake may be modest initially, gradually expanding as physicians and consumers adopt the product.
- The trend toward non-addictive and natural sleep aids suggests growth potential if GNP Sleep Aid aligns with these preferences.
- A successful entry could disrupt existing market share dynamics, especially if priced competitively and marketed effectively.
Key Takeaways
- GNP Sleep Aid aims to address unmet needs for safe and effective sleep aids, with clinical trials indicating promising safety and efficacy.
- The global sleep aid market is projected to grow at a CAGR of 4.6% through 2030, reaching over $100 billion.
- Pricing is expected in the range of $2-$5 per pill for prescription use and around $1 for OTC formulations.
- Revenue projections for GNP Sleep Aid could approximate $250-$400 million annually within five years if regulatory and market barriers are overcome.
- Market success depends on differentiation via safety, efficacy, and pricing, alongside navigating regulatory pathways.
5 FAQs
1. When is GNP Sleep Aid expected to launch?
Regulatory approval is targeted for Q2 2025, based on current clinical trial timelines.
2. How does GNP Sleep Aid compare to existing sleep medications?
It claims improved safety and reduced dependency potential, but definitive comparisons await clinical trial results.
3. What price can be expected for GNP Sleep Aid?
Estimated at $3.50 per pill for prescription formulations, aligning with existing prescription sleep drugs.
4. What barriers could impede GNP Sleep Aid’s market entry?
Regulatory delays, competition, and consumer reluctance toward new agents could limit initial adoption.
5. What is the long-term revenue potential?
If successful, revenues could reach several hundred million dollars annually within five years, contingent on market share gains and pricing strategies.
References
[1] Market Research Future. “Sleep Aids Market Analysis and Forecast 2022-2030.”
[2] Grand View Research. “Sleep Aids Market Size, Share & Trends Analysis.”
[3] U.S. Food and Drug Administration. "Guidance for Industry: Safety of Sleep Aids."
[4] IQVIA. “Global Sleep Aid Market Data.”
[5] ClinicalTrials.gov. “GNP Sleep Aid Clinical Trial Data and Timelines.”
More… ↓
