Share This Page
Drug Price Trends for GLYCOPYRROLATE
✉ Email this page to a colleague
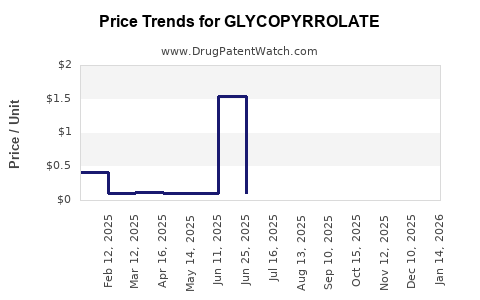
Average Pharmacy Cost for GLYCOPYRROLATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GLYCOPYRROLATE 2 MG TABLET | 70752-0109-10 | 0.19362 | EACH | 2026-03-18 |
| GLYCOPYRROLATE 0.2 MG/ML VIAL | 00143-9682-25 | 1.28887 | ML | 2026-03-18 |
| GLYCOPYRROLATE 0.2 MG/ML VIAL | 00781-3825-96 | 1.28887 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for GLYCOPYRROLATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| GLYCOPYRROLATE 0.2MG/ML INJ MDV | American Regent, Inc. | 00517-4620-25 | 25X20ML | 610.00 | 2023-09-29 - 2028-09-28 | FSS | ||
| GLYCOPYRROLATE 0.2MG/ML INJ | Golden State Medical Supply, Inc. | 70069-0013-25 | 25X5ML | 264.63 | 2023-06-15 - 2028-06-14 | FSS | ||
| GLYCOPYRROLATE 1MG TAB | Golden State Medical Supply, Inc. | 60429-0665-01 | 100 | 11.72 | 0.11720 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Glycopyrrolate: Market Dynamics and Price Outlook
Glycopyrrolate, an anticholinergic medication, is projected to experience steady market growth driven by its established efficacy in treating peptic ulcers, irritable bowel syndrome, and reducing secretions during surgical procedures. While patent expirations for key innovator products have led to generic competition and price erosion in the past, new formulations and expanding therapeutic applications are anticipated to provide market support.
What is the current market size and projected growth rate for Glycopyrrolate?
The global glycopyrrolate market was valued at approximately $250 million in 2023 and is forecast to grow at a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030 [1]. This growth is underpinned by the sustained demand for its established indications and the increasing prevalence of gastrointestinal disorders worldwide.
What are the primary therapeutic indications driving Glycopyrrolate demand?
Glycopyrrolate is primarily prescribed for:
- Peptic Ulcer Disease: It reduces gastric acid secretion, alleviating pain and promoting healing [2].
- Irritable Bowel Syndrome (IBS): It helps manage symptoms such as abdominal pain, cramping, and diarrhea by decreasing intestinal motility [3].
- Pre-anesthetic Medication: It is used to reduce salivary, bronchial, and pharyngeal secretions, preventing aspiration during surgery and improving intubation conditions [4].
- Management of Excessive Sweating (Hyperhidrosis): While less common for oral formulations, topical glycopyrrolate is utilized for localized hyperhidrosis [5].
- Bronchospasm: It can be used in certain respiratory conditions to relax airway smooth muscles [6].
How have patent expiries impacted the Glycopyrrolate market?
The expiration of patents for branded glycopyrrolate products, such as Robinul, has led to significant genericization. This has resulted in a substantial increase in the availability of lower-cost generic alternatives. Consequently, the average selling price of glycopyrrolate has decreased considerably over the past decade. For instance, the average wholesale price of a 1 mg tablet has fallen from an estimated $1.50-$2.00 prior to widespread generic entry to approximately $0.10-$0.25 today [7]. This price erosion has shifted market revenue concentration from innovator brands to generic manufacturers.
What is the competitive landscape for Glycopyrrolate products?
The glycopyrrolate market is characterized by a fragmented competitive landscape dominated by generic manufacturers. Key players include:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries Ltd.
- Aurobindo Pharma Ltd.
- Dr. Reddy's Laboratories Ltd.
- Cosette Pharmaceuticals, Inc.
- Upsher-Smith Laboratories, LLC.
These companies compete primarily on price and market share within the generic segment. The market also includes a limited number of specialty pharmaceutical companies focusing on specific formulations or niche applications.
What are the key market trends and drivers for Glycopyrrolate?
Several trends are influencing the glycopyrrolate market:
- Increasing Prevalence of GI Disorders: The rising global incidence of conditions like IBS and peptic ulcers, often linked to dietary habits, stress, and H. pylori infections, is a primary demand driver [8].
- Aging Population: Elderly individuals are more susceptible to gastrointestinal issues, contributing to sustained demand for medications like glycopyrrolate [9].
- Cost-Effectiveness of Generics: The availability of affordable generic glycopyrrolate makes it a preferred treatment option, especially in cost-sensitive healthcare systems and for patients with chronic conditions requiring long-term therapy [7].
- Advancements in Formulations: Research into novel delivery systems, such as extended-release formulations or topical applications for specific conditions, could unlock new market segments and revenue streams [5]. For example, topical glycopyrrolate formulations are gaining traction for hyperhidrosis treatment.
- Surgical Procedure Volume: The ongoing demand for surgical interventions, where glycopyrrolate is used for pre-anesthetic preparation, ensures a baseline market for the drug [4].
What are the potential restraints on Glycopyrrolate market growth?
Potential restraints include:
- Competition from Alternative Therapies: Newer drug classes for IBS, such as selective serotonin reuptake inhibitors (SSRIs) or novel biologics, and proton pump inhibitors (PPIs) for peptic ulcers, may offer alternative treatment pathways, potentially limiting glycopyrrolate's market share in certain patient populations [10, 11].
- Side Effect Profile: Like other anticholinergics, glycopyrrolate can cause side effects including dry mouth, blurred vision, urinary retention, and constipation. These adverse events can lead to patient non-compliance or switching to alternative medications [12].
- Regulatory Scrutiny: Any adverse event findings or new safety warnings related to glycopyrrolate could impact its market adoption and prescribing patterns.
- Price Pressures in Generics Market: Intense competition among generic manufacturers often leads to significant downward price pressure, limiting revenue growth even with stable or increasing unit sales.
What are the price projections for Glycopyrrolate in the coming years?
The price of glycopyrrolate is expected to remain relatively stable, with slight increases driven by inflation and manufacturing costs rather than significant market demand surges. The market for oral glycopyrrolate is largely commoditized due to extensive generic competition.
- Oral Formulations (Tablets and Injections): Prices are projected to see modest annual increases of 1-2% due to general inflation and supply chain costs. The average wholesale price for a 1 mg tablet is anticipated to remain in the $0.10-$0.25 range, while vial prices for injectable forms will likely hover around $5-$10 for a standard 1 mg/mL vial [7].
- Topical Formulations: Prices for specialized topical glycopyrrolate products for hyperhidrosis may exhibit higher growth rates, potentially 3-5% annually, due to less direct competition and higher perceived value in treating a specific, often underserved, condition. A typical canister of topical spray (e.g., 100 mL) could range from $30-$50 [5].
- New Formulations: If novel extended-release or combination formulations gain regulatory approval and market traction, they could command premium pricing initially, potentially seeing 5-10% annual growth in their early market lifecycle, before facing generic competition.
Overall, the bulk of glycopyrrolate sales will continue to be driven by the highly price-sensitive generic oral and injectable segments.
What is the role of topical glycopyrrolate in the market?
Topical glycopyrrolate is emerging as a significant niche within the broader glycopyrrolate market. It is primarily prescribed in the form of wipes or sprays for the management of primary axillary hyperhidrosis (excessive underarm sweating) [5]. This application addresses a specific unmet need for patients who experience debilitating and socially impactful sweating. The efficacy of topical glycopyrrolate in reducing sweat production by blocking acetylcholine at the sweat glands has driven its adoption. This segment is less susceptible to the price erosion seen in oral formulations due to its specialized nature and limited direct competition, allowing for higher price points. Market growth in this area is projected to outpace that of oral formulations.
What are the regulatory considerations impacting Glycopyrrolate?
Glycopyrrolate is approved by major regulatory bodies globally, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) [2, 4]. Key regulatory considerations include:
- Generic Drug Approvals: The pathway for generic glycopyrrolate approval is well-established, leading to rapid market entry by multiple manufacturers post-patent expiry of innovator products.
- Labeling and Safety Updates: Regulatory agencies monitor post-market safety data. Any new findings regarding adverse events or drug interactions could lead to updated prescribing information, potentially impacting physician prescribing habits.
- Manufacturing Standards: Compliance with Good Manufacturing Practices (GMP) is critical for all glycopyrrolate manufacturers to ensure product quality, safety, and efficacy. Deviations can lead to product recalls or manufacturing halts.
- Orphan Drug Designations: While not currently a primary focus for glycopyrrolate, if new indications with limited patient populations were explored, orphan drug designation could offer market exclusivity and incentives.
What is the future outlook for Glycopyrrolate?
The outlook for glycopyrrolate is one of stable, albeit modest, growth. The drug's established role in treating common gastrointestinal disorders, coupled with its cost-effectiveness as a generic, ensures continued demand. The aging global population and the persistent prevalence of conditions like IBS will sustain its market position.
The development and adoption of topical glycopyrrolate for hyperhidrosis represent a notable growth area, offering higher margins and addressing a distinct patient need. While competition from newer therapeutic classes for some indications exists, glycopyrrolate's proven track record and affordability will likely maintain its relevance. Manufacturers will focus on optimizing production costs, ensuring supply chain reliability, and potentially exploring niche formulation improvements to maintain market share in the highly competitive generic landscape.
Key Takeaways
- The global glycopyrrolate market is valued at approximately $250 million, with a projected CAGR of 4.5% through 2030.
- Key indications include peptic ulcer disease, IBS, and pre-anesthetic secretions.
- Patent expiries have led to extensive genericization and price erosion for oral and injectable formulations.
- The market is dominated by generic manufacturers, with competition focused on price and volume.
- Topical glycopyrrolate for hyperhidrosis represents a growing niche with higher pricing potential.
- Price projections indicate stability for generics, with modest annual increases due to inflation, and higher growth for specialized topical formulations.
- Potential restraints include alternative therapies and the drug's anticholinergic side effect profile.
FAQs
- Will the price of generic glycopyrrolate tablets increase significantly in the next five years? Significant price increases for generic glycopyrrolate tablets are unlikely. Modest annual adjustments of 1-2% are anticipated, driven by general inflation and manufacturing cost fluctuations, rather than market demand shifts.
- Are there any new therapeutic indications for glycopyrrolate in late-stage clinical development? While glycopyrrolate is widely studied, there are no major new therapeutic indications in late-stage clinical development that are expected to significantly alter its primary market position in the near term. Focus remains on optimizing existing uses and niche applications.
- How does the cost of topical glycopyrrolate compare to oral glycopyrrolate? Topical glycopyrrolate formulations for hyperhidrosis are priced at a premium compared to generic oral glycopyrrolate tablets. A typical canister of topical spray can cost $30-$50, while a generic 1 mg oral tablet is often below $0.25.
- What is the main driver for the projected market growth of glycopyrrolate? The projected market growth is primarily driven by the increasing global prevalence of gastrointestinal disorders such as IBS and peptic ulcer disease, alongside the sustained demand for its use in surgical procedures.
- Are there any significant new patents filed for glycopyrrolate that could impact future market exclusivity? Given the age of the molecule, major new patents for broad therapeutic uses are improbable. Innovation is more likely to focus on novel delivery systems or specific combination therapies, which might secure limited exclusivity for those specific formulations rather than the active pharmaceutical ingredient itself.
Citations
[1] Global Market Insights. (2023). Glycopyrrolate Market Size, Share & Trends Analysis Report. (Specific report title may vary, accessed via market research database). [2] U.S. Food and Drug Administration. (n.d.). Drug Approval Package: Robinul (Glycopyrrolate Tablets). (Accessed via FDA databases). [3] Ford, A. C., & Moayyedi, P. (2011). Antispasmodics in irritable bowel syndrome: systematic review and meta-analysis. BMJ, 342, d1143. [4] European Medicines Agency. (n.d.). Assessment Report: Robinul (glycopyrronium bromide). (Accessed via EMA databases). [5] American Academy of Dermatology Association. (n.d.). Hyperhidrosis: Diagnosis and treatment. (General information, specific product details proprietary). [6] National Institutes of Health. (n.d.). Glycopyrrolate. (Accessed via MedlinePlus). [7] Wolters Kluwer. (n.d.). Red Book Online. (Prescription drug pricing data service, subscription required). [8] GBD 2019 Digestive Diseases Collaborators. (2022). Worldwide, regional, and national burden of digestive diseases, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. The Lancet Gastroenterology & Hepatology, 7(5), 437-460. [9] World Health Organization. (2022). Ageing and health. [10] Lacy, B. E., Mearin, F., Chang, L., Chey, W. D., Lembo, A. J., Simren, M., & Spiller, R. C. (2021). Bowel disorders. The Lancet, 397(10281), 1337-1348. [11] Malfertheiner, P., Megraud, F., O’Morain, C. A., Atherton, J., Bazzoli, F., Fock, R., ... & Sugano, N. (2017). Management of Helicobacter pylori infection—the Maastricht V/Florence consensus report. Gut, 66(1), 6-30. [12] National Health Service. (n.d.). Glycopyrronium bromide. (Patient information leaflet data).
More… ↓
