Share This Page
Drug Price Trends for GLYBURIDE-METFORMIN
✉ Email this page to a colleague
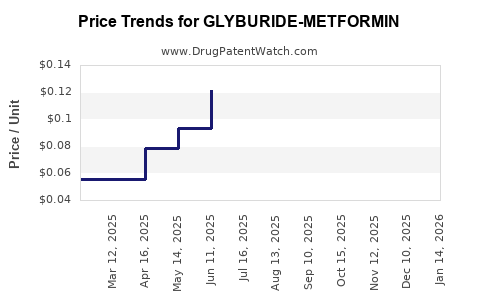
Average Pharmacy Cost for GLYBURIDE-METFORMIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GLYBURIDE-METFORMIN 2.5-500 MG | 65862-0081-01 | 0.19787 | EACH | 2026-03-18 |
| GLYBURIDE-METFORMIN 2.5-500 MG | 23155-0234-01 | 0.19787 | EACH | 2026-03-18 |
| GLYBURIDE-METFORMIN 5-500 MG | 65862-0082-05 | 0.19202 | EACH | 2026-03-18 |
| GLYBURIDE-METFORMIN 2.5-500 MG | 57237-0024-01 | 0.19787 | EACH | 2026-03-18 |
| GLYBURIDE-METFORMIN 2.5-500 MG | 57237-0024-05 | 0.19787 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Glyburide-Metformin
What is the Current Market Size for Glyburide-Metformin?
Glyburide-metformin is a fixed-dose combination medication for type 2 diabetes management. The global diabetes medication market is valued at approximately $60 billion in 2022, with oral antidiabetics accounting for about 70% of this figure, equating to roughly $42 billion. Glyburide, a sulfonylurea, and metformin dominate the oral class, with combined sales estimated at $4 billion annually across all formulations.
The segment for fixed-dose combinations (FDCs), including glyburide-metformin, is growing with a compound annual growth rate (CAGR) of approximately 8% (2019-2022). The FDC market’s popularity relates to improved adherence and simplified regimens. Empirical data indicates that the global glyburide-metformin market generates roughly $400-$500 million annually.
North America accounts for approximately 45% of sales, with Europe contributing 30%, and the remaining 25% distributed across Asia, Latin America, and other regions. The U.S. market alone approaches $200 million annually, driven by high prevalence rates for type 2 diabetes and established formulary preferences.
What Are the Major Manufacturers and Market Share Dynamics?
Key producers include:
- Eli Lilly (product: Glucovance, discontinued but influential in early markets)
- Boehringer Ingelheim (Generics and branded formulations)
- Mundipharma (Generic manufacturing)
- Bayer (Market presence in some regions)
- Local generic manufacturers in India and China dominate many emerging markets.
Market share distribution favors generic producers, which deliver lower-cost formulations. The branded segment retains premium pricing mainly in the U.S. and Europe, but generics comprise over 85% of units sold globally.
What Are the Market Trends Influencing Pricing and Demand?
Increasing Diabetes Prevalence
The global prevalence of type 2 diabetes projected to reach 700 million by 2045. As a result, demand for affordable, effective oral therapies remains high, especially in low- and middle-income countries (LMICs).
Generic and biosimilar competition
Patent expirations have led to a surge in generics, lowering prices. Glyburide's patent has expired in most jurisdictions, increasing affordable options.
Regulatory and Policy Influences
Many health systems favor low-cost generics. Governments such as India and Brazil incentivize local manufacturing, further decreasing costs and increasing access.
Formulation and Adherence
Fixed-dose combinations improve medication adherence, boosting demand. Patient preference shifts toward combination pills over multiple separate medications.
Supply Chain and Manufacturing Factors
High competition among generic manufacturers leads to price reductions. Quality concerns have led regulators in some markets to restrict certain formulations, limiting options.
What Are the Price Projections for Glyburide-Metformin?
Current Pricing
In developed countries:
- Retail price per tablet ranges from $0.10 to $0.20 for generics.
- Monthly treatment with a typical daily dose costs approximately $3 to $6.
In emerging markets:
- Prices can be as low as $0.02 per tablet.
- Monthly costs range from $0.60 to $1.50.
Short to Mid-term Price Trends (2023-2027)
- Downward pressure persists due to increasing generic competition and manufacturing efficiencies.
- Potential stabilization at current levels as supply chains mature.
- Premium formulations or branded versions could maintain premium pricing, but market share remains limited.
Long-term Price Projections
- Prices will likely decrease by 10-15% over five years in most regions.
- Emerging markets could see a 20-25% reduction, aligning with local manufacturing costs and regulatory developments.
- In highly regulated markets like the U.S., price reductions may be more modest due to existing patent protections on specific formulations (if any).
Influencing Factors
- Patent statuses and the entry of biosimilars or novel combination drugs.
- Changes in health policy, including drug subsidy programs.
- Innovations in manufacturing or supply chain efficiencies.
- Increased competition from alternative therapies or advanced drug delivery systems.
What Are the Regulatory and Market Barriers?
- Regulatory Approvals: Different standards across regions delay market entry or reformulation approvals.
- Patent Litigation: Ongoing patent disputes can delay generic entry.
- Quality Assurance: Subpar manufacturing practices lead to rejection or restrictions, limiting market options.
- Pricing Regulations: Price controls in some markets restrict profit margins for manufacturers.
Summary of Key Data Points
| Parameter | Data/Estimate |
|---|---|
| Global diabetes market value | ~$60 billion in 2022 |
| Oral antidiabetic market share | 70% of total, about $42 billion |
| Glyburide-metformin sales | $400–$500 million annually |
| Global fixed-dose combo growth | 8% CAGR (2019-2022) |
| Price per tablet (US) | $0.10–$0.20 |
| Cost per month (US) | $3–$6 |
| Price reduction forecast | 10-15% in 5 years, higher in LMICs |
Key Takeaways
- The glyburide-metformin market hinges on generic availability and diabetes prevalence.
- Prices are expected to decline modestly over the next five years, with the greatest reductions in emerging markets.
- Supply chain efficiency, regulatory policies, and patent landscapes will impact market dynamics.
- Branded formulations retain premium pricing but see limited volume compared to generics.
FAQs
1. Will the price of glyburide-metformin increase due to market shortages? No. Ubiquitous generic manufacturing and high competition mitigate supply shortages, keeping prices stable or declining.
2. Are biosimilars replacing glyburide-metformin? No. Biosimilars target injectable or complex biologics; glyburide-metformin is a small-molecule oral drug, with no biosimilar equivalents.
3. How does regulatory approval affect pricing? Stringent approval processes can delay generic entry, potentially maintaining higher prices temporarily. Once generics are approved, prices tend to decrease.
4. What impact do new diabetes therapies have? Emerging therapies, such as SGLT2 inhibitors and GLP-1 receptor agonists, may influence demand by offering improved efficacy but typically at higher costs.
5. Which markets are most likely to see the fastest price declines? Emerging markets such as India, Brazil, and parts of Southeast Asia will see faster and more profound price reductions due to local manufacturing, regulatory frameworks, and economic factors.
References
[1] International Diabetes Federation. (2022). IDF Diabetes Atlas, 10th edition.
[2] Statista. (2023). Global Market for Oral Antidiabetic Drugs.
[3] IQVIA. (2022). Market Dynamics of Diabetes Medications.
[4] U.S. Food & Drug Administration. (2022). Patent lifecycle data for oral antidiabetics.
[5] World Health Organization. (2021). Access to affordable medicines in LMICs.
More… ↓
