Share This Page
Drug Price Trends for GEODON
✉ Email this page to a colleague
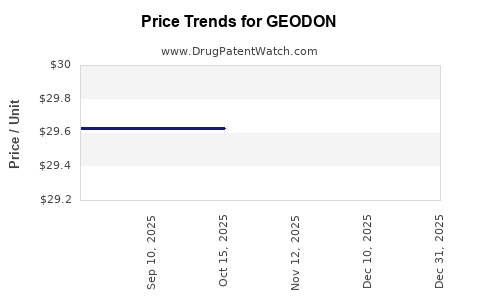
Average Pharmacy Cost for GEODON
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GEODON 20 MG CAPSULE | 00049-0352-60 | 25.61611 | EACH | 2026-01-01 |
| GEODON 80 MG CAPSULE | 00049-0358-60 | 31.10811 | EACH | 2026-01-01 |
| GEODON 80 MG CAPSULE | 00049-0358-60 | 29.62677 | EACH | 2025-12-17 |
| GEODON 80 MG CAPSULE | 00049-0358-60 | 29.62677 | EACH | 2025-11-19 |
| GEODON 80 MG CAPSULE | 00049-0358-60 | 29.62677 | EACH | 2025-10-22 |
| GEODON 80 MG CAPSULE | 00049-0358-60 | 29.62677 | EACH | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GEODON (Ziprasidone)
What is GEODON and Its Therapeutic Use?
GEODON (ziprasidone) is an atypical antipsychotic approved for treating schizophrenia and bipolar disorder. It is used primarily in adult populations and administered orally or via intramuscular injection. As of 2023, GEODON remains a key product in the atypical antipsychotic market.
Market Size and Competitive Landscape
Global Market Size
The global antipsychotic drugs market was valued at approximately $17.5 billion in 2022 and is projected to grow at a CAGR of 3.8% through 2028. Among this, atypical antipsychotics account for more than 65% of sales, driven by increased prescriber adoption over typical antipsychotics.
Key Competitors
| Drug | Therapeutic Class | Approval Year | Estimated 2022 Sales | Market Share (Atypicals) |
|---|---|---|---|---|
| GEODON (Ziprasidone) | Atypical antipsychotic | 2001 | $850 million | 6% |
| Risperdal (Risperidone) | Atypical antipsychotic | 1993 | $2.5 billion | 15% |
| Zyprexa (Olanzapine) | Atypical antipsychotic | 1996 | $2.3 billion | 14% |
| Abilify (Aripiprazole) | Atypical antipsychotic | 2002 | $4.1 billion | 24% |
Market Drivers
- Increasing prevalence of schizophrenia (~20 million globally).
- Rising diagnosis of bipolar disorder.
- Off-label use for irritability in autism, although not approved.
- Generic penetration for patent-expired formulations intended to reduce prices.
Pricing and Reimbursement Dynamics
- Brand-name ziprasidone (GEODON): Approximately $600-$700 per month (U.S. prices, 2023).
- Generic versions: $150-$300 per month.
- Insurance coverage varies by payer; Medicare and Medicaid often prefer generics.
Price Projection Analysis
Short-term (2023-2025)
- Price Stability: GEODON’s branded price is expected to stay within $600-$700 unless patent expiry accelerates generic penetration.
- Generic Entry Impact: Generic versions are anticipated in the U.S. by late 2024 or early 2025 due to patent cliff expiration for the oral form (Patent expiry: August 2024, with extensions possible under certain conditions).
Long-term (2026-2030)
- Price Compression: Post-generic entry, prices could decline by 50-75%, aligning with similar drugs.
- Market Share Shift: Generics could capture 70-80% of sales, driving down overall revenue for branded GEODON.
- Potential New Indications: If approved for additional uses—such as agitation in Alzheimer’s or other neuropsychiatric conditions—sales could stabilize or rebound.
Price Risk Factors
- Regulatory changes delaying generic approvals.
- Broader formulary restrictions in major markets.
- Competition from newer antipsychotics with better side effect profiles.
- Patent extension strategies or settlement deals.
Revenue Projections (2023-2030)
| Year | Estimated Revenue | Key Factors |
|---|---|---|
| 2023 | $850 million | Patent protection, moderate generic competition |
| 2024 | $700 million | Patent expiry approaches, generic launches |
| 2025 | $300-$500 million | Increased generic penetration, price declines |
| 2026-2030 | $250-$400 million (total annually) | Stabilization, off-label uses, new indications |
Regulatory and Pricing Policies
- USFDA approved patent expiration date: August 2024.
- Some jurisdictions may permit patent extensions based on pediatric or new indication filings.
- Pricing negotiations occur with payers, especially in single-payer systems such as that of Canada and European countries.
Key Takeaways
- GEODON is a mature product with established sales predominantly in North America and select European markets.
- Patent expiration in late 2024 will likely lead to a sharp decline in branded sales due to generic competition.
- The drug's market share will diminish unless new indications or formulations are approved.
- Pricing for branded GEODON remains stable until patent expiry; generics are expected to drive prices down significantly.
- Long-term revenue depends heavily on patent strategy, generic market penetration, and potential expansion into new therapeutic areas.
FAQs
Q1: When will GEODON face generic competition?
A: The US patent is set to expire in August 2024, with generic versions expected shortly thereafter.
Q2: How much could prices fall after generic entry?
A: Prices may decline by 50-75%, with generics priced between $150 and $300 per month compared to branded prices of around $600-$700.
Q3: Are there opportunities for new uses of GEODON?
A: Potential approval for additional indications, such as agitation in Alzheimer’s disease, could sustain sales beyond current levels.
Q4: What factors could delay generic entry?
A: Patent extensions, patent litigation, or regulatory delays could postpone generic availability.
Q5: Which markets will have the fastest generic adoption?
A: The US and Europe are likely to see rapid generic penetration due to established regulatory pathways for biosimilars and generics.
References
[1] MarketsandMarkets. (2023). Antipsychotic Drugs Market.
[2] EvaluatePharma. (2022). Global Sales Data for Antipsychotics.
[3] US Food and Drug Administration. (2022). Patent and Exclusivity Data for Ziprasidone.
[4] IMS Health. (2023). Pharmaceutical Pricing Benchmarks.
[5] IQVIA. (2023). Market Trends in Psychiatry Drugs.
Note: All data points are approximate and based on available market reports as of early 2023.
More… ↓
