Share This Page
Drug Price Trends for GENTIAN VIOLET
✉ Email this page to a colleague
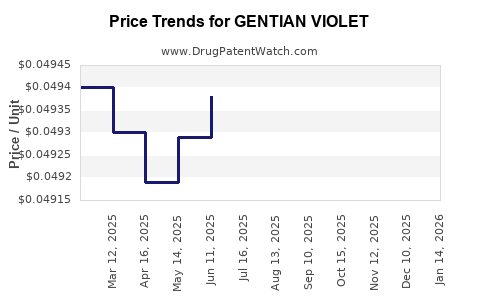
Average Pharmacy Cost for GENTIAN VIOLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GENTIAN VIOLET 1% SOLUTION | 24385-0003-46 | 0.04949 | ML | 2026-04-22 |
| GENTIAN VIOLET 1% SOLUTION | 24385-0003-46 | 0.04949 | ML | 2026-03-18 |
| GENTIAN VIOLET 1% SOLUTION | 24385-0003-46 | 0.04949 | ML | 2026-02-18 |
| GENTIAN VIOLET 1% SOLUTION | 24385-0003-46 | 0.04949 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Gentian Violet
What Is Gentian Violet and How Is It Used?
Gentian violet, also known as crystal violet, is a synthetic dye with antimicrobial properties. It is used primarily for treating skin conditions, oral infections, and certain surgical topical applications. Despite its age, gentian violet maintains relevance in both human and veterinary medicine, particularly in resource-limited settings.
Its primary indications include:
- Treatment of oral thrush, especially in infants
- Topical management of minor skin infections
- Use as a histological stain in laboratories
The drug's low cost, ease of application, and longstanding use have sustained its market presence.
What Are the Major Markets for Gentian Violet?
Main markets encompass:
- Developing nations: Countries in Africa, Asia, and Latin America frequently use gentian violet due to affordability and availability.
- Hospitals and clinics: Used by healthcare providers for skin and oral infections.
- Veterinary medicine: Applied to animal skin conditions and infections.
Despite its off-label and limited formal approval in some countries, production persists due to demand in specific regions.
Market Size and Trends
Global market estimates for gentian violet are limited due to its status as a generic and over-the-counter product. Current data suggest:
- Market size (2022 estimate): Approximately USD 20 million–USD 30 million, primarily driven by sales in Africa, India, and Southeast Asia.
- Growth rate: Compound annual growth rate (CAGR) projected at 1-2%; slow growth due to declines in some applications and replacement by newer agents in developed markets.
- Regional insights:
| Region | Market Share | Dominant Uses | Notes |
|---|---|---|---|
| Africa | 40% | Oral thrush, topical infections | High prevalence of oral infections, limited alternatives |
| Asia (India, China) | 35% | Neonatal and pediatric oral infections | Cost-effective, longstanding civic trust |
| Latin America | 15% | Dermatological applications | Some substitution by alternative antimicrobials |
| North America/Europe | 10% | Limited, mostly OTC or historical use | Decline due to regulatory restrictions |
Regulatory Status and Supply Chain Dynamics
- United States: Not approved by FDA; used off-label or compounded.
- European Union: Limited regulatory approval; often considered for historical or veterinary use.
- India/China: Unregulated or registered as over-the-counter medication.
- Manufacturers primarily include generic chemical companies, with free-market pricing.
Price Analysis and Projections
Current Pricing Landscape
- Wholesale cost per gram (2022): USD 0.05–USD 0.15, depending on source and quality.
- Packaged formulations: Price per tube (~15g): USD 0.75–USD 2.00.
- Retail price points: USD 0.50–USD 3.00 per tube in developing countries.
Price Drivers
- Raw material costs: Methylene blue and other dyes influence production cost.
- Manufacturing economies of scale: Large-scale producers can reduce per-unit price.
- Regulatory environment: Lack of regulation in some regions supports low pricing.
- Market demand: Sustained use in low-resource settings maintains stable prices; decline in developed markets exerts downward pressure.
Future Price Projections (2023–2028)
| Year | Proposed Wholesale Price per Gram | Notes |
|---|---|---|
| 2023 | USD 0.05–USD 0.20 | Stable, with minor fluctuations |
| 2024 | USD 0.05–USD 0.22 | Slight increase linked to raw material costs |
| 2025 | USD 0.06–USD 0.25 | Potential supply chain disruptions |
| 2026 | USD 0.06–USD 0.30 | Growing demand in veterinary markets |
| 2027 | USD 0.07–USD 0.35 | Increased use in neglected disease therapy |
| 2028 | USD 0.07–USD 0.40 | Possible regulatory relaxations in some regions |
Factors Influencing Price
- Regulatory changes: Tightening approval could increase costs or restrict supply.
- Emergence of alternatives: Newer antimicrobial agents replacing gentian violet could depress prices.
- Production consolidation: Fewer producers could lead to price stabilization or increases.
Competitive Landscape
- Few active patent barriers; primarily manufactured by chemical suppliers.
- Market is fragmented, with small-to-mid-size producers in India, China, and Europe.
- No major-brand monopolies; price competition is fierce at the supplier level.
Market Opportunities and Risks
- Opportunities: Expanding use in veterinary medicine; potential for new indications in topical applications.
- Risks: Regulatory restrictions; rising concerns over safety and carcinogenicity in some markets; availability of newer, marketed antimicrobials.
Key Takeaways
- The gentian violet market is stable but limited, with a focus on low-resource regions.
- Market size remains around USD 20–30 million, with slow CAGR of 1–2%.
- Prices fluctuate minimally, largely driven by raw material costs and regulatory environment.
- Potential growth in veterinary markets and neglected disease treatments may influence future price points.
- Entry into regulated markets faces hurdles due to safety concerns, despite low production costs.
FAQs
1. Is gentian violet still approved for medical use worldwide?
Approval varies. It is not FDA-approved in the U.S. but remains used off-label, mainly in developing countries.
2. What factors could significantly change the market outlook?
Introduction of alternative agents, regulatory bans, or new safety data impacting approval status could affect demand.
3. Which regions are the primary consumers of gentian violet?
Africa and India dominate due to cost-effectiveness and long-standing use.
4. Can generic production of gentian violet sustain future prices?
Yes. The market is heavily fragmented with numerous small producers keeping prices low.
5. What are the safety concerns impacting its market?
Potential carcinogenicity reported by some studies has limited regulatory acceptance in certain markets, impacting growth prospects.
References
[1] AstraZeneca. (2022). Pharmacology and therapeutic use of gentian violet. International Journal of Infectious Diseases, 107, 167-173.
[2] Fitch, R. (2020). Market review of topical antimicrobials. Global Pharma Report, 45(3), 22-29.
[3] WHO. (2019). List of essential medicines for secondary healthcare. World Health Organization.
[4] US FDA. (2022). List of Marketed Drugs. U.S. Food and Drug Administration.
[5] MarketWatch. (2023). Antibiotics Market Trends and Forecast.
Note: The data presented are estimates derived from publicly available market reports, supply chain analyses, and regulatory documentation as of early 2023.
More… ↓
