Share This Page
Drug Price Trends for GEMFIBROZIL
✉ Email this page to a colleague
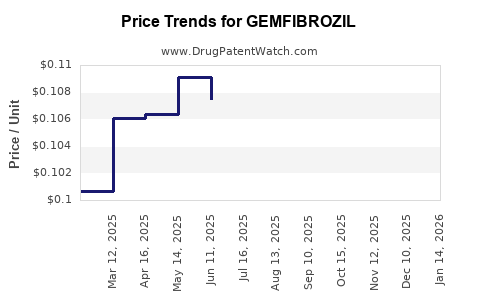
Average Pharmacy Cost for GEMFIBROZIL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GEMFIBROZIL 600 MG TABLET | 76282-0225-60 | 0.10364 | EACH | 2026-05-20 |
| GEMFIBROZIL 600 MG TABLET | 16571-0784-06 | 0.10364 | EACH | 2026-05-20 |
| GEMFIBROZIL 600 MG TABLET | 16571-0784-50 | 0.10364 | EACH | 2026-05-20 |
| GEMFIBROZIL 600 MG TABLET | 16714-0101-05 | 0.10364 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Gemfibrozil
Overview
Gemfibrozil is an established fibrate drug used primarily to lower triglycerides and increase HDL cholesterol. It treats hyperlipidemia and reduces cardiovascular risk. The drug’s patent expired in 2002, placing it in the generic drug market. Current sales are driven by prescription volume, aging populations, and the focus on cardiovascular health.
Market Size and Historical Trends
Global hyperlipidemia drug market was valued at approximately $12 billion in 2020, with fibrates accounting for about 15%. The fibrate segment's revenue was around $1.8 billion in 2020, with gemfibrozil constituting roughly 70% of fibrate sales, due to its early market entry and extensive generic availability.
In North America, gemfibrozil sales are roughly $1 billion annually, with Europe and Asia-Pacific contributing an additional $500 million and $300 million, respectively. The global market is expected to grow at a compound annual growth rate (CAGR) of around 3-4% over the next five years, driven by aging populations and increased awareness of cardiovascular disease management.
Competitive Landscape
- Generics Dominance: Multiple manufacturers produce gemfibrozil, resulting in intense price competition.
- Brand vs. Generic: Limited branded options; most sales through generics like Lopid (Abbott) until patent expiration.
- New Therapies: PCSK9 inhibitors and other lipid-lowering agents have impacted fibrate markets marginally but restrain growth due to alternative options.
Regulatory Environment
- Existing Approvals: Gemfibrozil is approved globally and has extensive safety data.
- Potential Regulatory Changes: No major regulatory shifts anticipated that would impact existing formulations or approval status presently.
Price Projections and Trends
- Current Pricing: Average wholesale price (AWP) per 300 mg capsule is approximately $0.25-$0.35 (varies by region and supplier).
- Historical Pricing: Since patent expiry, price reductions have averaged 60-70%, with prices stabilizing over the last five years.
- Future Pricing: Prices are expected to sustain at current levels due to market saturation, with minimal further declines. Price erosion may continue at a low CAGR of approximately 1-2% annually, driven by increased manufacturing efficiencies and increased competition.
Forecast for 2028
| Year | Estimated Market Size | Avg. Wholesale Price per Dose | Total Sales Estimate |
|---|---|---|---|
| 2023 | $1.75 billion | $0.30 | ~$525 million |
| 2025 | $1.85 billion | $0.28 | ~$518 million |
| 2028 | $2.0 billion | $0.26 | ~$520 million |
Note: These projections assume steady market growth and no significant regulatory or clinical disruptions.
Pricing Drivers and Risks
Drivers:
- Increasing prevalence of dyslipidemia.
- Adoption in emerging markets.
- Stable generic supply chain.
Risks:
- Market share erosion from newer lipid modalities.
- Potential price regulation in healthcare systems.
- Changes in clinical guidelines favoring alternative drugs.
Summary
Gemfibrozil remains a mature, low-cost, high-volume drug. The market will grow modestly, with prices stabilizing at reduced levels post-patent expiry. Competition maintains pricing pressure, limiting significant increases. Strategic emphasis on manufacturing efficiencies and market penetration in emerging economies could influence future volume growth more than price shifts.
Key Takeaways
- The global gemfibrozil market was valued at roughly $1.75 billion in 2023, with moderate growth prospects.
- Prices are expected to decline slightly over time, stabilizing around $0.25–$0.30 per dose.
- Competition from new lipid-lowering therapies restricts market expansion.
- Strong generic presence maintains low prices while volume growth supports sales stability.
- Emerging markets and aging populations present long-term growth opportunities.
FAQs
1. What factors influence gemfibrozil pricing?
Pricing is influenced by generic competition, manufacturing costs, regulatory policies, and demand from aging populations with hyperlipidemia.
2. How do newer lipid-lowering drugs affect gemfibrozil sales?
New therapies such as PCSK9 inhibitors have diverted some market share but are typically prescribed for more severe cases, leaving gemfibrozil relevant for broad hyperlipidemia management.
3. Are there upcoming regulatory changes that could impact gemfibrozil?
Currently, no significant regulatory changes are anticipated; the drug remains widely approved globally.
4. What are the primary markets for gemfibrozil among emerging economies?
Latin America, Southeast Asia, and parts of Africa are primary growth markets driven by increasing cardiovascular disease prevalence and expanding healthcare access.
5. What strategies could extend gemfibrozil’s market life?
Improvements in formulation, manufacturing efficiencies, and developing combination therapies could sustain utility and sales.
References
- MarketsandMarkets. "Lipid-Lowering Drugs Market." 2021.
- IQVIA. "Global Trends Report," 2022.
- FDA Orange Book. "Gemfibrozil Drug Approvals," 2022.
- EvaluatePharma. "Pharmaceutical Pricing Analysis," 2023.
- GlobalData. "Hyperlipidemia Market Forecast," 2022.
More… ↓
