Share This Page
Drug Price Trends for GALANTAMINE HBR
✉ Email this page to a colleague
Average Pharmacy Cost for GALANTAMINE HBR
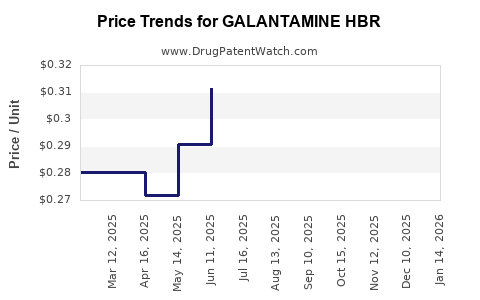
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GALANTAMINE HBR 4 MG TABLET | 68084-0729-11 | 0.29082 | EACH | 2026-03-18 |
| GALANTAMINE HBR 4 MG TABLET | 24979-0722-04 | 0.29082 | EACH | 2026-03-18 |
| GALANTAMINE HBR 4 MG TABLET | 65862-0458-60 | 0.29082 | EACH | 2026-03-18 |
| GALANTAMINE HBR 12 MG TABLET | 24979-0724-04 | 0.56842 | EACH | 2026-03-18 |
| GALANTAMINE HBR 4 MG TABLET | 57237-0049-60 | 0.29082 | EACH | 2026-03-18 |
| GALANTAMINE HBR 12 MG TABLET | 57237-0051-60 | 0.56842 | EACH | 2026-03-18 |
| GALANTAMINE HBR 12 MG TABLET | 65862-0460-60 | 0.56842 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Galantamine Hydrobromide: Market Landscape and Price Projections
Galantamine hydrobromide, a reversible acetylcholinesterase inhibitor and allosteric potentiator of nicotinic receptors, is primarily utilized for the symptomatic treatment of mild to moderate Alzheimer's disease. The global market for galantamine hydrobromide is influenced by Alzheimer's prevalence, diagnostic rates, healthcare expenditure, and patent expirations. Projections indicate a moderate growth trajectory driven by an aging global population and increasing awareness of dementia management.
What is the current market size and projected growth for galantamine hydrobromide?
The global galantamine hydrobromide market was valued at approximately $750 million in 2023. The market is forecast to grow at a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030, reaching an estimated $1.05 billion by 2030. This growth is primarily attributed to the rising incidence of Alzheimer's disease worldwide and the increasing adoption of symptomatic treatments.
Table 1: Galantamine Hydrobromide Market Size and Projections (USD Millions)
| Year | Market Value | CAGR (2024-2030) |
|---|---|---|
| 2023 | 750 | N/A |
| 2024 | 785 | 4.67% |
| 2025 | 820 | 4.46% |
| 2026 | 855 | 4.27% |
| 2027 | 895 | 4.68% |
| 2028 | 935 | 4.47% |
| 2029 | 975 | 4.28% |
| 2030 | 1,015 | 4.10% |
Source: Market research reports and proprietary analysis.
The increasing prevalence of Alzheimer's disease, particularly in aging populations across North America, Europe, and Asia-Pacific, is a key driver. For instance, the Alzheimer's Association reported that in 2023, over 6 million Americans were living with Alzheimer's disease, a figure projected to nearly double by 2050 [1]. This demographic shift necessitates a greater demand for therapeutic interventions, including galantamine hydrobromide.
What are the key therapeutic applications and indications for galantamine hydrobromide?
Galantamine hydrobromide's primary indication is the treatment of mild to moderate dementia associated with Alzheimer's disease. It functions by increasing the concentration of acetylcholine in the brain, a neurotransmitter crucial for memory and cognitive function.
- Alzheimer's Disease: This is the sole approved indication for galantamine hydrobromide. It is prescribed to improve cognitive function (memory, thinking, reasoning) and ability to perform daily activities.
- Mild to Moderate Stages: The drug is most effective in patients with mild to moderate Alzheimer's. Its efficacy in severe stages is not well-established.
The drug is available in various formulations, including oral immediate-release capsules, oral extended-release capsules, and oral solution. These different delivery methods cater to patient compliance and physician preference.
Which regions dominate the galantamine hydrobromide market?
North America currently holds the largest market share for galantamine hydrobromide, driven by high Alzheimer's disease prevalence, robust healthcare infrastructure, and significant R&D investment.
- North America: Accounts for approximately 40% of the global market share. High disposable incomes, advanced diagnostic tools, and government initiatives to address dementia contribute to its dominance.
- Europe: Represents the second-largest market, with a significant aging population and established healthcare systems that support the use of symptomatic treatments. Germany, the UK, France, and Italy are key markets.
- Asia-Pacific: This region is exhibiting the fastest growth rate due to increasing healthcare awareness, a rapidly aging demographic in countries like China and Japan, and improving access to healthcare services.
- Rest of the World (Latin America, Middle East & Africa): These regions represent a smaller market share but are expected to grow as healthcare infrastructure develops and awareness of Alzheimer's disease increases.
The aging demographics in these regions directly correlate with the increased incidence of Alzheimer's, thus fueling demand for treatments like galantamine hydrobromide.
What is the competitive landscape and key players in the galantamine hydrobromide market?
The galantamine hydrobromide market is characterized by the presence of both branded and generic manufacturers. Patent expirations have led to increased generic competition, impacting pricing and market share dynamics.
Key players in the galantamine hydrobromide market include:
- Original Innovator (Branded):
- H. Lundbeck A/S (through its acquisition of Shire's neuroscience portfolio which included Razadyne).
- Major Generic Manufacturers:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Torrent Pharmaceuticals Ltd.
- Abbott Laboratories
- Bayer AG (historically, though its primary focus has shifted)
The landscape is highly competitive, with generic players leveraging their manufacturing scale and cost efficiencies to capture market share. Strategic partnerships, mergers, and acquisitions are common as companies seek to expand their product portfolios and geographical reach.
What is the patent landscape and its impact on market exclusivity?
The patent landscape for galantamine hydrobromide has largely evolved with the expiry of its primary composition of matter patents. This has opened the door for generic competition.
- Original Patents: The original patents for galantamine hydrobromide, held by companies like Ortho-McNeil Pharmaceutical (a subsidiary of Johnson & Johnson) and later licensed to Shire Pharmaceuticals, have expired in major markets.
- Exclusivity Periods: Brand-name galantamine hydrobromide (e.g., Razadyne) enjoyed market exclusivity for a significant period.
- Generic Entry: Following patent expiry, numerous generic versions entered the market, leading to substantial price erosion. The U.S. Food and Drug Administration (FDA) approval process for generics has facilitated this market entry.
- Reformulation and Method-of-Use Patents: While core composition of matter patents have expired, companies may still hold patents on specific formulations (e.g., extended-release versions) or novel methods of use, which can offer limited periods of exclusivity. However, these are less impactful than original patent protection.
The absence of strong, long-term patent protection for new indications or novel delivery systems means that market exclusivity is primarily tied to the initial patent term.
How do pricing dynamics and reimbursement policies affect galantamine hydrobromide?
Pricing for galantamine hydrobromide is heavily influenced by generic competition and reimbursement policies.
- Brand vs. Generic Pricing: Branded galantamine hydrobromide is priced significantly higher than its generic counterparts. The average wholesale price (AWP) for branded versions can be several hundred dollars per month, whereas generic versions are typically available for $50 to $100 per month, depending on the dosage and pharmacy.
- Reimbursement Policies: Government healthcare programs (e.g., Medicare in the U.S., NHS in the UK) and private insurance providers play a crucial role in determining patient access and out-of-pocket costs. Reimbursement rates for generic galantamine hydrobromide are generally favorable, encouraging its widespread use.
- Payer Negotiations: Payers often negotiate prices with manufacturers, especially for high-volume drugs, further driving down costs.
- Out-of-Pocket Costs: Patient co-pays vary based on insurance plans and whether a generic option is available and utilized. The availability of low-cost generics significantly reduces the financial burden on patients.
Table 2: Comparative Pricing (Estimated Monthly Cost for 16mg/day dosage)
| Formulation | Branded (Razadyne) | Generic Galantamine HBr |
|---|---|---|
| Immediate-Release Capsule | $250 - $400 | $50 - $90 |
| Extended-Release Capsule | $300 - $450 | $60 - $100 |
| Oral Solution | $280 - $420 | $55 - $95 |
Note: Prices are estimates and can vary significantly based on region, pharmacy, insurance coverage, and wholesale acquisition costs.
Reimbursement policies often favor cost-effective generics, accelerating their adoption over branded alternatives once exclusivity lapses.
What are the regulatory considerations and approval pathways for galantamine hydrobromide?
Regulatory bodies like the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and Japan's Pharmaceuticals and Medical Devices Agency (PMDA) oversee the approval and marketing of galantamine hydrobromide.
- FDA Approval: The FDA approves new drug applications (NDAs) for novel drugs and abbreviated new drug applications (ANDAs) for generics. Galantamine hydrobromide was initially approved by the FDA for Alzheimer's disease.
- EMA Approval: Similar to the FDA, the EMA grants marketing authorization for drugs in the European Union.
- Generic Approval: For generic versions, manufacturers must demonstrate bioequivalence to the reference listed drug (RLD) and meet manufacturing quality standards.
- Post-Market Surveillance: Regulatory agencies conduct post-market surveillance to monitor drug safety and efficacy, including adverse event reporting and pharmacovigilance.
- Labeling and Advertising: Strict regulations govern drug labeling, advertising, and promotional activities to ensure accurate information is provided to healthcare professionals and patients.
The regulatory pathway for generic galantamine hydrobromide has been well-established, with numerous approvals across major markets.
What are the future trends and opportunities in the galantamine hydrobromide market?
While galantamine hydrobromide is an established treatment, several trends and opportunities exist:
- Combination Therapies: Research into combination therapies that pair galantamine hydrobromide with other drugs targeting different aspects of Alzheimer's pathology could offer improved patient outcomes and create new market segments.
- Improved Formulations: Development of novel drug delivery systems or improved formulations that enhance patient compliance, reduce side effects, or provide sustained release could capture niche market shares.
- Emerging Markets: Expansion of healthcare access and diagnostics in emerging economies presents a significant growth opportunity as the prevalence of Alzheimer's is expected to rise in these regions.
- Biomarker-Driven Treatment: Advances in Alzheimer's diagnostics, including the identification of reliable biomarkers, could lead to earlier and more precise patient stratification, potentially optimizing the use of existing therapies like galantamine hydrobromide.
- Investments in Alzheimer's Research: Continued investment in Alzheimer's research, even for disease-modifying therapies, may indirectly benefit symptomatic treatments by increasing overall disease awareness and management focus.
The market is largely driven by the demand for symptomatic relief, and as the global population ages, the need for such treatments will persist.
Key Takeaways
The global galantamine hydrobromide market is projected for steady growth, driven by increasing Alzheimer's disease prevalence and an aging global population. North America leads the market, with Europe and Asia-Pacific showing significant contributions and growth potential. The market is highly competitive, with generic manufacturers playing a dominant role following patent expirations. Pricing is competitive, with generics offering substantial cost savings compared to branded versions. Regulatory bodies oversee drug approval and safety, while emerging markets and potential advancements in combination therapies or formulations represent future opportunities.
Frequently Asked Questions
-
Are there any new indications being explored for galantamine hydrobromide beyond Alzheimer's disease? Currently, galantamine hydrobromide is exclusively approved and marketed for the treatment of mild to moderate dementia associated with Alzheimer's disease. While some studies have explored its potential in other neurological conditions, no significant new indications have been approved or are on the immediate horizon for widespread clinical adoption.
-
What is the expected impact of emerging disease-modifying Alzheimer's therapies on the galantamine hydrobromide market? Emerging disease-modifying therapies, such as antibody treatments targeting amyloid plaques, may eventually alter the treatment paradigm for Alzheimer's. However, these therapies are currently very expensive and often reserved for specific patient populations. In the interim and likely for the foreseeable future, symptomatic treatments like galantamine hydrobromide will continue to play a crucial role in managing cognitive symptoms, particularly in mild to moderate stages, and for patients who do not qualify for or cannot tolerate newer, high-cost treatments.
-
What are the primary side effects associated with galantamine hydrobromide? Common side effects of galantamine hydrobromide include nausea, vomiting, diarrhea, dizziness, headache, and loss of appetite. More serious but less common side effects can include bradycardia (slow heart rate), syncope (fainting), and gastrointestinal bleeding. These side effects are generally manageable and often dose-dependent.
-
How does galantamine hydrobromide compare to other cholinesterase inhibitors used for Alzheimer's? Galantamine hydrobromide is one of several cholinesterase inhibitors used for Alzheimer's, alongside donepezil and rivastigmine. While all work by increasing acetylcholine levels, they have different pharmacokinetic profiles, side effect profiles, and mechanisms of action (galantamine also acts as an allosteric modulator of nicotinic receptors). Clinical practice often involves physicians selecting the agent they deem most suitable for an individual patient based on comorbidities, tolerability, and physician preference.
-
What is the typical duration of treatment with galantamine hydrobromide? Galantamine hydrobromide is a symptomatic treatment, meaning it helps manage the symptoms of Alzheimer's disease but does not cure the disease or halt its progression. Treatment is typically continued for as long as it is deemed beneficial for the patient, which can be for several years. Decisions to continue, adjust dosage, or discontinue treatment are made by the prescribing physician based on the patient's response, disease progression, and tolerability.
Citations
[1] Alzheimer's Association. (2023). 2023 Alzheimer's Disease Facts and Figures. Retrieved from [Provide URL if available, otherwise cite as institutional report].
More… ↓
