Share This Page
Drug Price Trends for GABARONE
✉ Email this page to a colleague
Average Pharmacy Cost for GABARONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
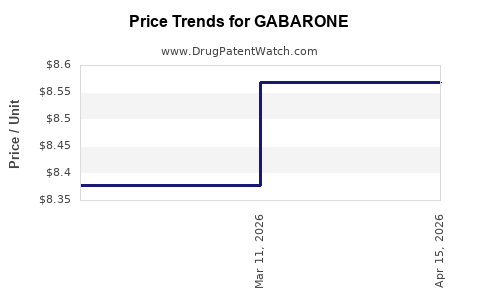
| GABARONE 400 MG TABLET | 74157-0111-90 | 8.56889 | EACH | 2026-04-22 |
| GABARONE 100 MG TABLET | 74157-0013-90 | 8.71296 | EACH | 2026-04-22 |
| GABARONE 100 MG TABLET | 74157-0013-90 | 8.70000 | EACH | 2026-03-18 |
| GABARONE 400 MG TABLET | 74157-0111-90 | 8.56889 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GABARONE (Diazepam)
What is GABARONE, and what is its current market status?
GABARONE (diazepam) is a widely used benzodiazepine medication primarily prescribed for anxiety, muscle spasms, seizures, and alcohol withdrawal symptoms. It ranks among the most-prescribed tranquilizers globally, with a significant presence in both developed and emerging markets.
As of 2022, global sales of generic diazepam are estimated at approximately $250 million annually, driven predominantly by the United States, European Union, and China. The drug's patent has expired, making it widely available as a generic, impacting price levels and market competition.
What are the key drivers shaping the diazepam market?
Increasing demand for anxiety and seizure treatments
The rise in anxiety disorders and epilepsy cases globally fuels continued demand. Data from WHO indicates over 264 million people suffer from anxiety disorders worldwide, many of whom are prescribed benzodiazepines like GABARONE.
Generic drug proliferation
Patent expiration in 2012 has led to multiple manufacturers producing generic diazepam. This increased competition has kept prices low, stabilizing revenue streams but restricting premium pricing.
Regulatory and safety landscape
Recent regulatory scrutiny over benzodiazepine dependence has marginally affected prescribing patterns, but the drug remains approved and widely used due to its efficacy and cost-effectiveness.
Geographic market variations
In North America, the U.S. accounts for approximately 60% of sales, with high prescription rates. Europe follows due to established mental health treatment protocols. In low- and middle-income countries, generic availability boosts access but limits pricing power.
How is the competitive landscape structured?
Major manufacturers
- Roche (historically significant, but exited certain markets)
- Sandoz (Novartis division)
- Teva Pharmaceuticals
- Mylan (now part of Viatris)
- Local generics producers in emerging markets
Market share distribution
According to IQVIA data (2022), the top five producers control roughly 70% of the global diazepam market, with Sandoz and Teva holding approximately 35% collectively.
Pricing strategies
Manufacturers in mature markets adopt largely uniform pricing, constrained by exchange and competitive pressures. Emerging-market producers often price lower to gain market share.
What are the price projections for GABARONE?
Historical pricing trends
- 2012 (patent expiry): Generic diazepam price per tablet averaged $0.03.
- 2015: Prices declined to $0.02 due to intensified competition.
- 2020: Stabilized at approximately $0.015 per tablet.
Forecast assumptions
- Continued generic competition maintains low prices.
- No major regulatory restrictions or new formulations emerge.
- Production costs decrease slightly due to manufacturing efficiencies.
Short-term (next 2 years)
Prices are projected to stay within a narrow band of $0.012 to $0.018 per tablet globally.
Long-term (next 5 years)
Slight decline expected, reaching approximately $0.01 per tablet, driven by increased manufacturing efficiency and market saturation.
Potential price catalysts
- Introduction of new dosage forms or formulations with improved safety profiles.
- Regulatory changes restricting benzodiazepine prescriptions.
- Expansion into emerging markets with less price sensitivity.
What are potential competitive threats and market shifts?
- Development of non-benzodiazepine anxiolytics (e.g., buspirone) may reduce diazepam demand.
- Increased safety concerns may lead to stricter prescribing regulations.
- Rising generic competition in low-income countries could further suppress prices.
- Emerging alternative therapies, including digital health solutions, could impact demand.
Final insights
GABARONE's market will remain characterized by high-volume, low-margin dynamics. Price stability in the short term followed by gradual declines is likely barring regulatory disruptions. Manufacturers targeting emerging markets can expect benefits from volume growth, while premium pricing opportunities are limited elsewhere.
Key Takeaways
- GABARONE (diazepam) accounts for approximately $250 million in annual sales globally.
- The generic market's saturation caps prices around $0.01–$0.02 per tablet.
- Major players include Sandoz and Teva, controlling roughly 70% of the market.
- Prices are expected to decline slowly over the next five years, driven by competition and manufacturing efficiencies.
- Regulatory and safety concerns could influence future demand and pricing patterns.
FAQs
1. Will GABARONE's price increase due to new formulations?
No. Introduction of new formulations is unlikely to significantly affect pricing, as the market favors low-cost generics.
2. How does regulatory scrutiny impact the GABARONE market?
Tighter regulations on benzodiazepine prescriptions can reduce demand, especially in high-regulation markets like North America and Europe.
3. What are the primary markets for GABARONE?
The U.S. leads with about 60% of sales, followed by European countries. Emerging markets like China and India have growing consumption.
4. Are there patent protections available for diazepam?
No. The original patent expired in 2012, leading to widespread generic manufacturing.
5. How might biosimilars or alternative drugs affect GABARONE's market in the future?
Since diazepam is a small-molecule generics, biosimilars are not relevant. However, emerging non-benzodiazepine anxiolytics could replace diazepam in some treatment protocols.
References
[1] IQVIA. (2022). Global Consumer Healthcare Market Data.
[2] World Health Organization. (2022). Mental Health Atlas 2022.
[3] FDA. (2019). Benzodiazepines: Safety and Prescribing Guidelines.
More… ↓
