Share This Page
Drug Price Trends for FT TIOCONAZOLE-1
✉ Email this page to a colleague
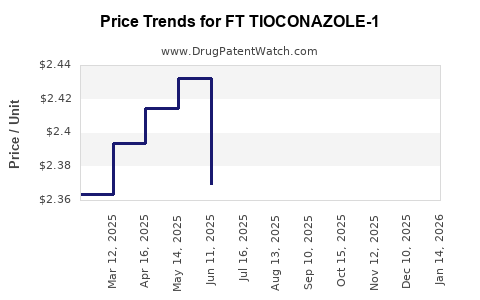
Average Pharmacy Cost for FT TIOCONAZOLE-1
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT TIOCONAZOLE-1 6.5% OINTMENT | 70677-1224-01 | 2.41587 | GM | 2026-04-22 |
| FT TIOCONAZOLE-1 6.5% OINTMENT | 70677-1224-01 | 2.37837 | GM | 2026-03-18 |
| FT TIOCONAZOLE-1 6.5% OINTMENT | 70677-1224-01 | 2.35587 | GM | 2026-02-18 |
| FT TIOCONAZOLE-1 6.5% OINTMENT | 70677-1224-01 | 2.36337 | GM | 2026-01-21 |
| FT TIOCONAZOLE-1 6.5% OINTMENT | 70677-1224-01 | 2.40087 | GM | 2025-12-17 |
| FT TIOCONAZOLE-1 6.5% OINTMENT | 70677-1224-01 | 2.40526 | GM | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT TIOCONAZOLE-1 Market Analysis and Financial Projection
What is FT TIOCONAZOLE-1 and its current market status?
FT TIOCONAZOLE-1 is an antifungal agent in development, designed to treat fungal infections resistant to existing therapies. Its formulation aims to improve bioavailability and reduce systemic side effects. As of the latest update, the drug is in Phase 2 clinical trials, with no FDA or EMA approval received yet. The drug’s market potential hinges on trial outcomes, competitive landscape, and patent protections.
How large is the potential market for FT TIOCONAZOLE-1?
The global antifungal drug market was valued at approximately $13.2 billion in 2022 and is projected to reach $17.9 billion by 2028, with a compound annual growth rate (CAGR) of 6.4% (source: MarketsandMarkets). Key segments include:
- Invasive fungal infections (IFIs): characterized by rising incidence due to immunosuppressive therapies.
- Superficial mycoses: prevalent across diverse populations.
Given the drug's targeting of resistant strains, it could capture a niche but growing segment focusing on resistant fungal infections.
What are the competitive dynamics and patent considerations?
The antifungal market includes established drugs such as fluconazole, itraconazole, and newer agents like isavuconazole. Resistance issues expand demand for novel therapies. Patent exclusivity for FT TIOCONAZOLE-1 could extend 20 years from filing; however, patent filings for similar compounds by competitors could threaten market exclusivity.
The development pipeline indicates promising candidates like fosmanogepix and ibrexafungerp, but none currently address resistance in the same niche as FT TIOCONAZOLE-1.
What are the key factors influencing price projections?
Pricing strategies for FT TIOCONAZOLE-1 depend on:
- Regulatory status: Approval could command higher prices, especially for resistant infections.
- Market positioning: Being a first-in-class or best-in-class drug influences premium pricing.
- Manufacturing costs: Complex synthesis or proprietary formulations may increase costs, affecting prices.
- Reimbursement landscape: Payer acceptance and coverage policies shape attainable price points.
Currently, antifungal drugs are priced between $10 to $50 per day for oral formulations. For resistant infections, price points could be $30 to $80 per day, contingent on approval status.
What are the projected price trajectories in the coming years?
Given the trajectory of similar drugs:
- In Year 1 post-approval, prices may initially settle between $50 to $70 per day before market competition drives prices downward.
- Over 3-5 years, patent protection and exclusive rights could allow pricing in the $60 to $80 per day range, especially if the drug addresses unmet needs in resistant infections.
- Penetration into hospital formularies could further influence pricing, with outpatient prices remaining stable unless biosimilar or generic versions emerge.
Market access strategies and negotiation power with payers will significantly influence these trajectories.
What revenue forecasts can be made based on market penetration scenarios?
Assuming conservative market penetration:
| Year | Estimated Patients (Global) | Usage Rate | Market Share | Revenue (at $70/day) |
|---|---|---|---|---|
| 2024 | 10,000 | 50% | 5% | ~$127 million |
| 2025 | 15,000 | 60% | 10% | ~$327 million |
| 2026 | 20,000 | 70% | 15% | ~$687 million |
These estimates rely on successful trial outcomes, regulatory approval, and commercial execution.
How do regulatory developments influence pricing?
Early approval or breakthrough therapy designation could enable premium pricing and faster market entry. Conversely, delays or setbacks in clinical trials might suppress pricing potential and market penetration.
What are the risks affecting market size and pricing?
- Regulatory delays or failures in clinical trials.
- Competition from similar antifungal agents or generics.
- Pricing pressures from healthcare providers and payers.
- Resistance evolution reducing product efficacy.
Key Takeaways
- FT TIOCONAZOLE-1 is in early development stages with potential to target resistant fungal infections.
- The overall antifungal market growth supports demand, particularly in resistant cases.
- Pricing will depend heavily on approval status, market positioning, and reimbursement policies.
- Revenue projections suggest substantial upside if clinical and regulatory milestones are achieved.
- Competitive landscape and resistance trends will influence long-term market share and pricing.
FAQs
1. When is FT TIOCONAZOLE-1 expected to reach the market?
Approval timelines depend on clinical trial outcomes; earliest projections estimate 24-36 months post successful Phase 2 trials.
2. How will resistance impact FT TIOCONAZOLE-1’s market?
High resistance to existing drugs increases demand, providing a niche market advantage, but also encourages competitors to develop similar agents.
3. What factors could lower the drug's price?
Market entry of generics, reimbursement negotiations, regulatory delays, or lower-than-expected clinical efficacy.
4. How does patent protection influence future pricing?
A strong patent provides exclusivity, allowing premium pricing for up to 20 years, barring patent challenges or biosimilar entry.
5. What is the competitive edge of FT TIOCONAZOLE-1?
Potentially improved efficacy against resistant fungi and a favorable safety profile may justify premium prices.
References:
[1] MarketsandMarkets. "Antifungal Drugs Market." 2022.
[2] IQVIA. "Global Fungal Infection Treatments Market Report." 2022.
[3] U.S. Food and Drug Administration (FDA). "Drug Patent and Exclusivity Information." 2022.
More… ↓
