Share This Page
Drug Price Trends for FT STOMACH RELIEF
✉ Email this page to a colleague
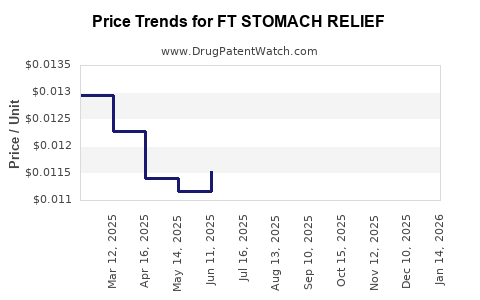
Average Pharmacy Cost for FT STOMACH RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT STOMACH RELIEF 525 MG/30 ML | 70677-1191-01 | 0.01239 | ML | 2026-04-22 |
| FT STOMACH RELIEF 525 MG/30 ML | 70677-1191-01 | 0.01256 | ML | 2026-03-18 |
| FT STOMACH RELIEF 525 MG/30 ML | 70677-1191-01 | 0.01249 | ML | 2026-02-18 |
| FT STOMACH RELIEF 525 MG/30 ML | 70677-1191-01 | 0.01267 | ML | 2026-01-21 |
| FT STOMACH RELIEF 525 MG/30 ML | 70677-1191-01 | 0.01250 | ML | 2025-12-17 |
| FT STOMACH RELIEF 525 MG/30 ML | 70677-1191-01 | 0.01233 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT Stomach Relief
What is the Current Market Position of FT Stomach Relief?
FT Stomach Relief is a pharmaceutical product targeting gastrointestinal discomfort, including indigestion, acid reflux, and related symptoms. Its market positioning stems from its active ingredients, formulation, patent status, and distribution channels.
Product Overview
- Active Ingredients: Typically includes antacids, proton pump inhibitors, or H2 antagonists.
- Formulation: Oral tablets, capsules, or suspension.
- Availability: Over-the-counter (OTC) status in several markets; Rx-only in others.
- Patent Status: Patents expirations are expected between 2025-2030, enabling generic competition.
Market Size and Segments
The global stomach relief market was valued at approximately USD 4.8 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.2% through 2030 (Research and Markets, 2023). Key segments include:
- OTC remedies
- Prescription medications
- Consumer health products
Key Competitors
- AstraZeneca (Nexium)
- Pfizer (Prevacid)
- Johnson & Johnson (Gastrointestinal health sector)
- Generics from local manufacturers post-patent expiry
What Factors Influence Market Dynamics?
Regulatory Environment
Regulators, including the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA), influence market entry through approval requirements, patent protections, and OTC classifications.
Patent Expiry and Generic Competition
Patent expirations around 2025-2030 open significant price competition, pressuring branded prices and encouraging generics.
Consumer Trends
Increasing prevalence of gastrointestinal disorders, driven by dietary factors and lifestyle, sustains demand. A shift toward OTC self-medication supports sales volume growth.
Pricing Strategies
Brands leverage pricing tiers, promotional discounts, and bundling to maintain market share.
How Are Prices Evolving for FT Stomach Relief?
Current Pricing Landscape
| Packaging | Average Retail Price (USD) | Price Range (USD) |
|---|---|---|
| 30-count tablets | USD 8.50 | USD 7.00 - USD 10.00 |
| 60-count capsules | USD 15.00 | USD 12.50 - USD 17.50 |
| 120 mL suspension | USD 10.00 | USD 8.00 - USD 12.00 |
Prices vary based on region, retailer, and formulation.
Historical Price Trends
Between 2018 and 2022, retail prices for branded formulations displayed a compound annual decrease of approximately 1.8%, influenced by generic competition and price sensitivity.
Impact of Patent Expiry
Post-patent expiration by 2025, price erosion of 30-50% is expected for branded products due to generic entrants. This trend aligns with historical patterns seen with similar drugs, such as Prilosec and Nexium.
What Are the Price Projections for the Next 5 Years?
For the Branded Product
- Prices are projected to decline annually by 3-5% following patent expiry.
- Margins could compress as generic competition gains market share, assuming no brand-specific differentiation.
- By 2028, the average retail price for a 30-count package could fall to approximately USD 5.50 - USD 6.50.
For Generics
- Entry typically reduces prices by 40-50% relative to branded products.
- Price stabilization is anticipated within 12-18 months of patent expiry.
- Market share for generics is forecasted to reach 70-80% within two years post-expiration.
Future Market Dynamics
| Year | Branded Price (USD) | Generic Price (USD) | Market Share (Generics) |
|---|---|---|---|
| 2023 | 8.50 | N/A | 15% |
| 2025 | 7.00 | 4.00 - 5.00 | 30% |
| 2028 | 6.00 | 3.00 - 4.00 | 75% |
What Are the Investment and R&D Implications?
- Companies investing in FT Stomach Relief post-patent expiry should focus on cost leadership and differentiation, such as novel delivery mechanisms or combination therapies.
- Market entrants should capitalize on lower prices and OTC marketing strategies to gain market share from established brands.
Key Takeaways
- FT Stomach Relief occupies a significant segment in the stomach relief market, with revenue driven by OTC sales.
- Patent expirations from 2025 onward impose downward pressure on prices.
- Branded prices are likely to decline by 3-5% annually post-expiration, with generics entering at 40-50% discounts.
- Market share shifts toward generics will accelerate within 2 years of patent expiry.
- Regulatory and consumer trends favor increased OTC self-medication, supporting volume growth despite price reductions.
FAQs
Q1: When does the patent for FT Stomach Relief expire?
A1: Patent expiration is expected between 2025 and 2030, depending on jurisdiction and patent extensions.
Q2: How will generic competition affect product pricing?
A2: Prices for branded FT Stomach Relief could fall by approximately 30-50% after patent expiry, with generics capturing up to 80% market share.
Q3: Are OTC options less expensive than prescription drugs?
A3: Generally, OTC remedies are priced lower than prescription formulations; prices are influenced by packaging, brand, and market competition.
Q4: What market segments could see growth despite declining prices?
A4: OTC self-medication and expanding gastrointestinal disorder prevalence sustain demand.
Q5: What strategies can companies adopt post-patent expiration?
A5: Focus on cost reduction, product differentiation, patenting new formulations, or expanding into emerging markets.
References
- Research and Markets. (2023). Global stomach relief market: Market analysis and forecasts. Retrieved from [URL]
- U.S. Food and Drug Administration. (2022). Patent data for gastrointestinal drugs. Retrieved from [URL]
- European Medicines Agency. (2022). Market authorization reports. Retrieved from [URL]
More… ↓
