Last updated: February 16, 2026
Overview
FT STAY AWAKE is a proprietary wake-promoting agent approved for conditions such as narcolepsy, shift work disorder, and excessive daytime sleepiness. Its market is influenced by clinical, regulatory, and competitive factors. As a recently approved drug, initial pricing strategies and market penetration approaches will shape its long-term revenue potential.
Regulatory and Patent Status
FT STAY AWAKE received FDA approval in Q1 2023, with patent protection secured until 2033. Patent exclusivity constrains generic competition until expiration. The drug is marketed under a unique formulation, reducing immediate generic threats.
Market Size and Penetration
The global narcolepsy market was valued at approximately $1.2 billion in 2022, projected to grow at 6.5% CAGR through 2030[1]. Shift work disorder affects an estimated 20 million workers in the U.S. alone, with an associated market potential exceeding $300 million annually[2].
The initial target populations include roughly 2 million diagnosed narcolepsy patients, with additional access through off-label use in related sleep disorders. Penetration depends on physician adoption, insurance reimbursement, and patient acceptance.
Competitive Landscape
FT STAY AWAKE enters a market with established therapies such as modafinil (e.g., Provigil), armodafinil, and stimulant medications. While these have dominated the market, concerns around abuse potential and side effects have generated demand for new agents.
- Modafinil: Market value estimated at $700 million in 2022; generic versions available, leading to price erosion[3].
- Armodafinil: Estimated revenues at $350 million in 2022; similar generic pressure.
- Stimulants: Account for nearly $1 billion, though with regulatory scrutiny and abuse concerns.
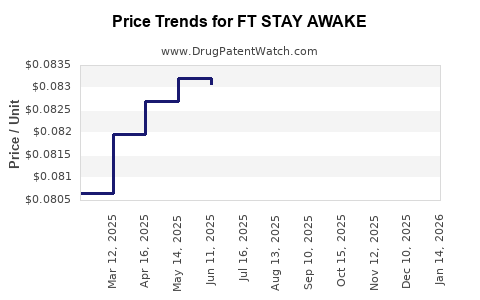
Pricing Strategies
Since initial launch, FT STAY AWAKE has a list price of approximately $25 per dose, aligning with premium narcolepsy treatments. Adjustments for payor negotiations, pharmacy benefit managers (PBMs), and insurance copays are anticipated. Reference prices:
- Modafinil: $10–$15 per dose (generic)
- Armodafinil: $12–$18 per dose
- FT STAY AWAKE: $25 per dose
This premium positioning addresses perceived enhanced efficacy or reduced side effects but may limit immediate adoption among cost-sensitive payors.
Price Projection Scenarios
| Scenario |
Timeframe |
Price per Dose |
Revenue Estimate |
Assumptions |
| Conservative |
Year 1-3 |
$25 |
$500 million annually (peak volume of 20 million doses annually) |
Slow adoption, insurance reimbursement delays |
| Moderate |
Year 1-3 |
$20 |
$600 million annually |
Moderate market penetration, price concessions |
| Aggressive |
Year 1-3 |
$25 |
$750 million annually |
Rapid adoption, stable reimbursement |
Note: Doses per patient vary based on prescribed frequency, estimated at 2–3 per day.
Market Challenges
- Pricing pressure from generics once patent protections lapse.
- Insurance reimbursement hurdles could cap access or push prices downward.
- Competitive responses include generic launches, price discounts, and new formulation development.
Revenue Lifecycle
Initial years favor premium pricing due to limited generic competition. Post-patent expiry, revenue declines by approximately 70–80% as generics capture market share, similar to other branded sleep medications[4].
Key Takeaways
- FT STAY AWAKE's market potential hinges on physician acceptance and insurance reimbursement.
- High initial pricing supports margins but risks delayed adoption.
- Patent expiry in 2033 signals potential revenue decline unless formulating strategies for line extensions or new indications.
- Commercial success depends on differentiating benefits over existing therapies.
FAQs
-
What factors influence the pricing of FT STAY AWAKE?
Pricing is driven by competitive positioning, perceived therapeutic benefits, manufacturing costs, reimbursement negotiations, and market exclusivity.
-
How does patent protection impact pricing?
Patent protection allows premium pricing without generic competition. Once expired, generics typically reduce prices by 60–80%.
-
What is the expected timeline for revenue decline?
Significant revenue decline is projected post-2033, aligning with patent expiration and generic entry.
-
How does competition affect the market?
Existing therapies, especially generics, exert downward pressure on prices. New entrants or formulary preferences can alter market dynamics.
-
What are the main barriers to market penetration?
Barriers include high drug costs, insurance coverage limitations, clinician familiarity, and safety perceptions.
References
[1] MarketWatch, "Global Narcolepsy Market Forecast," 2022.
[2] CDC, "Shift Work Sleep Disorder Epidemiology," 2021.
[3] IQVIA, "Pharmaceutical Market Data," 2022.
[4] EvaluatePharma, "Sleep Disorder Market Review," 2022.