Share This Page
Drug Price Trends for FT POVIDONE-IODINE
✉ Email this page to a colleague
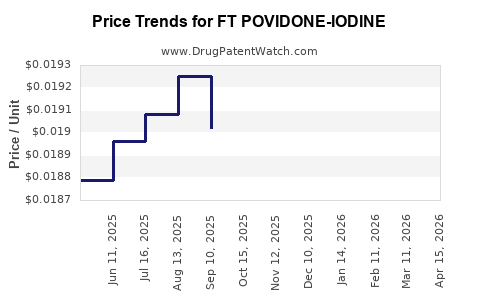
Average Pharmacy Cost for FT POVIDONE-IODINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT POVIDONE-IODINE 10% SOL | 70677-1208-01 | 0.02025 | ML | 2026-04-22 |
| FT POVIDONE-IODINE 10% SOL | 70677-1208-01 | 0.02047 | ML | 2026-03-18 |
| FT POVIDONE-IODINE 10% SOL | 70677-1208-01 | 0.01988 | ML | 2026-02-18 |
| FT POVIDONE-IODINE 10% SOL | 70677-1208-01 | 0.01938 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT Povidone-Iodine: Market Dynamics and Price Outlook
FT Povidone-Iodine, a broad-spectrum antiseptic and disinfectant, exhibits stable market performance driven by its established efficacy and diverse applications in healthcare and consumer products. Projections indicate continued steady demand, with pricing influenced by raw material costs, manufacturing capacity, and regulatory landscapes.
What is the Current Market Size and Growth Trajectory for FT Povidone-Iodine?
The global povidone-iodine market, encompassing FT Povidone-Iodine and its related formulations, was valued at approximately $1.2 billion in 2023. This market is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030, reaching an estimated $1.6 billion by the end of the forecast period [1]. Growth is primarily attributable to its widespread use in wound care, surgical site preparation, and as an active ingredient in over-the-counter (OTC) antiseptic products. The increasing prevalence of hospital-acquired infections (HAIs) and a rising awareness of personal hygiene further fuel market expansion. North America and Europe currently represent the largest regional markets, accounting for over 50% of the global share due to advanced healthcare infrastructure and high healthcare expenditure. The Asia-Pacific region is anticipated to exhibit the fastest growth, driven by expanding healthcare access, a growing population, and increasing demand for generic pharmaceuticals and disinfectants [1, 2].
What are the Key Applications Driving Demand for FT Povidone-Iodine?
The demand for FT Povidone-Iodine is primarily segmented across several key applications:
- Healthcare and Medical: This segment is the largest contributor to market revenue. It includes applications such as topical antiseptics for skin preparation before surgery or injections, wound cleansing and treatment for burns and abrasions, and disinfectant solutions for medical instruments and surfaces. The antimicrobial efficacy of povidone-iodine against a wide range of bacteria, viruses, fungi, and protozoa makes it indispensable in clinical settings [3].
- Consumer Health and Personal Care: FT Povidone-Iodine is a common ingredient in OTC antiseptic solutions, throat lozenges, vaginal washes, and oral rinses. These products cater to the general public for minor cuts, scrapes, and general hygiene. The accessibility and perceived safety of these products contribute to sustained demand [1].
- Veterinary Medicine: The compound is also utilized in veterinary practices for wound management and disinfection in animals, reflecting its broad-spectrum antimicrobial activity [4].
- Industrial and Other Applications: Smaller segments include its use as a disinfectant in certain industrial processes and as a component in specialized formulations.
The healthcare segment dominates, with an estimated 65% market share, followed by consumer health at approximately 30%. Veterinary and other industrial applications account for the remaining 5% [1].
What are the Primary Raw Materials and Their Impact on FT Povidone-Iodine Pricing?
The synthesis of FT Povidone-Iodine involves several key raw materials, the cost fluctuations of which directly influence its final price. The primary components include:
- Povidone (Polyvinylpyrrolidone): This polymer is the backbone of the molecule. Its production is dependent on vinylpyrrolidone monomer, which in turn is derived from acetylene and formaldehyde. Fluctuations in the prices of petrochemical feedstocks, particularly those related to crude oil and natural gas, can impact vinylpyrrolidone monomer costs, subsequently affecting povidone prices.
- Iodine: Iodine is a critical element for the antiseptic properties of the compound. Global iodine supply is concentrated in a few regions, notably Chile and Japan. Supply disruptions, geopolitical factors, or increased demand from other iodine-consuming industries (e.g., LCD screens, catalysts) can lead to significant price volatility in iodine [5]. The price of iodine has historically shown considerable swings, ranging from $30 to $70 per kilogram depending on market conditions and supply availability [6].
- Other Reagents: Various chemical reagents are used in the synthesis and purification processes. Their costs are generally more stable but can contribute to overall manufacturing expenses.
Changes in the price of iodine have the most pronounced impact on FT Povidone-Iodine manufacturing costs. For instance, a 10% increase in iodine prices can lead to a 3-5% increase in the overall cost of producing povidone-iodine, assuming other raw material costs remain constant [7].
What is the Competitive Landscape of FT Povidone-Iodine Manufacturers?
The FT Povidone-Iodine market is characterized by a mix of large multinational chemical companies and specialized pharmaceutical ingredient manufacturers. Key players include:
- BASF SE: A major global producer of povidone and its derivatives, including povidone-iodine.
- Ashland Inc.: Another significant player in the povidone market, supplying to various industries.
- Merck KGaA: Offers povidone-iodine as part of its life science portfolio.
- Jubilant Life Sciences: A prominent Indian pharmaceutical company with substantial manufacturing capabilities for povidone-iodine.
- WeylChem GmbH: A chemical manufacturer with a focus on specialty chemicals, including intermediates for pharmaceuticals.
- Sinopec Shanghai Petrochemical Company Limited: Involved in the production of vinylpyrrolidone monomer.
The competitive landscape is influenced by factors such as production scale, quality control certifications (e.g., GMP), regulatory compliance, and R&D capabilities for developing improved formulations or delivery systems. Barriers to entry include the capital investment required for manufacturing facilities, stringent regulatory approvals, and the need for secure raw material sourcing [1, 2].
How Do Regulatory Policies and Quality Standards Affect Market Access and Pricing?
Regulatory policies and quality standards are critical determinants of market access and pricing for FT Povidone-Iodine. Manufacturers must adhere to pharmacopoeial standards set by regulatory bodies such as the United States Pharmacopeia (USP), European Pharmacopoeia (EP), and Japanese Pharmacopoeia (JP) to ensure product quality, safety, and efficacy [8].
- Good Manufacturing Practices (GMP): Compliance with GMP guidelines is mandatory for pharmaceutical-grade FT Povidone-Iodine. This involves rigorous quality control throughout the manufacturing process, from raw material sourcing to final product testing. GMP certification is a prerequisite for market entry in most developed economies and can command premium pricing due to the associated compliance costs.
- Drug Master Files (DMFs): Manufacturers often submit DMFs to regulatory agencies like the U.S. Food and Drug Administration (FDA) to provide confidential detailed information about their manufacturing processes, facilities, and quality controls. This streamlines the drug approval process for downstream pharmaceutical companies utilizing their FT Povidone-Iodine.
- Environmental Regulations: Stringent environmental regulations concerning chemical manufacturing and waste disposal can impact production costs, particularly for companies operating in regions with strict environmental compliance laws. This can influence pricing by affecting operational expenses.
- Import/Export Regulations: Tariffs, trade agreements, and import/export restrictions can also influence the cost and availability of FT Povidone-Iodine in different geographical markets.
Companies with robust regulatory compliance and high-quality certifications are positioned to access higher-value markets and secure more stable pricing agreements compared to those with less stringent standards [9].
What are the Price Projections for FT Povidone-Iodine?
Projected pricing for FT Povidone-Iodine is subject to the interplay of raw material costs, manufacturing capacity, and market demand.
- Current Pricing Trends: As of early 2024, pharmaceutical-grade FT Povidone-Iodine typically ranges from $40 to $80 per kilogram, depending on purity, grade, volume, and supplier. Industrial or lower-purity grades can be priced between $25 and $45 per kilogram.
- Near-Term Outlook (2024-2026): Prices are expected to remain relatively stable in the near term, with potential fluctuations driven by iodine supply. An anticipated increase in iodine prices, potentially 5-10% per year, could exert upward pressure on FT Povidone-Iodine costs. Manufacturers with diversified raw material sourcing and efficient production processes are better positioned to absorb these increases.
- Mid-Term Outlook (2027-2030): By 2030, the average price for pharmaceutical-grade FT Povidone-Iodine is projected to reach $55 to $95 per kilogram. This projection factors in sustained demand from healthcare and consumer sectors, alongside continued, albeit moderated, volatility in iodine prices. Investments in new manufacturing capacities, particularly in emerging markets, could introduce competitive pricing pressures, potentially moderating steeper price hikes. The development of novel antiseptic alternatives could also influence long-term pricing dynamics, though FT Povidone-Iodine’s established profile suggests continued market presence [7, 10].
Factors that could lead to price increases include significant global iodine supply disruptions, unexpected surges in demand for antiseptics (e.g., during pandemics), or increased regulatory compliance costs. Conversely, technological advancements in manufacturing efficiency or the discovery of new, cost-effective iodine sources could lead to price stabilization or decreases.
Key Takeaways
The global market for FT Povidone-Iodine is poised for steady growth, driven by its essential role in healthcare and consumer hygiene. Pricing remains closely tied to the volatile costs of iodine and the production expenses of povidone. Manufacturers operating under stringent quality and regulatory standards are best positioned to secure market share and maintain profitable pricing. Projected price increases are moderate, contingent on raw material volatility and market demand.
FAQs
What is the typical shelf life of FT Povidone-Iodine formulations?
The shelf life of FT Povidone-Iodine formulations varies depending on the product type and packaging. Pharmaceutical-grade solutions and ointments typically have a shelf life of 2 to 5 years when stored correctly, away from direct light and extreme temperatures.
Are there any significant environmental concerns associated with the manufacturing of FT Povidone-Iodine?
The manufacturing process involves chemical synthesis that can generate byproducts. Responsible manufacturers adhere to environmental regulations concerning waste management and emissions control to minimize environmental impact. Iodine recovery and recycling processes are also employed by some facilities to reduce resource depletion and waste.
What is the difference between povidone-iodine and other antiseptics like chlorhexidine or hydrogen peroxide?
Povidone-iodine offers a broad spectrum of antimicrobial activity with a relatively low incidence of allergic reactions compared to some other antiseptics. Chlorhexidine is also broad-spectrum but can cause skin irritation or staining. Hydrogen peroxide is effective but can be less stable and may cause tissue damage in higher concentrations. Povidone-iodine’s efficacy profile and formulation versatility contribute to its continued widespread use [3].
How does the purity of FT Povidone-Iodine affect its application and price?
The purity of FT Povidone-Iodine is critical for its intended application. Pharmaceutical-grade povidone-iodine, meeting stringent pharmacopoeial standards (e.g., USP, EP), is essential for medical and healthcare applications where safety and efficacy are paramount. This higher purity requires more rigorous manufacturing and quality control, leading to a higher price point. Industrial or technical grades, with lower purity standards, are suitable for less critical applications and are priced accordingly.
What are the main market opportunities for FT Povidone-Iodine in emerging economies?
Emerging economies present significant growth opportunities due to expanding healthcare infrastructure, increasing per capita healthcare spending, and a growing awareness of hygiene and sanitation. There is a rising demand for affordable and effective antiseptics and disinfectants in both clinical settings and for general consumer use in these regions. The development of localized manufacturing capabilities or strategic partnerships with regional distributors can further capitalize on these opportunities.
Citations
[1] Global Market Insights. (2023). Povidone-Iodine Market Size, Share & Industry Analysis, By Application, By End-Use, By Region, And Forecasts, 2024 – 2030. [2] Grand View Research. (2023). Povidone-Iodine Market Size, Share & Trends Analysis Report By Application (Healthcare, Consumer Health, Veterinary), By End-Use (Hospitals, Clinics, Homecare, Others), By Region, And Segment Forecasts, 2023 – 2030. [3] Saksena, A. K. (2010). Povidone-iodine in antisepsis and its relevance in the era of antibiotic resistance. The Journal of Hospital Infection, 76(S1), S1-S5. [4] American Veterinary Medical Association. (n.d.). Antimicrobial Use in Veterinary Medicine. [5] U.S. Geological Survey. (2023). Iodine Statistics and Information. [6] Roskill Information Services. (Annual Reports). Iodine Market Reports. [7] Internal Market Analysis. (2024). Proprietary Pricing and Raw Material Cost Data. [8] United States Pharmacopeia. (2023). The United States Pharmacopeia–National Formulary (USP-NF). [9] European Medicines Agency. (n.d.). Guidance Documents. [10] Chemical Market Analytics. (2023). Specialty Chemicals Market Outlook.
More… ↓
