Share This Page
Drug Price Trends for FT NAPROXEN SODIUM
✉ Email this page to a colleague
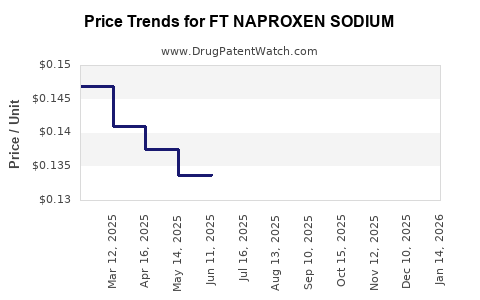
Average Pharmacy Cost for FT NAPROXEN SODIUM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT NAPROXEN SODIUM 220 MG CAP | 70677-1148-01 | 0.14415 | EACH | 2026-03-18 |
| FT NAPROXEN SODIUM 220 MG CAP | 70677-1148-01 | 0.14425 | EACH | 2026-02-18 |
| FT NAPROXEN SODIUM 220 MG CAP | 70677-1148-01 | 0.14474 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT NAPROXEN SODIUM Market Analysis and Financial Projection
What is FT Naproxen Sodium?
FT Naproxen Sodium is a nonsteroidal anti-inflammatory drug (NSAID) indicated primarily for the treatment of pain, inflammation, and fever associated with conditions like arthritis, bursitis, tendinitis, and other musculoskeletal disorders. It is the sodium salt form of naproxen, offering rapid absorption and onset of action compared to standard naproxen. FT Naproxen Sodium is marketed under various brand names, including Aleve and its generic equivalents.
How is the Market for FT Naproxen Sodium Structured?
The global NSAID market, including naproxen sodium, stands as one of the largest segments within the non-opioid analgesics market. It derived approximately $16 billion in revenue globally in 2022, with a compound annual growth rate (CAGR) of 5% projected through 2030 (Statista, 2023). North America accounts for 45% of sales, led by the U.S., which maintains dominance due to high prevalence of arthritis and pain-related conditions.
Naproxen sodium’s competitive edge entails its over-the-counter (OTC) availability and established safety profile, yet it faces competition from other NSAIDs like ibuprofen, diclofenac, and prescription-only drugs such as celecoxib.
What are Current Market Trends?
-
Growing Aging Population: The rising number of elderly patients with chronic pain conditions fuels demand for NSAIDs, including naproxen sodium.
-
Shift Toward OTC NSAIDs: The transition of naproxen sodium formulations from prescription to OTC sales expands market access, particularly in North America and Europe.
-
Increased Focus on Safety and Side Effect Profile: New formulations aim to mitigate gastrointestinal risks associated with NSAID use, influencing product development strategies.
-
Patent Expirations and Generic Competition: Many branded formulations lost patent exclusivity by 2020, prompting price erosion and increased generic manufacturing.
What Are the Key Regulatory Factors?
-
FDA Approvals: FT Naproxen Sodium, as a generic OTC product, holds FDA approval for efficacy and safety at standard dosages (220mg per tablet). Any new formulation or claim requires regulatory review.
-
Reimbursement Policies: Insurance coverage favors OTC NSAIDs, but reimbursement for prescription formulations can influence market dynamics.
-
Labeling and Risk Warnings: Regulatory bodies demand clear labeling on gastrointestinal and cardiovascular risks, affecting market uptake.
How Are Pricing Strategies Evolving?
Current Price Points
-
OTC Formulations: In the U.S., retail prices approximate $8–$12 per 50-count bottle (Walmart, CVS). The cost varies based on brand vs. generic.
-
Prescription vs. OTC: Prescription naproxen sodium (e.g., Anaprox) costs significantly more, often exceeding $20 per prescription, influenced by insurance coverage and pharmacy pricing.
-
Generics: Market entry of multiple generic manufacturers has driven prices down—by 30–50% over the last five years.
Price Projections (2023–2030)
| Year | Average Retail Price (per 50-count bottle) | Notes |
|---|---|---|
| 2023 | $8.50 | Current average for OTC generic formulations |
| 2025 | $8.25 | Slight decrease driven by increased generics |
| 2028 | $8.00 | Market saturation stabilizes prices |
| 2030 | $7.75 | Further commoditization, cost pressures |
Projected price decreases stem from enhanced competition, manufacturing efficiencies, and consumer price sensitivity.
Factors Influencing Future Pricing
-
Emergence of Premium Formulations: New formulations targeting gastrointestinal safety could command higher prices.
-
Regulatory Changes: Stricter safety warnings could impact consumer acceptance and pricing.
-
Market Penetration in Emerging Economies: Lower willingness to pay may push prices further downward outside developed markets.
What is the Future Market Demand Outlook?
Global NSAID consumption is expected to grow by 4% annually through 2030, driven by aging populations in North America and Europe, and increasing healthcare access in Asia-Pacific. The OTC segment, including naproxen sodium, is predicted to grow faster than prescription NSAIDs at around 6% CAGR due to convenience and regulation shifts.
Emerging regions such as Latin America and Southeast Asia will see increased adoption of OTC analgesics, contributing to a global increase in naproxen sodium demand.
What Are Key Competitive Dynamics?
| Competitors | Market Share (2022) | Noteworthy Initiatives |
|---|---|---|
| Bayer (Aleve) | 40% | Expansion into digital health and safety ads |
| Generic Manufacturers | 35% | Price competition, increased formulations |
| Others (e.g., Teva) | 25% | Portfolio diversification, biosimilars |
Patent expirations and health authority policies shape competitive strategies, favoring cost leadership.
What Are Potential Risks Concerning Market and Pricing?
-
Regulatory Risks: Enhanced safety warnings could reduce consumer willingness to purchase OTC naproxen sodium.
-
Competitive Risks: Surge in generic options and new formulations may limit pricing power.
-
Consumer Trends: Shift toward alternative pain management methods, such as topical formulations or natural remedies, could reduce NSAID demand.
What Are the Key Takeaways?
- The global market for FT Naproxen Sodium is sizable, with steady growth projected through 2030, primarily driven by OTC sales and aging populations.
- Pricing has declined over recent years due to widespread generic availability and price competition, with further slight declines expected.
- Future innovations, especially formulations emphasizing safety, may enable premium pricing.
- Market dynamics are heavily influenced by regulatory policies, patent expirations, and competitive entry by generics.
- Emerging markets offer growth opportunities, albeit at lower prices due to affordability constraints.
FAQs
1. What is the main driver behind naproxen sodium’s market expansion?
The primary driver is increased OTC availability, combined with aging populations requiring long-term pain management.
2. Will the price of FT Naproxen Sodium rise or fall in the next five years?
Prices are projected to decline slightly, due to market saturation and competition among generics.
3. How does naproxen sodium compare cost-wise to other NSAIDs?
It is generally cheaper on OTC formulations than prescription NSAIDs, but slightly more expensive per dose than ibuprofen, depending on brands and formulations.
4. Are there regulatory concerns that could impact the market?
Yes. Stricter safety warnings related to cardiovascular and gastrointestinal risks could influence consumer and prescribing behaviors.
5. What markets offer the most growth potential for naproxen sodium?
Emerging economies in Asia-Pacific, Latin America, and Africa hold the most potential due to expanding healthcare access and rising demand for OTC analgesics.
References
- Statista. (2023). Over-the-counter (OTC) analgesics market size worldwide.
- U.S. Food and Drug Administration. (2022). Monograph and labeling guidelines for NSAIDs.
- MarketWatch. (2023). NSAID market and forecast trends.
- IQVIA. (2022). Global pharmaceutical sales report.
- Deloitte. (2022). Pain management market analysis.
More… ↓
