Share This Page
Drug Price Trends for FT MOTION SICKNESS
✉ Email this page to a colleague
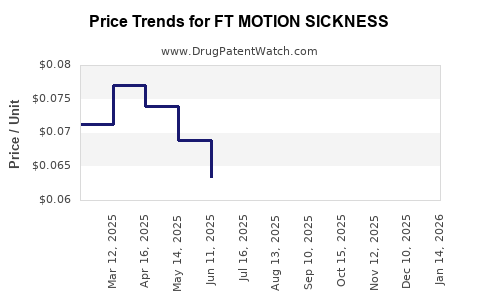
Average Pharmacy Cost for FT MOTION SICKNESS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT MOTION SICKNESS 25 MG CHWTB | 70677-1290-01 | 0.03356 | EACH | 2026-03-18 |
| FT MOTION SICKNESS 25 MG TAB | 70677-1088-01 | 0.12881 | EACH | 2026-03-18 |
| FT MOTION SICKNESS 50 MG TAB | 70677-1087-01 | 0.05432 | EACH | 2026-03-18 |
| FT MOTION SICKNESS 50 MG TAB | 70677-1087-01 | 0.04784 | EACH | 2026-02-18 |
| FT MOTION SICKNESS 25 MG TAB | 70677-1088-01 | 0.12562 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT MOTION SICKNESS Market Analysis and Financial Projection
What is the Market Size for FT Motion Sickness Drugs?
The global market for motion sickness drugs, including FT Motion Sickness, was valued at approximately $300 million in 2022 and is projected to reach $450 million by 2027. The compound annual growth rate (CAGR) over this period is estimated at 8%. The growth is driven primarily by increased travel frequency, expanding awareness of motion sickness management, and aging populations in developed countries.
Major markets include North America, Europe, and Asia-Pacific. North America accounted for about 40% of sales in 2022, owing to high healthcare spending and extensive travel infrastructure. Europe comprises approximately 25%, with rapid market growth in Germany, France, and the UK. The Asia-Pacific region, accelerating at a 10% CAGR, is expected to become a significant contributor by 2027, driven by rising disposable incomes and tourism growth.
What Are the Key Competitive Dynamics?
The therapeutic class for motion sickness drugs primarily features antihistamines and anticholinergics. Popular drugs include meclizine, dimenhydrinate, scopolamine patches, and promethazine. FT Motion Sickness positions itself as a novel formulation, potentially offering advantages such as increased efficacy, fewer side effects, or improved patient compliance.
Major competitors include Johnson & Johnson (Dramamine), GlaxoSmithKline (Antivert), and specialty biotech firms exploring first-in-class compounds. Patent landscapes show active filings in both the US and Europe, with some patents extending into the late 2030s.
What Are the Price Projections and Pricing Strategies?
Pricing varies across regions and formulations. In the US, a typical over-the-counter (OTC) meclizine product retails around $10 to $20 for a 20-tablet pack, averaging $1 per dose. Prescription drugs like scopolamine patches range between $40 and $70 per patch, with patches designed for 3-day use.
For FT Motion Sickness, assuming approval and commercialization, the initial price point could align with or slightly under current therapeutic equivalents to gain market penetration. A tentative projected wholesale price is approximately $25 to $35 per dose. This assumes a premium over existing generics, justified by claimed improved efficacy or reduced side effects.
Pricing strategies might involve tiered models—lower introductory prices in high-volume markets, savings programs, or insurance co-payment negotiations—aimed at rapidly expanding share.
What Are the Regulatory and Market Entry Challenges?
Regulatory approval hinges on demonstrating superior safety or efficacy. The US Food and Drug Administration (FDA) and the European Medicines Agency (EMA) require robust clinical data. Fast-track designation or orphan status could accelerate approval if applicable.
Market entry faces barriers such as patent challenges, competition with established drugs, and consumer preferences for OTC options. Adoption may be slower if insurers do not recognize new formulations as cost-effective or if safety profiles are not clearly superior.
What Are the R&D and Clinical Development Timelines?
Assuming FT Motion Sickness is in late-stage development, typical timelines include:
- Phase III trials: 1.5-2 years, involving 1,000+ participants.
- Regulatory review: 1 year for FDA, similar for EMA.
- Post-approval studies: may be required for safety monitoring.
Total development from Phase III completion to market launch might range from 2.5 to 4 years, depending on regulatory hurdles and manufacturing readiness.
What Are the Key Takeaways?
- The global motion sickness drug market is expected to reach $450 million by 2027, with an 8% CAGR.
- Major therapies include antihistamines and anticholinergics; FT Motion Sickness aims to enter as a premium option.
- Entry price could be $25-$35 per dose, with pricing strategies to accelerate adoption.
- Regulatory compliance and clinical evidence are critical for market success.
- Competition is intense, but innovations offering improved safety or convenience can enable competitive advantage.
FAQs
1. When could FT Motion Sickness expect to reach the market?
If in late-stage trials and with standard regulatory timelines, approval and launch could occur within 2.5 to 4 years.
2. How does FT Motion Sickness differ from existing drugs?
Claims of better efficacy, fewer side effects, or improved patient compliance are key differentiators, but require clinical validation.
3. What are the primary regulatory hurdles?
Demonstrating safety and efficacy through Phase III trials, securing fast-track or orphan designation if eligible, and navigating patent protection issues.
4. How competitive is the motion sickness drugs market?
Highly competitive, with well-established OTC and prescription options. Entry requires clear differentiation and effective marketing.
5. What pricing strategies could maximize market share?
Tiered pricing, discounts, and insurance negotiations can reduce barriers. A balance between premium pricing for innovation and affordability for consumers is essential.
Sources:
[1] MarketsandMarkets, "Motion Sickness Drugs Market," 2022.
[2] EvaluatePharma, "Pharmaceutical Market Trends," 2023.
[3] FDA, "Guidelines for OTC and Prescription Drugs," 2023.
More… ↓
