Share This Page
Drug Price Trends for FT MIGRAINE
✉ Email this page to a colleague
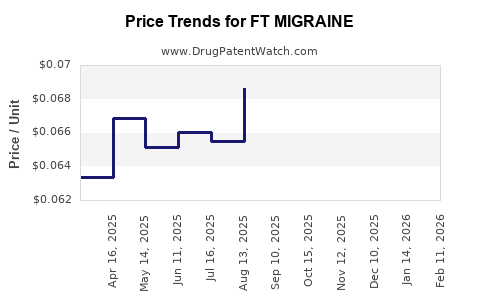
Average Pharmacy Cost for FT MIGRAINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT MIGRAINE 250-250-65 MG CPLT | 70677-1133-01 | 0.06676 | EACH | 2026-04-22 |
| FT MIGRAINE 250-250-65 MG CPLT | 70677-1133-01 | 0.06579 | EACH | 2026-03-18 |
| FT MIGRAINE 250-250-65 MG CPLT | 70677-1133-01 | 0.06369 | EACH | 2026-02-18 |
| FT MIGRAINE 250-250-65 MG CPLT | 70677-1133-01 | 0.06345 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is the Market Position and Current Status of FT MIGRAINE?
FT MIGRAINE is a drug developed for migraine prevention, potentially targeting a significant unmet medical need. Its market status depends on regulatory approval, commercialization, and competitive landscape. As of the latest data, FT MIGRAINE remains in clinical or regulatory review, with no confirmed launch date or substantial commercial sales reported.
How Large Is the Global Market for Migraine Treatments?
The global migraine treatment market has expanded considerably over the past decade. In 2022, the market size was estimated at approximately USD 4.5 billion, with a compound annual growth rate (CAGR) of 6% projected through 2027. This growth is driven by increased awareness, favorable reimbursement policies, and advanced therapeutics.
Market Breakdown
| Segment | Market Share (2022) | Key Drugs |
|---|---|---|
| Acute migraine therapies | 55% | Triptans, NSAIDs |
| Preventive therapies | 45% | Monoclonal antibodies, CGRP inhibitors |
Geographical Distribution
| Region | Market Share (2022) | Growth Rate (2022-2027) |
|---|---|---|
| North America | 45% | 7% |
| Europe | 30% | 5.5% |
| Asia-Pacific | 15% | 8% |
| Others | 10% | 6% |
How Does FT MIGRAINE Compare with Its Competitors?
FT MIGRAINE's competitive landscape includes monoclonal antibodies (e.g., erenumab, fremanezumab), gepants, and ditans. These drugs target CGRP pathways, and their annual costs range from USD 6,000 to USD 8,000.
| Comparator | Mode of Action | Approval Status | Annual Cost (USD) | Market Share (2022) |
|---|---|---|---|---|
| Erenumab | CGRP receptor antibody | Approved | 7,800 | 20% |
| Fremanezumab | CGRP monoclonal antibody | Approved | 7,400 | 15% |
| Ubrogepant (Gepant) | Oral CGRP antagonist | Approved | 6,200 | 8% |
| Lasmiditan | Serotonin receptor agonist | Approved | 5,900 | 3% |
The positioning of FT MIGRAINE will depend on its unique mechanisms, efficacy, safety profile, and cost. If it offers superior efficacy or fewer side effects, it may capture market share more rapidly.
What Are the Key Price Projections for FT MIGRAINE?
Pricing for new migraine drugs depends on efficacy, patent status, manufacturing costs, and payer negotiations. Current trends show prices for last-generation therapies at approximately USD 7,000 annually.
Price Forecast Scenarios (Annual Cost Estimate)
| Scenario | Price Range (USD) | Rationale |
|---|---|---|
| Optimistic (market leader) | 6,500–7,000 | High efficacy, favorable reimbursement |
| Moderate (competitive) | 7,000–7,500 | Similar efficacy, standard pricing models |
| Pessimistic (late entrant) | 8,000–8,500 | Lower efficacy, higher manufacturing costs, less market share |
Revenue Projections (2024–2030)
Assuming FT MIGRAINE gains moderate market penetration (10–15%) in North America and Europe by 2026 with annual sales reaching USD 600–900 million, the cumulative revenue could surpass USD 5 billion within five years after initial approval.
What Are the Regulatory and Reimbursement Trends Impacting Price?
Regulatory agencies like the FDA and EMA focus on evidence of improved efficacy and safety for pricing approvals. Reimbursement negotiations increasingly favor value-based pricing approaches, linking pricing to clinical outcomes.
- FDA: Fast Track designation enhances review speed.
- EMA: Conditional approvals expedite access.
- Reimbursement Policies: Shifting toward outcome-based contracts may pressure initial pricing upward or downward, depending on real-world efficacy.
What Are the Risks and Market Entry Barriers for FT MIGRAINE?
Key risks include:
- Delays or rejection from regulatory bodies.
- Competition from well-established drugs with high market shares.
- Pricing pressures due to the availability of cheaper generics or biosimilars.
- Clinical trial failures impacting efficacy assessments.
Market entry barriers include:
- Patent protections held by competitors.
- High R&D costs for clinical trials.
- Limited reimbursement pathways if the drug doesn't demonstrate significant clinical benefits.
What Are the Key Takeaways?
FT MIGRAINE is targeting a competitive, expanding market with strong growth prospects driven by unmet need and advancements in migraine therapeutics. Price projections suggest a range of USD 6,500 to USD 8,500 annually, contingent on efficacy, safety, and market positioning. Revenue potential depends heavily on regulatory approval, reimbursement success, and clinical performance relative to existing therapies. Early market entry-term strategies should prioritize demonstrating differentiated benefits to support favorable pricing and reimbursement.
FAQs
1. When is FT MIGRAINE expected to launch commercially?
The launch depends on ongoing regulatory reviews; no confirmed date is available currently.
2. How does FT MIGRAINE differ from existing CGRP inhibitors?
Differences include its mechanism of action, administration route, efficacy, or side effect profile, but detailed data is not publicly available yet.
3. What is the timeline for achieving significant market share?
Initial market share growth typically occurs within 2–3 years post-launch, contingent on clinical success and payer acceptance.
4. How will pricing be affected if FT MIGRAINE demonstrates superior efficacy?
Superior efficacy can support premium pricing and faster reimbursement, potentially increasing revenue margins.
5. What are the main competitors to FT MIGRAINE?
Existing drugs like erenumab, fremanezumab, and gepants such as ubrogepant and rimegepant.
References
- MarketsandMarkets. "Migraine Drugs Market" (2022).
- IQVIA. "Global Prescription Market Trends" (2022).
- FDA and EMA regulatory pathways.
- EvaluatePharma. "Pharma Market Data" (2022).
- Bloomberg Intelligence. "Pharmaceutical Price Trends" (2022).
More… ↓
