Share This Page
Drug Price Trends for FT MAGNESIUM CITRATE SOLUTION
✉ Email this page to a colleague
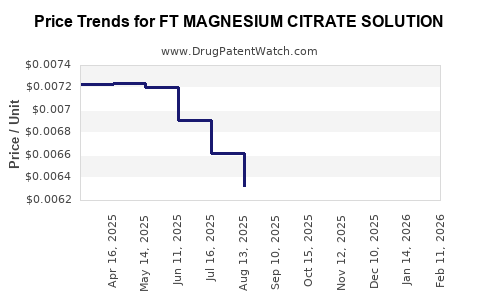
Average Pharmacy Cost for FT MAGNESIUM CITRATE SOLUTION
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT MAGNESIUM CITRATE SOLUTION | 70677-1111-01 | 0.00617 | ML | 2026-02-18 |
| FT MAGNESIUM CITRATE SOLUTION | 70677-1112-01 | 0.00617 | ML | 2026-02-18 |
| FT MAGNESIUM CITRATE SOLUTION | 70677-1111-01 | 0.00612 | ML | 2026-01-21 |
| FT MAGNESIUM CITRATE SOLUTION | 70677-1112-01 | 0.00612 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT Magnesium Citrate Solution
Overview
FT Magnesium Citrate Solution is a laxative indicated primarily for bowel preparation before diagnostic procedures like colonoscopies. It also has uses in managing constipation. The product's market dynamics are driven by the demand for gastrointestinal diagnostics, aging populations, and increasing prevalence of digestive disorders. The global gastrointestinal drugs market is expected to grow at a compound annual growth rate (CAGR) of approximately 6.2% from 2022 to 2028, reaching an estimated value of $11.8 billion by 2028 [1].
Market Size and Growth Drivers
| Parameter | Value / Estimate |
|---|---|
| 2022 Global GI drug market size | $7 billion |
| CAGR (2022-2028) | 6.2% |
| Key growth drivers | Aging populations, rising digestive disorders, expanding minimally invasive diagnostic procedures |
Competitive Landscape
Major competitors include Bayer, Salix Pharmaceuticals, and generic manufacturers. Bayer’s Miralax (polyethylene glycol 3350), branded for bowel prep and constipation, commands a significant market share. The ubiquity of generic options impacts pricing and margins. Regulatory approvals vary by region:
- FDA approved in the US
- EMA approval in Europe
- Multiple approvals in Asia and developing markets
Pricing Landscape
The price for a 240 mL bottle of FT Magnesium Citrate Solution ranges between $8 and $15 in the U.S., with variations depending on supplier, packaging, and bulk discounts. Generic magnesium citrate solutions are priced competitively, often under $10 per bottle.
Price Projection Factors
Factors influencing future pricing include:
- Regulatory Changes: Faster approvals or patent expiries could increase competition, pushing prices downward.
- Manufacturing Costs: Stabilization or reduction in raw material costs, including magnesium salts and excipients.
- Market Penetration: Entry into emerging markets with lower price sensitivity.
- Reimbursement Policies: Insurance coverage affects out-of-pocket costs, influencing consumer demand.
- Product Differentiation: Enhanced formulations or improved tolerability could command premium pricing.
Projected Price Range (2023-2028)
| Year | Estimated Price Range (per 240 mL) | Notes |
|---|---|---|
| 2023 | $8.00 – $12.00 | Current market levels |
| 2024 | $8.00 – $11.50 | Slight pressure from generics |
| 2025 | $8.00 – $11.00 | Increasing market penetration |
| 2026 | $7.50 – $10.50 | Patent expiries influence prices |
| 2027 | $7.50 – $10.00 | Growing presence in emerging markets |
| 2028 | $7.00 – $9.50 | Competitive landscape stabilizes |
Market Entry and Expansion Opportunities
- Emerging Markets: Countries such as India, China, and Southeast Asian nations are experiencing increased adoption of gastrointestinal diagnostics, opening avenues for lower-cost formulations.
- Formulation Innovation: Developing ready-to-drink or flavored options could enhance patient adherence and justify premium pricing.
- Strategic Partnerships: Collaborations with healthcare providers or contract manufacturing organizations can reduce costs and accelerate market penetration.
Regulatory and Patent Considerations
- Patent protection for formulations or delivery mechanisms may delay generic entry until around 2026.
- Regulatory pathways for OTC status vary by jurisdiction; success in obtaining over-the-counter approval can expand market reach and influence price structures.
Conclusion
FT Magnesium Citrate Solution exists in a mature market with moderate growth prospects. Price stability is influenced by generic competition, manufacturing costs, and regional regulatory environments. Price declines are expected to be limited due to product standardization, but novel formulations or market expansion into emerging regions can offset downward pressure.
Key Takeaways
- The global GI drug market is expanding at roughly 6.2%, driven by demographic shifts.
- Current retail prices range from $8 to $15 per 240 mL bottle in the US.
- Price projections suggest a gradual decline to around $7 to $9.50 by 2028, barring significant regulatory or market developments.
- Competition from generics and formulary inclusion impacts pricing strategies.
- Opportunities exist in emerging markets and through formulation innovation.
FAQs
-
What are the main competitors to FT Magnesium Citrate Solution?
Bayer’s Miralax, generic magnesium citrate solutions, and other bowel preparatory products. -
How does patent expiration influence pricing?
Patent expiry around 2026 may lead to increased generic competition, exerting downward pressure on prices. -
What regulatory hurdles exist for market expansion?
Gaining approval for OTC status or regulatory harmonization can extend product access; compliance with regional pharmacopoeias also impacts market entry. -
How can manufacturers differentiate their magnesium citrate products?
Through improved formulations, flavoring, packaging, and patient comfort features. -
What is the impact of reimbursement policies on pricing?
Reimbursement coverage influences consumer out-of-pocket costs, affecting demand and pricing strategies.
Sources
[1] Research and Markets, "Gastrointestinal Drugs Market Analysis," 2022.
More… ↓
