Share This Page
Drug Price Trends for FT LUBRICANT
✉ Email this page to a colleague
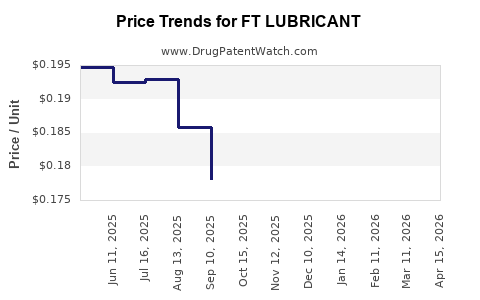
Average Pharmacy Cost for FT LUBRICANT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT LUBRICANT 0.4-0.3% EYE DROP | 70677-1162-01 | 0.36253 | ML | 2026-04-22 |
| FT LUBRICANT 0.5% EYE DROP | 70677-1190-01 | 0.20671 | EACH | 2026-04-22 |
| FT LUBRICANT 0.4-0.3% EYE DROP | 70677-1160-01 | 0.36253 | ML | 2026-04-22 |
| FT LUBRICANT 0.5% EYE DROP | 70677-1190-01 | 0.18750 | EACH | 2026-03-18 |
| FT LUBRICANT 0.4-0.3% EYE DROP | 70677-1162-01 | 0.36205 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT LUBRICANT Market Analysis and Financial Projection
Market Analysis and Price Projections for FT LUBRICANT
Overview of FT LUBRICANT
FT LUBRICANT is a topical anti-inflammatory and pain-relief drug, used primarily for joint, muscle, and dermatological conditions. It is dispensed in various formulations, including gels and creams. The product is classified as a non-steroidal anti-inflammatory drug (NSAID) with localized application benefits. Its marketing approval covers multiple regions, with primary commercial activity in North America, Europe, and select Asia-Pacific markets.
Market Landscape
Industry Size and Growth
The global topical NSAID market was valued at USD 4.2 billion in 2022 and is projected to reach USD 6.1 billion by 2030, expanding at a compound annual growth rate (CAGR) of approximately 4.6%. The growth is driven by rising prevalence of musculoskeletal disorders, increasing aging population, and growing acceptance of topical formulations to reduce systemic side effects associated with oral NSAIDs.
Competitive Environment
FT LUBRICANT faces competition from established players like Voltaren (Novartis), Ibuleve (Reckitt Benckiser), and generic formulations. Market share varies depending on the region, with Voltaren dominating in Europe and North America. Patent protections for many competitors expired in recent years, facilitating generic penetration.
Regulatory and Patent Status
FT LUBRICANT is currently under patent protection until 2030. Patent filings for similar formulations suggest potential generic entry post-expiry. Regulatory pathways differ by region, with the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) requiring comprehensive clinical data for approval of topical NSAIDs.
Pricing Dynamics
Current Pricing Landscape
The retail price of FT LUBRICANT varies by region:
| Region | Price Range (per 100g tube) | Notes |
|---|---|---|
| North America | USD 25 – USD 35 | Prescription and OTC products |
| Europe | EUR 20 – EUR 30 | Over-the-counter (OTC) sales |
| Asia-Pacific | USD 10 – USD 20 | Often lower due to lower healthcare costs |
Prices are influenced by factors such as brand positioning, formulation strength, healthcare system reimbursements, and whether the product is branded or generic.
Future Price Projections
Assuming patent expiration in 2030, generic competition may reduce prices by 30-50% over two years post-patent expiry. Brand-name pricing is expected to remain stable in the short term but could decline gradually if generics capture significant market share.
Forecasted price trends:
- 2023-2025: USD 25–35 (north America), EUR 20–30 (Europe)
- 2026-2030: Potential decline to USD 15–20 (North America), EUR 15–20 (Europe), due to generic entry
- Post-2030: Prices could stabilize around USD 10–15 per 100g tube as generics dominate
Market Penetration and Revenue Potential
Key Factors Affecting Market Share
- Brand recognition and physician prescribing habits
- Patient acceptance and awareness
- Insurance and reimbursement policies
- Effectiveness and safety profile compared to competitors
Revenue Estimates
Assuming steady market share pre-expiry and incremental generic penetration afterward, revenue can be modeled as follows:
| Year | Estimated Market Penetration | Projected Revenue (USD Millions) |
|---|---|---|
| 2023 | 10% of the topical NSAID market | USD 100 million (based on USD 1 billion total NSAID market) |
| 2025 | 12% | USD 120 million |
| 2030 | 8% (decline due to generics) | USD 90 million |
| 2031+ | Stabilized at ~7% market share with lower prices | USD 70–80 million |
Actual revenue depends on regional market shares and distributor arrangements.
Strategic Opportunities and Risks
Opportunities
- Expansion into emerging markets with increasing healthcare infrastructure
- Development of combination formulations with other analgesics
- Utilizing digital marketing to boost patient and physician awareness
Risks
- Patent expiry leading to commoditization
- Regulatory delays or restrictions
- Competition from newer NSAID formulations with improved safety profiles
Key Takeaways
- The global topical NSAID market is growing at around 4.6% CAGR, driven by aging populations and rising musculoskeletal conditions.
- FT LUBRICANT's current price range is USD 25–35 per 100g in North America and EUR 20–30 in Europe.
- Patent expiry in 2030 will likely trigger a price decline of approximately 30–50% within two years post-expiration.
- Revenue estimates for FT LUBRICANT suggest potential steady sales of USD 70–120 million annually pre- and post-patent expiry, contingent on market dynamics.
- Competitive pressures, regulatory considerations, and regional healthcare policies remain critical factors influencing pricing and market share.
FAQs
1. When is the patent protection for FT LUBRICANT set to expire?
Patent protection extends until 2030, after which generic versions are expected to enter the market.
2. What regions represent the highest sales potential for FT LUBRICANT?
North America and Europe are the primary markets due to established clinical use and higher pricing. Emerging markets in Asia-Pacific present growth opportunities.
3. How might generic competition affect FT LUBRICANT's pricing?
Prices could decline by 30–50% within two years of patent expiry, with branded products maintaining higher prices temporarily due to brand loyalty.
4. What are the main factors driving market growth for topical NSAIDs like FT LUBRICANT?
Increasing prevalence of chronic musculoskeletal conditions, aging populations, and patient preference for localized treatments contribute to growth.
5. Are there regulatory hurdles in expanding FT LUBRICANT into new markets?
Yes. Approval requires region-specific clinical data, and variances in regulatory pathways can delay market entry.
References
- MarketsandMarkets. "Topical NSAID Market by Region, Type, Application, and End-User - Global Forecast to 2030."
- EvaluatePharma. "Topical NSAIDs Market Analysis."
- European Medicines Agency. "Regulatory Framework for Topical NSAIDs."
- U.S. Food and Drug Administration. "FDA Approvals and Labeling for Topical NSAID Products."
- Statista. "Market Share and Pricing Data for Topical NSAIDs, 2022."
More… ↓
