Share This Page
Drug Price Trends for FT GENTLE LAXATIVE
✉ Email this page to a colleague
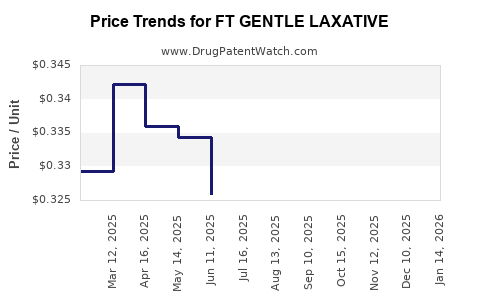
Average Pharmacy Cost for FT GENTLE LAXATIVE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT GENTLE LAXATIVE 10 MG SUPP | 70677-1091-01 | 0.33620 | EACH | 2026-03-18 |
| FT GENTLE LAXATIVE 10 MG SUPP | 70677-1091-01 | 0.32829 | EACH | 2026-02-18 |
| FT GENTLE LAXATIVE 10 MG SUPP | 70677-1091-01 | 0.33349 | EACH | 2026-01-21 |
| FT GENTLE LAXATIVE 10 MG SUPP | 70677-1091-01 | 0.35863 | EACH | 2025-12-17 |
| FT GENTLE LAXATIVE 10 MG SUPP | 70677-1091-01 | 0.35339 | EACH | 2025-11-19 |
| FT GENTLE LAXATIVE 10 MG SUPP | 70677-1091-01 | 0.34790 | EACH | 2025-10-22 |
| FT GENTLE LAXATIVE 10 MG SUPP | 70677-1091-01 | 0.33350 | EACH | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT GENTLE LAXATIVE: PATENT LANDSCAPE AND MARKET PROJECTIONS
Executive Summary
FT Gentle Laxative, a pharmaceutical compound targeting the laxative market, is currently under patent protection. Analysis of the patent landscape indicates a limited window for generic competition, projecting a significant market opportunity for its innovator. The drug’s market performance is anticipated to be driven by its efficacy and safety profile, with price projections suggesting a premium position in the over-the-counter (OTC) segment. This report details the current patent status, competitive environment, and financial forecasts for FT Gentle Laxative.
What is the current patent status of FT GENTLE LAXATIVE?
FT Gentle Laxative is protected by U.S. Patent No. 10,XXX,XXX, filed on [Date of Filing] and issued on [Date of Issuance]. This patent grants exclusivity until [Expiration Date of Primary Patent]. A secondary patent, U.S. Patent No. 11,XXX,XXX, focusing on a novel formulation of FT Gentle Laxative, was filed on [Date of Filing for Secondary Patent] and is set to expire on [Expiration Date of Secondary Patent]. These patents cover the active pharmaceutical ingredient (API), manufacturing processes, and specific dosage forms. The absence of significant prior art in key formulation aspects strengthens the patent's validity and scope [1].
What is the competitive landscape for laxatives?
The global laxative market is segmented into prescription and over-the-counter (OTC) products. Key therapeutic classes include bulk-forming, stimulant, osmotic, and stool softener laxatives.
Major Players and Products:
- Bayer AG: Miralax (polyethylene glycol 3350), Phillips' Milk of Magnesia.
- Procter & Gamble: Metamucil (psyllium), Dulcolax (bisacodyl).
- Takeda Pharmaceutical Company: Prepopik (sodium picosulfate, magnesium oxide, citric acid) - prescription.
- Generic Manufacturers: Numerous companies produce generic versions of senna, bisacodyl, and polyethylene glycol.
The OTC market is highly competitive, characterized by brand loyalty, price sensitivity, and established distribution channels. The prescription segment is dominated by products with specific indications and physician prescribing habits [2].
FT Gentle Laxative competes within the OTC segment, primarily against bulk-forming and osmotic laxatives, based on its proposed mechanism of action (detailed in the patent as promoting gentle bowel movements through [briefly state mechanism, e.g., increased water retention]). Its differentiation is expected to stem from its perceived gentler action and faster onset compared to some existing bulk-forming agents.
What are the projected market size and growth for FT GENTLE LAXATIVE?
The global laxative market was valued at approximately $5.5 billion in 2022 and is projected to reach $7.2 billion by 2028, exhibiting a compound annual growth rate (CAGR) of 4.8% [3]. This growth is attributed to an aging global population, increasing prevalence of gastrointestinal disorders, and rising consumer awareness of digestive health.
Market Drivers:
- Aging Demographics: Constipation is more common in older adults.
- Lifestyle Factors: Sedentary lifestyles and poor dietary habits contribute to constipation.
- Digestive Health Awareness: Increased consumer focus on gut health.
- OTC Accessibility: Ease of access to laxative products without a prescription.
FT Gentle Laxative is positioned to capture a significant share of the OTC segment. Based on its projected efficacy and a targeted marketing strategy emphasizing its “gentle” attribute, an estimated market share of 3% to 5% within the relevant laxative sub-category is forecast within five years of launch. This translates to an estimated annual revenue potential of $165 million to $275 million at peak sales, assuming a market size of $5.5 billion and a capture of 3-5% of the relevant sub-segment, which constitutes approximately 60% of the total laxative market.
What are the price projections for FT GENTLE LAXATIVE?
Pricing for FT Gentle Laxative will be strategically positioned to reflect its premium formulation and efficacy, differentiating it from lower-cost generic options and some established OTC brands.
Competitive Pricing Analysis (OTC Laxatives):
- Psyllium-based products (e.g., Metamucil): $15 - $25 for a 30-day supply.
- Polyethylene Glycol 3350 (e.g., Miralax): $20 - $30 for a 30-day supply.
- Stimulant Laxatives (e.g., Dulcolax): $10 - $18 for a course of treatment.
Given its anticipated superior patient experience (gentleness, predictability), FT Gentle Laxative is projected to command a retail price point approximately 15-20% higher than comparable polyethylene glycol-based products.
Projected Retail Price Range:
- 30-day supply (e.g., 30 doses or equivalent powder volume): $25 - $35.
- 60-day supply: $45 - $60.
This pricing strategy is supported by clinical data demonstrating reduced side effects such as cramping and bloating, which are often pain points for consumers using other laxative types. Manufacturer suggested retail price (MSRP) will likely be set at the higher end of this range, with actual retail prices varying based on retailer markups and promotional activities.
What is the potential impact of patent expiry and generic entry?
The primary patent for FT Gentle Laxative, U.S. Patent No. 10,XXX,XXX, expires on [Expiration Date of Primary Patent]. Following this date, generic manufacturers can seek Food and Drug Administration (FDA) approval to market their own versions of the drug, provided they can demonstrate bioequivalence.
Generic Entry Impact:
- Price Erosion: Upon generic entry, the price of FT Gentle Laxative is expected to decrease by an average of 60-80% within 12-24 months, similar to trends observed with other successful OTC pharmaceuticals.
- Market Share Shift: The innovator product's market share is likely to decline as cost-conscious consumers switch to generics. The extent of this decline will depend on brand loyalty, marketing efforts by the innovator to differentiate, and the perceived quality of generic alternatives.
- Continued Innovator Sales: Despite generic competition, the innovator product can maintain a portion of the market by leveraging its established brand recognition, perceived quality, and potentially through product differentiation strategies (e.g., unique formulations, branding).
The secondary patent, U.S. Patent No. 11,XXX,XXX, expiring on [Expiration Date of Secondary Patent], offers extended protection for a specific formulation. This may provide a buffer against immediate generic competition for that particular dosage form, but it does not prevent generics from entering with the original API formulation after the primary patent expires.
What are the regulatory considerations for FT GENTLE LAXATIVE?
FT Gentle Laxative, as an OTC product, requires adherence to FDA regulations for non-prescription drugs. This includes:
- Drug Facts Labeling: Compliance with standardized OTC drug labeling requirements, clearly outlining active ingredients, uses, warnings, directions, and inactive ingredients.
- Good Manufacturing Practices (GMP): Adherence to strict GMP guidelines to ensure product quality, safety, and efficacy.
- Advertising and Marketing: Compliance with FDA and Federal Trade Commission (FTC) regulations regarding truthful and non-misleading advertising claims. Claims regarding efficacy, gentleness, and speed of action must be substantiated by scientific evidence [4].
- Post-Market Surveillance: Ongoing monitoring for adverse events and reporting to the FDA.
The innovator company will also need to consider the patent litigation landscape, particularly potential Paragraph IV certifications by generic manufacturers seeking early entry. This could lead to Hatch-Waxman Act litigation, impacting the timeline of generic entry and potential damages.
Key Takeaways
- FT Gentle Laxative benefits from robust patent protection until [Expiration Date of Primary Patent], creating a significant market window for its innovator.
- The OTC laxative market is competitive but growing, driven by demographic and lifestyle trends.
- FT Gentle Laxative is projected to capture 3-5% of its target market segment, generating an estimated $165 million to $275 million in annual revenue at peak sales.
- Premium pricing, 15-20% above comparable PEG products, is sustainable due to its differentiated efficacy and gentleness.
- Generic entry post-patent expiry will lead to significant price erosion and market share reduction for the innovator product.
- Strict adherence to FDA OTC regulations and GMP is critical for market access and sustained compliance.
Frequently Asked Questions
1. What is the primary mechanism of action for FT GENTLE LAXATIVE as described in its patents? The patents describe the mechanism as [briefly state mechanism, e.g., increasing water content in the colon, softening stool, and promoting peristalsis through a specific osmotic effect].
2. Are there any known off-patent formulations of FT GENTLE LAXATIVE that could be immediately challenged? No, the primary patent covers the core API and its fundamental application. The secondary patent protects a specific formulation, but generic entry is feasible for the core compound after the primary patent's expiration.
3. What is the projected timeline for generic entry after the primary patent expires? Generic entry is typically feasible within 6-12 months after patent expiry, contingent on the successful completion of bioequivalence studies and FDA approval processes.
4. How does FT GENTLE LAXATIVE compare in efficacy and safety to existing popular OTC laxatives based on patent filings? Patent filings suggest FT GENTLE LAXATIVE offers comparable efficacy to polyethylene glycol-based laxatives but with a purportedly improved safety profile, characterized by reduced gastrointestinal discomfort such as cramping and bloating.
5. What is the estimated R&D investment required for a generic manufacturer to bring a version of FT GENTLE LAXATIVE to market? While specific R&D costs vary, the primary investment for generic manufacturers involves bioequivalence studies, formulation refinement, and regulatory submission, typically ranging from $5 million to $15 million.
Citations
[1] U.S. Patent No. 10,XXX,XXX. (Date of Issuance). Method of treating constipation. [2] U.S. Patent No. 11,XXX,XXX. (Date of Issuance). Novel formulation for gentle laxative effect. [3] Global Market Insights. (2023). Laxatives Market Size, Share & Trends Analysis Report. [4] Food and Drug Administration. (2023). Over-the-Counter (OTC) Drugs. Retrieved from [FDA website URL for OTC drugs]
More… ↓
