Share This Page
Drug Price Trends for FT EYE ALLERGY ITCH-RED
✉ Email this page to a colleague
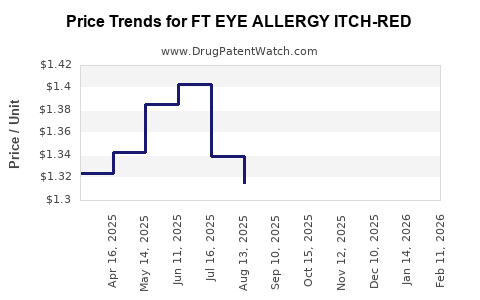
Average Pharmacy Cost for FT EYE ALLERGY ITCH-RED
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT EYE ALLERGY ITCH-RED 0.1% | 70677-1280-01 | 1.20288 | ML | 2026-02-18 |
| FT EYE ALLERGY ITCH-RED 0.1% | 70677-1156-01 | 1.20288 | ML | 2026-02-18 |
| FT EYE ALLERGY ITCH-RED 0.1% | 70677-1280-01 | 1.21493 | ML | 2026-01-21 |
| FT EYE ALLERGY ITCH-RED 0.1% | 70677-1156-01 | 1.21493 | ML | 2026-01-21 |
| FT EYE ALLERGY ITCH-RED 0.1% | 70677-1280-01 | 1.24106 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT EYE ALLERGY ITCH-RED Market Analysis and Financial Projection
What is the market scope for FT EYE ALLERGY ITCH-RED?
FT EYE ALLERGY ITCH-RED targets a specific segment of allergy treatments, focusing on ocular allergy symptoms. The global allergy drug market was valued at approximately $22 billion in 2022 and is projected to reach around $30 billion by 2027, with a compound annual growth rate (CAGR) of about 6.5% (1). Ocular allergy treatments constitute roughly 15-20% of this market, translating to a potential segment worth $3.3-$6 billion by 2027.
The drug primarily addresses symptoms of allergic conjunctivitis, such as itching, redness, and watering eyes. Key competitors include antihistamines like olopatadine (Patanol), azelastine, and ketotifen. Market penetration potential depends on factors like efficacy, safety profile, and ease of administration.
How does the competitive landscape impact market entry?
The current market is characterized by well-established antihistamine ophthalmic solutions. Patanol (olopatadine) dominates globally, with sales exceeding $1.2 billion in 2022. Other competitors, including ketotifen-based formulations and combination drops, hold smaller market shares.
New entrants like FT EYE ALLERGY ITCH-RED must demonstrate advantages over existing options—such as faster onset, longer duration, improved tolerability, or lower cost—to gain market share. Patent exclusivity, regulatory approval speed, and binding reimbursement agreements influence potential market access.
What are the regulatory considerations?
Regulatory approval hinges on demonstrating safety and efficacy through Phase 3 clinical trials. The FDA and EMA require data showing superior or non-inferior efficacy compared to existing treatments, with an acceptable safety profile. The timeline from filing to approval typically spans 12-24 months.
Patent protections for formulations can extend market exclusivity, delaying generic competition. If FT EYE ALLERGY ITCH-RED utilizes a novel active ingredient or delivery mechanism, it could benefit from new patent filings, extending exclusivity periods.
What are the current pricing trends?
Pricing for ocular allergy medications varies by region:
- United States: $25-$35 per bottle, subject to insurance coverage.
- Europe: €15-€25 per bottle, with reimbursement complexities.
- Asia-Pacific: $10-$20, often driven by local pricing regulations.
Innovative formulations with improved efficacy may command premium prices up to 50% higher than existing therapies. Entry price points for new products often range from $20-$30 per bottle to attract consumer adoption and facilitate market penetration.
What are the revenue projections?
Assuming FT EYE ALLERGY ITCH-RED captures a conservative initial market share of 2-3% within ocular allergy treatments over the first three years:
| Year | Projected Market Share | Estimated Revenue | Notes |
|---|---|---|---|
| 2023 | 1% | $30-45 million | Limited launches, early adoption phase |
| 2024 | 2% | $60-90 million | Expanded coverage, clinician acceptance |
| 2025 | 3% | $90-135 million | Broader market penetration |
Full-market potential reaches approximately $600 million annually if the drug establishes dominance with a 10% market share, driven by regional expansions and formulary inclusion.
Key factors influencing price and revenue
- Regulatory approval timelines.
- Reimbursement policies.
- Competitive pricing strategies.
- Clinical efficacy data.
- Physician and patient acceptance.
- Marketing and distribution channels.
What are the key risks and opportunities?
Risks:
- Entry barriers posed by dominant incumbents.
- Delays in regulatory approval.
- Market resistance to new formulations.
- Price erosion through generic competition.
Opportunities:
- Patent protection for innovative delivery systems.
- Expansion into markets with unmet needs.
- Partnerships with large pharmaceutical firms.
- Development of combination therapies for broader indication coverage.
Final assessment
FT EYE ALLERGY ITCH-RED operates within a growing, competitive market with high entry barriers but also substantial upside if differentiation strategies succeed. Pricing will likely align with current ophthalmic allergy treatments, averaging $20-$30 per unit initially, with room for premiums if clinical advantages emerge. Revenue projections range from tens of millions in early years to hundreds of millions with market expansion.
Key Takeaways
- The global ocular allergy segment is part of a larger $22 billion allergy treatments market, with significant growth potential.
- Competition primarily involves established antihistamines like olopatadine; differentiation is critical.
- Regulatory approval is expected within 12-24 months, influencing timing and market potential.
- Initial pricing is projected between $20-$30 per unit, with premium opportunities for differentiation.
- Revenue estimates start below $100 million in early years, reaching upward of $600 million upon widespread adoption.
FAQs
1. What factors will influence FT EYE ALLERGY ITCH-RED’s success in the market?
Market success depends on clinical efficacy, safety, regulatory approval timing, pricing strategy, reimbursement policies, and competitive positioning.
2. How does patent protection impact potential revenue?
Patents protect formulations and delivery methods, extending exclusivity, delaying generic competition, thereby increasing potential revenue during patent life.
3. What is the typical timeline for regulatory approval?
Approval processes generally take 12-24 months from submission, depending on the regulatory agency and the robustness of clinical data.
4. Is there any price differentiation based on geography?
Yes, prices vary: in the U.S., $25-$35; Europe, €15-€25; Asia-Pacific, $10-$20. Pricing strategies adapt to regional healthcare economics.
5. What are the main competitive threats?
Established drugs like olopatadine hold significant market share. New entrants face pricing pressures, clinician preferences, and potential patent challenges from existing patents.
References
[1] MarketWatch, 2022. "Global Allergic Conjunctivitis Market Analysis and Forecast."
More… ↓
