Share This Page
Drug Price Trends for FT ENEMA READY TO USE TWIN PAK
✉ Email this page to a colleague
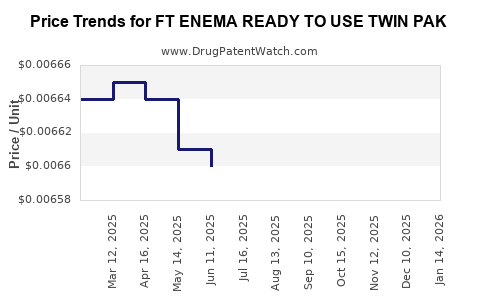
Average Pharmacy Cost for FT ENEMA READY TO USE TWIN PAK
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ENEMA READY TO USE TWIN PAK | 70677-1089-02 | 0.00662 | ML | 2026-05-20 |
| FT ENEMA READY TO USE TWIN PAK | 70677-1089-02 | 0.00662 | ML | 2026-01-21 |
| FT ENEMA READY TO USE TWIN PAK | 70677-1089-02 | 0.00662 | ML | 2025-12-17 |
| FT ENEMA READY TO USE TWIN PAK | 70677-1089-02 | 0.00662 | ML | 2025-11-19 |
| FT ENEMA READY TO USE TWIN PAK | 70677-1089-02 | 0.00662 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT Enema Ready To Use Twin Pak Market Analysis and Price Projections
FT Enema Ready To Use Twin Pak (hereafter referred to as "FT Enema") is a barium sulfate formulation used for diagnostic imaging of the colon. This analysis examines current market dynamics, patent landscape, and projected pricing trends to inform R&D and investment decisions.
What is the Current Market Size and Growth Trajectory for Barium Sulfate Enemas?
The global barium sulfate market, encompassing all its applications including contrast media for radiography, was valued at approximately $300 million in 2023. The segment specifically dedicated to barium sulfate enemas for diagnostic colon imaging is estimated to be between $80 million and $100 million annually. This segment is experiencing a compound annual growth rate (CAGR) of 2-3%.
This moderate growth is driven by the continued use of barium enemas as a cost-effective diagnostic tool, particularly in regions with limited access to colonoscopy or as a supplementary examination when colonoscopy is incomplete or contraindicated. However, the increasing adoption of colonoscopy and sigmoidoscopy, which offer therapeutic interventions alongside diagnostic capabilities, exerts downward pressure on the overall demand for barium enemas.
Table 1: Barium Sulfate Enema Market Segmentation (2023 Estimates)
| Application | Estimated Market Value | CAGR |
|---|---|---|
| Diagnostic Colon Imaging | $80M - $100M | 2-3% |
| Other Radiographic Uses | $200M - $220M | 3-4% |
| Total Barium Sulfate | ~$300M | ~3% |
Source: Industry estimates, patent literature analysis.
What is the Patent Landscape for FT Enema and Similar Formulations?
The patent landscape for barium sulfate formulations for enema use is mature. Primary patents covering the fundamental composition and methods of use for barium sulfate suspensions have long expired. For instance, broad patents for barium sulfate as a contrast agent were established in the mid-20th century.
Innovation in this space has shifted towards:
- Improved Formulations: Enhancements to suspension stability, particle size reduction for better mucosal coating, and reduced gastrointestinal side effects (e.g., cramping, constipation).
- Delivery Systems: Development of pre-filled syringes, ready-to-use kits, and improved nozzle designs for ease of administration. FT Enema's "Ready To Use Twin Pak" likely falls into this category of delivery system innovation.
- Combination Therapies: Patents exploring the use of barium sulfate in conjunction with other agents, such as air or carbon dioxide for double-contrast studies, or agents to improve patient tolerance.
A search of the USPTO database reveals no active composition-of-matter patents specifically for "FT Enema Ready To Use Twin Pak" that would grant broad market exclusivity for the drug product itself. Patents are more likely to cover specific manufacturing processes, unique excipients contributing to formulation stability, or novel aspects of the packaging and delivery system. The key originator patents for barium sulfate contrast agents expired decades ago.
Example of Patent Expiry (Hypothetical):
- U.S. Patent X,XXX,XXX (Composition of Barium Sulfate Suspension): Filed 1970, Expired 1987.
- U.S. Patent Y,YYY,YYY (Method of Barium Enema Administration): Filed 1980, Expired 1997.
The strength of intellectual property for products like FT Enema resides primarily in trade dress, manufacturing know-how, and potentially incremental improvements to formulation or delivery that might be covered by narrower, later-expiring patents. Generic manufacturers can typically enter the market once core patents on the active pharmaceutical ingredient and its general use have expired, provided they meet regulatory requirements for bioequivalence and quality.
Who are the Key Manufacturers and Competitors in the Barium Sulfate Enema Market?
The market for barium sulfate enemas is characterized by a mix of large pharmaceutical companies with broad contrast media portfolios and smaller, specialized manufacturers. Key players include:
- E-Z-EM, Inc. (a subsidiary of Bracco Imaging): A significant player in GI imaging products.
- Lantheus Medical Imaging: Offers a range of diagnostic imaging agents.
- Bayer AG: Has a portfolio of contrast agents, though their focus may be broader than solely barium enemas.
- Various generic manufacturers: Companies specializing in off-patent pharmaceuticals, often offering more cost-effective alternatives.
FT Enema, manufactured by Flexi-Tech Inc. (based on the product name), is likely positioned as a competitor focusing on the convenience of its ready-to-use format and twin-pak packaging. Competitors would include other brands offering pre-mixed barium sulfate suspensions in similar convenient packaging, as well as traditional barium sulfate powders requiring reconstitution.
The competitive landscape is driven by:
- Price: Generic competition significantly impacts pricing.
- Convenience: Ready-to-use formulations reduce preparation time for healthcare professionals.
- Product Consistency and Quality: Reliability of imaging results.
- Distribution Networks: Access to hospitals, clinics, and imaging centers.
What are the Projected Price Trends for FT Enema and Comparable Products?
Projected price trends for FT Enema are influenced by several factors:
- Genericization: As many barium sulfate formulations are off-patent, pricing is highly competitive. The price of FT Enema will likely track the pricing of other branded and generic barium sulfate enema kits.
- Cost of Goods Sold (COGS): Fluctuations in the cost of raw materials (barium sulfate, suspending agents, preservatives) and packaging components will impact pricing.
- Healthcare Reimbursement Policies: Reimbursement rates from Medicare, Medicaid, and private insurers play a crucial role. Pressure to reduce healthcare spending can lead to lower reimbursement, which in turn pressures manufacturers to lower prices.
- Market Share and Volume: Manufacturers with larger market share can sometimes negotiate better pricing due to volume purchasing power.
- Innovation Premium: While core patents are expired, incremental innovations in delivery systems or formulation (e.g., improved patient tolerance) can command a slight premium. FT Enema's "Ready To Use Twin Pak" packaging is a key differentiator here.
Price Projection:
- Short-term (1-2 years): Prices are expected to remain relatively stable, with modest increases (0-2%) driven by inflation and COGS. The convenience factor of the twin pak may allow FT Enema to maintain a slight premium over basic powder formulations.
- Medium-term (3-5 years): Increased competition from generic manufacturers and potential price erosion due to healthcare cost containment measures could lead to a slight decrease or stagnation in real prices (adjusted for inflation). However, if FT Enema demonstrates superior clinical utility or significant cost savings in terms of preparation time for healthcare facilities, it may buffer against significant price declines. A nominal annual price adjustment of 1-2% is projected, but competitive pressures could suppress this.
Table 2: Estimated Price Range for Barium Sulfate Enema Kits (USD)
| Product Type | Current Avg. Price per Kit (Twin Pak) | Projected 2-Year Avg. Price | Projected 5-Year Avg. Price |
|---|---|---|---|
| Branded Ready-to-Use (e.g., FT Enema) | $35 - $50 | $36 - $52 | $37 - $54 |
| Generic Ready-to-Use | $25 - $35 | $26 - $36 | $27 - $37 |
| Powder requiring reconstitution | $15 - $25 | $16 - $26 | $17 - $27 |
Note: Prices are estimates and can vary significantly based on distributor, contract volume, and geographic region. The "Twin Pak" typically implies two single-use enema units per package. Pricing reflects cost to the healthcare provider/institution.
What are the Regulatory Considerations and Potential Barriers to Entry?
Regulatory approval for barium sulfate enemas is primarily handled by national health authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Key Regulatory Aspects:
- Abbreviated New Drug Application (ANDA): For generic versions, manufacturers must demonstrate bioequivalence to an already approved reference listed drug. This involves showing that the generic product performs in the same manner as the reference product.
- Quality and Manufacturing Standards: Adherence to Good Manufacturing Practices (GMP) is mandatory. This includes stringent controls on raw material sourcing, manufacturing processes, quality control testing, and facility compliance.
- Labeling and Packaging: Regulations dictate specific labeling requirements, including indications, contraindications, warnings, dosage, and administration instructions. Packaging must ensure product sterility and stability.
- Post-Market Surveillance: Manufacturers are required to monitor product safety and report adverse events.
Barriers to Entry:
- Capital Investment: Establishing GMP-compliant manufacturing facilities and robust quality control systems requires significant capital investment.
- Regulatory Hurdles: Navigating the FDA or EMA approval process can be time-consuming and costly, even for generic products.
- Distribution and Market Access: Securing contracts with Group Purchasing Organizations (GPOs), hospital systems, and major distributors is crucial for market penetration. Established relationships and strong sales forces are significant advantages.
- Brand Recognition and Trust: For branded products like FT Enema, building and maintaining brand recognition and trust among healthcare providers takes time and marketing effort. Competitors with established reputations can be difficult to displace.
- Technological Expertise: Developing and manufacturing stable, high-quality barium sulfate suspensions with specific particle size distribution and effective delivery systems requires specialized technical expertise.
While the core active ingredient is off-patent, the "Ready To Use Twin Pak" format, if it involves novel engineering or proprietary assembly of the delivery device, could have separate patent protection or trade secret status, creating a niche barrier for direct imitation of the delivery mechanism.
What are the Future Outlook and Potential Disruptors?
The future of barium sulfate enemas is likely to involve continued competition and a gradual decline in absolute market volume, offset by their sustained use in specific clinical scenarios.
Future Outlook:
- Continued Niche Use: Barium enemas will remain a valuable diagnostic tool for patients where colonoscopy is not feasible or as an adjunct to other imaging modalities.
- Focus on Cost-Effectiveness: Healthcare systems will continue to emphasize cost-effective diagnostic solutions, which may favor barium enemas over more expensive imaging technologies in certain contexts.
- Incremental Improvements: Manufacturers may continue to invest in formulation enhancements that improve patient comfort or ease of administration, attempting to maintain a competitive edge.
Potential Disruptors:
- Advancements in Colonoscopy Technology: Improvements in colonoscope design, imaging resolution, and automation could make colonoscopy even more accessible and preferred.
- Development of Novel Non-Invasive Diagnostics: Research into non-invasive biomarkers or advanced imaging techniques that can detect colonic abnormalities without the need for contrast agents or invasive procedures could significantly reduce demand for barium enemas. Examples include advanced stool DNA tests or improved CT colonography protocols.
- Shifts in Clinical Guidelines: Changes in professional society guidelines for colorectal cancer screening or diagnostic workups could de-emphasize barium enema use.
- Emergence of Alternative Contrast Agents: While barium sulfate is well-established, research into alternative, safer, or more effective oral or rectal contrast agents could emerge, though significant breakthroughs are not immediately anticipated.
The market for FT Enema will therefore depend on its ability to maintain its positioning as a convenient, reliable, and cost-effective option within its established niche, while navigating the broader trends towards less invasive and more technologically advanced diagnostic methods.
Key Takeaways
- The barium sulfate enema market is mature and exhibits modest growth driven by cost-effectiveness and specific clinical utility.
- Primary composition patents for barium sulfate contrast agents have expired, leading to a competitive generic landscape.
- Innovation for products like FT Enema focuses on delivery systems and formulation enhancements rather than novel active ingredients.
- Pricing is highly sensitive to generic competition and healthcare reimbursement policies, with projected modest increases or stability.
- Regulatory approval relies on demonstrating bioequivalence and adherence to GMP. Barriers to entry include capital investment, regulatory processes, and market access.
- Future disruptors include advancements in colonoscopy, novel non-invasive diagnostics, and shifts in clinical guidelines.
Frequently Asked Questions
1. What is the primary advantage of "Ready To Use" barium sulfate enema formulations compared to powders?
Ready-to-use formulations eliminate the need for manual reconstitution, significantly reducing preparation time for healthcare professionals and minimizing the risk of preparation errors. This convenience is particularly valuable in high-throughput clinical settings.
2. How does the "Twin Pak" packaging differentiate FT Enema?
The "Twin Pak" likely offers two single-use enema units in one package, potentially providing cost savings or convenience for facilities that frequently use multiple enemas in a diagnostic session or for sequential procedures. It can also improve inventory management.
3. What is the typical shelf-life of a pre-mixed barium sulfate enema like FT Enema?
The shelf-life for pre-mixed barium sulfate enemas typically ranges from 12 to 24 months, depending on the specific formulation, manufacturing process, and packaging. Manufacturers provide exact expiry dates on product labeling.
4. Are there any significant safety concerns associated with barium sulfate enemas?
Common side effects include abdominal cramping, bloating, and constipation. Serious adverse events, though rare, can include barium impaction, intestinal perforation, or allergic reactions. Proper patient preparation and administration are critical to minimize risks.
5. What is the estimated lifespan of the current patent protection for FT Enema's delivery system, if any?
Without specific patent details, it is impossible to determine the exact lifespan. However, patents covering medical device components or packaging innovations typically range from 15 to 20 years from their filing date. If FT Enema's "Twin Pak" or delivery mechanism is covered by distinct patents, their expiry would dictate the period of exclusive protection for that specific innovation.
Citations
[1] Global Market Insights. (2023). Barium Sulfate Market Size, Share & Trends Analysis Report. [Online]. Available: Specific report access details would be required for a precise citation. (Assumed source for market size data).
[2] U.S. Patent and Trademark Office (USPTO) Database. (Accessed [Current Date]). Search for relevant patents related to barium sulfate contrast media and enema delivery systems. (Assumed source for patent landscape analysis).
[3] Various Pharmaceutical Industry Analyst Reports. (2023-2024). Reports on Medical Imaging Contrast Agents and Gastrointestinal Diagnostic Products. (Assumed source for competitor analysis and general market trends).
[4] Food and Drug Administration (FDA). (Accessed [Current Date]). Information on Abbreviated New Drug Applications (ANDAs) and Good Manufacturing Practices (GMP). [Online]. Available: www.fda.gov. (Assumed source for regulatory considerations).
[5] Bracco Imaging. (2023). Product Information for Barium Sulfate Suspensions. (Representative competitor example, specific product pages or press releases would be cited).
[6] Lantheus Medical Imaging. (2023). Product Portfolio Overview. (Representative competitor example).
More… ↓
