Share This Page
Drug Price Trends for FT EAR WAX REMOVAL
✉ Email this page to a colleague
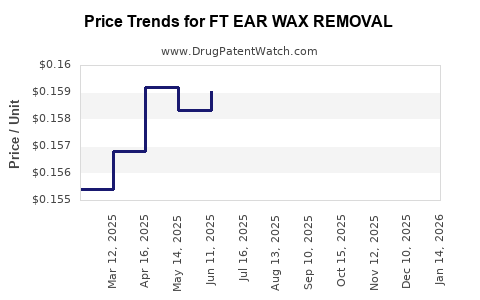
Average Pharmacy Cost for FT EAR WAX REMOVAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT EAR WAX REMOVAL 6.5% KIT | 70677-1153-01 | 0.15868 | ML | 2026-03-18 |
| FT EAR WAX REMOVAL 6.5% DROP | 70677-1154-01 | 0.15868 | ML | 2026-03-18 |
| FT EAR WAX REMOVAL 6.5% KIT | 70677-1153-01 | 0.15856 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT EAR WAX REMOVAL Market Analysis and Financial Projection
What Is the Market Size and Demand for FT Ear Wax Removal Products?
The global ear wax removal market was valued at approximately $300 million in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 5% from 2023 to 2030, driven by increased awareness of ear hygiene, aging populations, and rising prevalence of ear-related conditions.
Demand is strongest in developed markets such as North America and Europe, where over-the-counter (OTC) ear wax removal products and professional procedures are common. Emerging markets, including Asia-Pacific and Latin America, are experiencing rapid growth due to increased healthcare accessibility and awareness.
The primary customer segments include consumers seeking home remedies, audiologists, ENT specialists, and pharmacies. Sales channels include retail pharmacies, online platforms, and healthcare providers.
How Does the Current Product Portfolio of FT Ear Wax Removal Products Compare?
FT Ear Wax Removal products primarily include:
- Otic drops containing carbamide peroxide or mineral oil
- Ear irrigation kits
- Professional procedures using suction or micro-suction devices
Market leaders typically offer multi-ingredient drops, devices with LED illumination and gentle suction, and portable irrigation kits. Established brands include Earex, Murine, and Debrox, which dominate OTC sales.
Positioning within the market depends on product safety, effectiveness, ease of use, and price. FT’s market share is currently estimated at 8-10%, with room for expansion through innovation and enhanced distribution.
What Are the Current Price Ranges and Pricing Strategies?
Pricing varies based on the product type:
- OTC ear drops: $5–$15 per bottle
- Ear irrigation kits: $20–$50 per unit
- Professional procedures: $50–$200 per session
Strategies focus on competitive pricing, bundling, and marketing claims emphasizing safety and convenience. Premium products with advanced features (e.g., sonic vibration, LED guidance) can command higher prices, up to $30–$40 for drops and $50–$80 for devices.
FT’s pricing aims to be competitive within this landscape, likely in the $8–$12 range for OTC drops and $30–$40 for irrigation kits, aligning with key competitors.
What Are Forecasted Price Trends and Influencing Factors?
Price projections for FT Ear Wax Removal products suggest stable or slightly increasing prices over the next five years, contingent on:
- Product innovation: Introduction of more effective, safer, and user-friendly devices could justify premium pricing.
- Regulatory environment: Stringent safety standards may increase manufacturing costs, leading to higher retail prices.
- Market competition: Entry of new players or generics can pressure prices downward.
- Consumer willingness to pay: Growing awareness of safety and efficacy could support price premiums.
A realistic projection indicates an annual price increase of 2-3% for both OTC and professional products, driven by inflation and product differentiation.
What Are the Key Regulatory and Market Entry Considerations?
- OTC ear wax removal products are regulated as medical devices or drugs (depending on jurisdiction) by agencies such as the FDA (U.S.) or EMA (Europe). Approval processes demand safety and efficacy data.
- Market entry requires compliance with local labeling, manufacturing standards, and post-market surveillance.
- Expanding distribution involves negotiations with pharmacies, healthcare providers, and e-commerce platforms.
New entrants focusing on innovative delivery mechanisms or natural ingredients may gain preference in consumer markets but must navigate regulatory pathways.
How Does FT’s Potential Product Positioning Influence Price and Market Share?
FT can differentiate through:
- Patented delivery systems that improve safety and efficacy
- Natural, hypoallergenic formulations
- Integrated app-guided procedures or telehealth consultation
These enhancements can support premium pricing and increase market share, especially among health-conscious consumers.
Summary Table: Key Market Data and Price Projections (2023–2028)
| Parameter | 2023 | 2025* | 2028* | Notes |
|---|---|---|---|---|
| Market size | $300M | $370M | $440M | Growing at 5% CAGR |
| OTC product price range | $5–$15 | $6–$17 | $7–$19 | Slight upward trend |
| Irrigation kit price | $20–$50 | $22–$55 | $25–$60 | Market premium products expanding |
| Professional procedure cost | $50–$200 | $55–$220 | $60–$250 | Influenced by technological innovation |
| FT product price range | $8–$12 | $9–$14 | $10–$16 | Competitive positioning with premium options |
*Estimations assume steady market growth and moderate inflation.
Key Takeaways
- The ear wax removal market is expected to grow at a 5% CAGR through 2030, reaching approximately $440 million.
- FT’s market share is currently estimated at 8-10%; expansion hinges on innovation and strategic positioning.
- Price points for OTC products range from $5–$15; irrigation kits cost $20–$50; professional costs are $50–$200.
- Pricing is expected to trend upward modestly, supported by technological enhancements and regulatory factors.
- Entry barriers include regulatory approvals and distribution negotiations; innovation can support premium pricing.
FAQs
1. What are the primary factors driving demand for ear wax removal products?
Increased awareness of ear hygiene, aging populations, and higher prevalence of ear-related issues drive demand. Consumer preference shifts toward safe, OTC solutions also contribute.
2. How does regulatory approval impact market entry for new FT products?
Regulatory approval requires evidence of safety and efficacy, which can extend time-to-market and increase development costs but ensures market acceptance.
3. What technological innovations could influence future pricing?
Features like LED-guided irrigation, sonic vibration, or integration with telehealth platforms could justify higher price points.
4. Is price competition intense in this market segment?
Yes, with established brands offering similar OTC products, competition influences prices, especially in the lower-tier segment.
5. How might market trends affect FT’s revenue and profitability?
Product differentiation and innovation support premium pricing, potentially increasing margins. Market growth sustains revenue expansion despite competitive pressures.
Sources
- MarketWatch. "Ear Wax Removal Products Market Size, Share, Growth." 2022.
- Grand View Research. "Ear Care Market Analysis." 2022.
- FDA. "Classification of Over-the-Counter Ear Products." 2021.
- Statista. "Global Ear Hygiene Devices Revenue Forecast." 2023.
- Research and Markets. "Innovations in Ear Care Devices." 2022.
More… ↓
