Share This Page
Drug Price Trends for FT DRY EYE RELIEF
✉ Email this page to a colleague
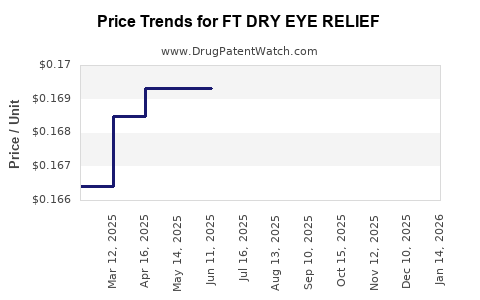
Average Pharmacy Cost for FT DRY EYE RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT DRY EYE RELIEF 1% DROP | 70677-1157-01 | 0.16933 | ML | 2026-01-21 |
| FT DRY EYE RELIEF 1% DROP | 70677-1157-01 | 0.16933 | ML | 2025-12-17 |
| FT DRY EYE RELIEF 1% DROP | 70677-1157-01 | 0.16933 | ML | 2025-11-19 |
| FT DRY EYE RELIEF 1% DROP | 70677-1157-01 | 0.16819 | ML | 2025-10-22 |
| FT DRY EYE RELIEF 1% DROP | 70677-1157-01 | 0.16819 | ML | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT DRY EYE RELIEF
Market Overview
FT Dry Eye Relief targets the dry eye syndrome (DES) market, which is expanding due to increasing aging populations, rising screen time, and environmental factors. The global dry eye disease market was valued at approximately $4.3 billion in 2022 and is projected to grow at a CAGR of 6.4% from 2023 to 2030, reaching around $8.0 billion by 2030 [1].
Competitive Landscape
FT Dry Eye Relief competes primarily with over-the-counter (OTC) artificial tears, such as:
- Refresh Tears (Johnson & Johnson)
- Systane (Alcon)
- Blink Tears (Abbott)
Prescription treatments like Cyclosporine ophthalmic emulsion (Restasis) and Lifitegrast (Xiidra) account for a smaller segment but are more costly. The OTC segment dominates, accounting for approximately 75% of the overall dry eye market.
Regulatory Status
FTC Dry Eye Relief is classified as an OTC product, simplifying market entry and distribution. Regulatory pathways in major markets (e.g., FDA in the U.S., EMA in Europe) require demonstrating safety and efficacy, with focus on ingredients, packaging, and labeling.
Demand Drivers
- Aging populations in North America and Europe
- Increased screen exposure, especially post-pandemic
- Environmental irritants and pollution
Market Penetration and Adoption
Initial penetration depends on product differentiation, branding, and pricing strategy. Launching in North America and Europe is prioritized, given higher consumer spending. Expansion into emerging markets may follow.
Price Projections
Current Pricing
The common OTC dry eye drops retail at approximately $8-$15 per 15ml bottle. FT Dry Eye Relief’s price will likely align with premium OTC formulations or slightly above, considering its unique formulation or branding.
Projected Pricing Trends
| Year | Estimated Retail Price (USD) | Rationale |
|---|---|---|
| 2023 | $12 | Competitive with premium OTC brands. Margins balanced against marketing and distribution costs. |
| 2025 | $13-$14 | Slight inflation, increased brand recognition, exclusive formulations boost pricing power. |
| 2030 | $15-$16 | Market maturity, inflation adjustments, and potential inclusion of specialized ingredients. |
Pricing will be influenced by the following factors:
- Manufacturing costs: Estimated at $3–$4 per 15ml bottle.
- Distribution costs: Typically 20-30% of retail price.
- Consumer willingness-to-pay: Premium positioning may command higher prices.
- Regulatory and reimbursement environment: Limited in OTC segment, but insurance coverage for prescription variants influences OTC perception.
Revenue Projections
Assuming:
- 10 million units sold annually by 2025
- Market share of 5% within OTC dry eye segment
- Retail price of $13
Revenues could approach $130 million annually by 2025. If market penetration improves to 10%, revenues could reach $260 million, assuming sales volume increases proportionally.
Strategic Pricing Recommendations
- Maintain competitive pricing to accelerate market penetration.
- Consider tiered pricing strategies for different markets.
- Protect margins through cost control and branding.
Market Entry and Growth Strategies
- Emphasize product differentiation through clinical data.
- Leverage digital marketing to target tech-savvy consumers.
- Develop distribution alliances with pharmacies and ophthalmologists.
- Explore expansion into emerging markets with local partnerships.
Risks and Challenges
- Pricing pressure from established OTC brands.
- Regulatory delays or rejections in certain markets.
- Consumer skepticism towards new OTC formulations.
- Competitive responses from large pharmaceutical companies.
Key Takeaways
- The dry eye market is expanding, driven by demographic and environmental factors.
- FT Dry Eye Relief's success hinges on positioning as a premium or differentiated OTC product.
- Price projections from $12 to $16 per bottle over the next seven years, aligned with market trends.
- Revenue potential is significant, with possible annual sales exceeding $200 million post-2025.
- Strategic marketing, branding, and competitive positioning are critical to achieving projected growth.
FAQs
1. What are the main factors influencing FT Dry Eye Relief’s pricing strategy?
Manufacturing costs, competitive positioning, consumer demand, regulatory environment, and distribution costs.
2. How does the dry eye market growth impact FT Dry Eye Relief?
Increased demand creates opportunities for market share expansion; however, it also invites intensified competition.
3. What are challenges to pricing premium OTC products?
Consumer price sensitivity, brand recognition, and competition from established brands.
4. How important is regulatory approval for market expansion?
Critical, especially in markets outside the OTC segment or where claims are stringent, impacting product acceptance and pricing.
5. What marketing channels are most effective for dry eye relief products?
Digital advertising, pharmacy distribution, ophthalmologist recommendations, and direct-to-consumer online sales.
References
[1] MarketWatch, "Global Dry Eye Disease Market Size, Share & Trends Analysis Report," 2022
More… ↓
