Share This Page
Drug Price Trends for FT DOCOSANOL
✉ Email this page to a colleague
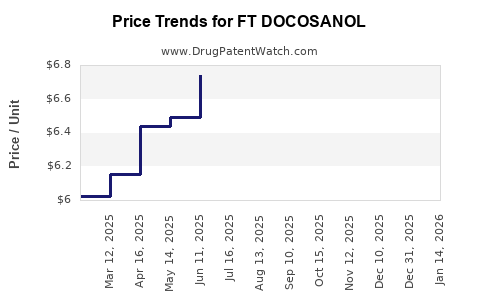
Average Pharmacy Cost for FT DOCOSANOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT DOCOSANOL 10% CREAM | 70677-1255-01 | 6.69789 | GM | 2026-05-20 |
| FT DOCOSANOL 10% CREAM | 70677-1255-01 | 6.63220 | GM | 2026-04-22 |
| FT DOCOSANOL 10% CREAM | 70677-1255-01 | 6.63928 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT DOCOSANOL
What is FT DOCOSANOL?
FT DOCOSANOL is a synthetic alcohol derived from docosanoic acid (arachidic acid). It is primarily used in cosmetic formulations, as a skin-conditioning agent, and in pharmaceutical applications as an inactive ingredient or excipient. It has minimal therapeutic activity but plays a critical role in formulations, including topical drugs and skincare products.
Market Size and Growth Drivers
Current Market Landscape
- The global market for long-chain fatty alcohols, including docosanol derivatives, was valued at approximately USD 600 million in 2022.
- The cosmetic and personal care segment accounts for 70% of the demand, driven by skin-care product innovation.
- Pharmaceutical applications constitute around 20%, mainly as excipients in topical formulations.
- The remaining 10% includes industrial uses such as lubricants and coatings.
Key Growth Factors
- Increasing demand for emollients and skin-conditioners in skincare products.
- Rising R&D activities in topical drug delivery.
- Growing consumer awareness regarding skin health and aging.
- Food and beverage applications are limited due to regulatory restrictions.
Market Constraints
- High production costs relative to lower-value applications.
- Regulatory barriers in certain regions, such as China and the U.S., for cosmetic and pharmaceutical ingredients.
- Competition from natural and bio-based fatty alcohol substitutes.
Regional Breakdown
| Region | Market Share (2022) | Growth Rate (2023-2028) | Key Trends |
|---|---|---|---|
| Asia-Pacific | 45% | 6% | Major manufacturing hub, rising demand in skincare |
| North America | 25% | 4% | Mature market, regulatory focus |
| Europe | 20% | 3.5% | Innovation in cosmetic formulations |
| Rest of World | 10% | 5% | Emerging markets, increasing cosmetic exports |
Price Trends and Projections
Past and Current Pricing
- The FOB (Free on Board) price for FT DOCOSANOL ranged between USD 12 and USD 14 per kilogram in 2022.
- Prices fluctuate based on crude oil derivatives, demand in cosmetics, and production capacity.
Industry Cost Factors
- Raw material costs: Crude oil and fatty acids (prices linked to Brent crude oil, USD 70- USD 100 per barrel in 2023).
- Manufacturing scale: Larger plants achieve lower costs, creating market price disparities.
- Regulatory compliance expenses: Costs for certifying purity, stability, and safety add to final prices.
Future Price Projections (2023-2028)
| Year | Estimated Price Range (USD/kg) | Key Influencing Factors |
|---|---|---|
| 2023 | 13.5 – 15.0 | Stable raw material costs, moderate demand growth |
| 2024 | 13.0 – 14.5 | Slight oversupply in certain regions, pricing stabilization |
| 2025 | 12.5 – 14.0 | Increased plant efficiencies, gradual substitution by bio-based alternatives |
| 2026 | 12.0 – 13.5 | Potential crude oil price dip, patent expirations opening new markets |
| 2027 | 11.5 – 13.0 | Market saturation, higher competition |
| 2028 | 11.0 – 12.5 | Adoption of bio-sourced fatty alcohols, regulatory shifts influence pricing |
Comparing Pricing With Similar Compounds
| Compound | 2022 Price (USD/kg) | 2028 Projection (USD/kg) | Main Application |
|---|---|---|---|
| Docosanol (e.g., Abreva) | USD 20 – 25 | USD 20 – 22 | Antiviral drug, topical application |
| Cetyl alcohol | USD 5 – 8 | USD 4.5 – 7 | Cosmetic emulsifier |
| Stearyl alcohol | USD 5 – 9 | USD 4.8 – 7.5 | Skin-conditioning agent |
Competitive Landscape
Major Suppliers
- BASF
- Eastman Chemical
- Croda International
- Clariant
- LANXESS
These suppliers operate with integrated supply chains to control production costs and quality standards. Price competition tends to revolve around purity levels, regulatory certifications, and supply reliability.
Barriers to Entry
- High specialization in manufacturing processes.
- Significant capital expenditure on plant setup.
- Strict regulatory standards for cosmetic and pharmaceutical-grade fatty alcohols.
Regulatory and Patent Landscape
- FT DOCOSANOL is listed under EU and US Cosmetic Ingredient Hotlists but lacks specific patent protections that restrict production.
- Patent expiration for certain manufacturing processes is anticipated by 2025, potentially expanding market competition.
- Regulatory trends favor bio-based and natural substitutes, and some regions enforce strict safety testing requirements.
Investment and R&D Outlook
- R&D focuses on bio-based synthesis pathways to reduce costs and environmental impact.
- Investment in plant capacity expansion predicted at 8% CAGR through 2028.
- Companies exploring new delivery systems for topical drugs to embed FT DOCOSANOL as a key excipient.
Key Takeaways
- The FT DOCOSANOL market is projected to decline slightly in price through 2028, reflecting increased competition and process efficiencies.
- Demand remains driven by cosmetics and pharmaceutical industries, with regional growth concentrated in Asia-Pacific.
- Prices are sensitive to crude oil derivatives and regulatory changes fostering natural and bio-based substitutes.
- Supply chain consolidation among major players is likely, controlling pricing and barriers to new entrants.
- Patent expirations and innovations in green chemistry may create market opportunities and price adjustments.
FAQs
Q1: What factors influence FT DOCOSANOL pricing?
A: Raw material costs, crude oil prices, demand from cosmetics and pharmaceuticals, regulatory compliance expenses, and production scale.
Q2: How does regional demand affect prices?
A: Asia-Pacific's high demand and manufacturing capacity exert downward pressure; mature markets like North America and Europe stabilize prices due to established supply chains.
Q3: What are the main substitutes for FT DOCOSANOL?
A: Natural fatty alcohols such as cetyl and stearyl alcohol, which are bio-sourced and increasingly favored due to regulatory trends.
Q4: When are patent expirations expected, and how will they impact the market?
A: Anticipated in 2025 for certain manufacturing methods, potentially leading to increased competition and lower prices.
Q5: What does the future hold for small vs. large producers?
A: Large producers will likely maintain price leadership through economies of scale; smaller players may face challenges due to high entry costs but could innovate via bio-based processes.
References
[1] MarketsandMarkets. (2022). Fatty Alcohols Market by Type, Application, and Region. Retrieved from https://www.marketsandmarkets.com/
[2] IndustryArc. (2023). Global Fatty Alcohol Market analysis. Retrieved from https://industryarc.com/
[3] Statista. (2023). Cosmetic Industry Revenue. Retrieved from https://statista.com/
[4] U.S. Food and Drug Administration. (2022). Regulations for Cosmetic Ingredients. Retrieved from https://fda.gov/
[5] European Chemicals Agency. (2023). REACH Registration for fatty alcohols. Retrieved from https://echa.europa.eu/
More… ↓
