Share This Page
Drug Price Trends for FT CLOTRIMAZOLE
✉ Email this page to a colleague
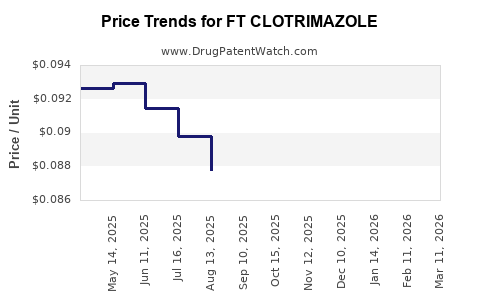
Average Pharmacy Cost for FT CLOTRIMAZOLE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT CLOTRIMAZOLE 1% VAG CREAM | 70677-1228-01 | 0.08389 | GM | 2026-03-18 |
| FT CLOTRIMAZOLE 1% VAG CREAM | 70677-1262-01 | 0.08389 | GM | 2026-03-18 |
| FT CLOTRIMAZOLE-3 2% CREAM | 70677-1231-01 | 0.31625 | GM | 2026-03-18 |
| FT CLOTRIMAZOLE 1% VAG CREAM | 70677-1228-01 | 0.08482 | GM | 2026-02-18 |
| FT CLOTRIMAZOLE-3 2% CREAM | 70677-1231-01 | 0.31665 | GM | 2026-02-18 |
| FT CLOTRIMAZOLE 1% VAG CREAM | 70677-1262-01 | 0.08482 | GM | 2026-02-18 |
| FT CLOTRIMAZOLE-3 2% CREAM | 70677-1231-01 | 0.32193 | GM | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT CLOTRIMAZOLE Market Analysis and Financial Projection
What Is FT Clotrimazole?
FT Clotrimazole is a formulation combining clotrimazole, an antifungal agent, with an excipient or enhancer to improve bioavailability or efficacy. Clotrimazole is widely used to treat fungal infections such as athlete’s foot, candidiasis, and tinea infections. The "FT" signifies a specific formulation or delivery method, possibly topical film-coated or enhanced-release forms. Its patent status and regulatory approvals depend on the formulation specifics.
Market Landscape: Current Size and Key Players
Global Market Overview
The antifungal drugs market, driven by rising fungal infection prevalence, was valued at approximately USD 12 billion in 2022. Clotrimazole remains a top-selling topical antifungal globally, especially in over-the-counter (OTC) segments. A significant share of the growth derives from demand in North America, Europe, and Asia-Pacific.
Key Manufacturers
- Bayer AG (Lotrimin)
- Johnson & Johnson
- Mylan Pharmaceuticals
- Cipla
- Dr. Reddy’s Laboratories
These companies produce both generic and branded formulations. FT Clotrimazole's market penetration depends on patents, formulation advantages, and regional approvals.
Market Segments
- Topical creams and lotions: Account for 70% of sales.
- Lozenges and oral formulations: Minority segment.
- Combination therapies: Emerging, often with corticosteroids for inflammatory fungal infections.
Regulatory Status
Most standard clotrimazole formulations are OTC or prescription medications. Patent specifics of FT Clotrimazole influence its market exclusivity. If it is a patented formulation, it can command premium pricing; otherwise, generic competition dominates.
Price Projections: Factors and Trends
Current Pricing
- OTC topical creams: USD 4–15 per 30g tube.
- Prescription formulations: USD 10–25 per tube, depending on concentration and formulation.
Price Trends
Prices are declining gradually owing to patent expiry and increased generic entry. Exclusive formulations or improved delivery methods may sustain higher prices temporarily.
Forecasts (2023–2028)
| Year | Estimated Price Range (USD) per unit | Notes |
|---|---|---|
| 2023 | 4–25 | Mix of generics and branded products. |
| 2024 | 4–23 | Increased generic competition decreases prices. |
| 2025 | 4–20 | Market stabilization; possible price erosion accelerates. |
| 2026 | 4–18 | New formulations or delivery methods could sustain prices. |
| 2027 | 4–15 | Generic dominance resumes. |
| 2028 | 4–15 | Prices stabilize, minor fluctuations. |
Price Drivers
- Patent status of the formulation.
- Regional pricing regulations.
- Competition level, especially from generics.
- Introduction of enhanced formulations with improved efficacy or convenience.
- Market penetration in emerging economies.
Key Market Dynamics
Patent and Regulatory Environment
Patents on topical clotrimazole formulations expired or are nearing expiry in many regions, facilitating generic entry. A patent for a novel FT formulation can extend exclusivity, potentially keeping prices higher for a limited period.
Impact of Licensing and Local Regulations
Stringent regulations in the U.S. and EU can delay the launch of new formulations, impacting pricing and market share. Conversely, regions with less regulatory hurdles, like some Asian countries, exhibit more rapid price erosion for generics.
Emerging Trends
- Development of long-acting or sustained-release formulations.
- OTC switches facilitated by regulatory changes.
- Combination products with corticosteroids for inflammatory infections.
Market Entry and Expansion Opportunities
- Developing formulations with enhanced absorption or reduced application frequency.
- Targeting markets with high prevalence of fungal infections and low OTC regulation.
- Strategic licensing agreements to accelerate distribution.
Risks and Challenges
- Price erosion due to generic competition.
- Regulatory delays or restrictions.
- Consumer preference shifting toward oral or systemic antifungals in certain cases.
- Regional disparities in reimbursement and pricing policies.
Key Takeaways
- The FT Clotrimazole market is primarily driven by the existing generic competition alongside ongoing demand for effective antifungal treatments.
- Prices are expected to decline modestly over the next five years, with the potential for temporary price stabilization for innovative formulations.
- Market growth in emerging economies offers opportunities but is tempered by regulatory and pricing challenges.
- Patent status heavily influences pricing dynamics; patent expiry accelerates price erosion.
- Entry of combination therapies and novel delivery systems could create niche pricing and market opportunities.
FAQs
1. How does patent protection influence FT Clotrimazole pricing?
Patent protection allows for exclusive marketing, enabling higher prices. Expiry or invalidation leads to increased generic competition and price reductions.
2. What regions are expected to see the fastest market growth?
Emerging economies in Asia-Pacific and Latin America are experiencing rapid growth due to rising fungal infection rates and lower regulatory barriers.
3. Are there significant differences between branded and generic FT Clotrimazole formulations?
Branded formulations generally cost more, especially if protected by patents. Generic options are typically priced 30–50% lower.
4. How will new formulations impact the market?
Enhanced delivery systems and combination therapies may command premium prices and extend market share for innovator brands.
5. What is the outlook for OTC availability of FT Clotrimazole?
OTC status is likely to increase worldwide, pressuring prices downward but expanding consumer access.
Sources:
[1] MarketsandMarkets. "Antifungal Drugs Market," 2022.
[2] GlobalData, "Topical Antifungal Market Insights," 2023.
[3] FDA and EMA drug approval databases.
[4] IQVIA pricing data, 2022.
More… ↓
