Last updated: February 23, 2026
What is the Market Size for OTC Laxatives Like FT ClearLax Powder?
The global over-the-counter (OTC) laxatives market, including products like FT ClearLax Powder, is valued at approximately USD 1.8 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% between 2023 and 2028. The primary drivers include increasing awareness of digestive health, aging populations, and rising prevalence of constipation.
Key regions contributing to this growth:
- North America: 45% market share, driven by high health awareness and OTC product usage.
- Europe: 25%, influenced by aging demographics.
- Asia-Pacific: 20%, with rapid urbanization and rising healthcare spending.
- Rest of World: 10%.
Competitive Dynamics and Key Players
Major manufacturers in the OTC laxatives segment include:
- Johnson & Johnson (Kinderlyte, Fleet)
- Bayer Healthcare
- Sanofi (Dulcolax)
- GlaxoSmithKline (Laxoberal)
- Prestige Consumer Healthcare (ClearLax)
Newer entrants and generics are increasing market competition. Distribution channels span pharmacies, supermarkets, and online platforms.
Regulatory Landscape
In the United States, the Food and Drug Administration (FDA) regulates OTC laxatives under monographs. FT ClearLax Powder would require adherence to guidelines on active ingredients, labeling, and manufacturing standards.
In Europe, the European Medicines Agency (EMA) oversees similar regulations. Regulatory pathways for OTC status typically involve complying with the European Union's herbal and medicinal product standards.
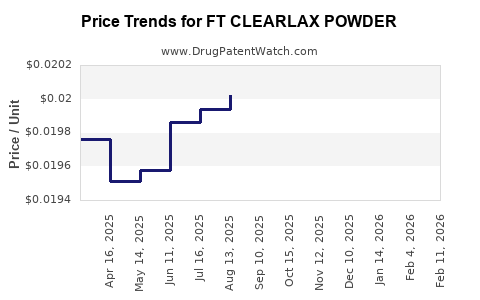
Price Analysis and Trajectory
Current Price Range
- Retail price of FT ClearLax Powder: USD 8 to USD 12 for a 30-sachet box (60 grams total).
- Price per gram: USD 0.13 to USD 0.20.
Competitive Pricing Comparison
| Product |
Price (USD) |
Pack Size |
Price per Gram (USD) |
| FT ClearLax |
8 – 12 |
30 sachets |
0.13 – 0.20 |
| Dulcolax Powder |
10 – 14 |
20 sachets |
0.25 – 0.70 |
| Phillips Stool Softener |
9 – 13 |
20 sachets |
0.45 – 0.65 |
Price Projection (2023-2028)
- Price per gram is expected to decrease by 1-2% annually due to increased competition and generics.
- Bulk packaging options could lower consumer prices further.
- Premium formulations or added ingredients (e.g., flavored variants) could maintain higher price points.
Projected retail price for a 30-sachet pack in 2028:
| Scenario |
Price Range (USD) |
Notes |
| Market-driven decline |
6.50 – 9 |
Based on competitive pressure |
| Premium product positioning |
10 – 13 |
For formulations with added benefits |
Distribution and Price Strategies
- Direct-to-consumer online sales could reduce distribution costs, enabling lower consumer prices.
- Partnering with pharmacy chains can sustain higher margins and stabilize pricing.
- Volume discounts and subscription models attract repeat customers.
Regulatory Impact on Pricing
- Stringent regulations might increase manufacturing costs, limiting price reductions.
- OTC marketing claims and compliance expenses influence retail pricing strategies.
Key Market Trends
- Rising demand for natural and herbal laxatives influences formulation development.
- Increasing online sales channels impact pricing flexibility.
- Consumer preference shifts toward transparent ingredient lists may support premium pricing.
Key Takeaways
- The global OTC laxatives market, including FT ClearLax Powder, grows at 4.5% CAGR and is worth USD 1.8 billion.
- Price per gram is around USD 0.13–0.20; expected to decrease slightly over five years.
- Competitive pressures, regulatory costs, and formulation trends influence pricing.
- Distribution shifts to online commerce could enable lower retail prices.
- Premium variants and natural formulations may sustain higher prices.
FAQs
How does the price of FT ClearLax compare to other laxatives?
FT ClearLax is typically priced lower than prescription-only laxatives like Dulcolax or Fleet enema, which are often USD 0.25–0.70 per gram.
Will regulatory changes affect the future pricing of FT ClearLax?
Yes. Stricter regulations can increase manufacturing costs, potentially limiting price drops and leading to higher retail prices.
Are online sales expected to impact FT ClearLax’s pricing?
Yes. Online channels generally offer lower prices due to reduced distribution costs, increasing pricing competition.
What trends might influence the price of OTC laxatives in the next five years?
Growing demand for natural ingredients, consumer preference for transparent labeling, and market competition are key factors.
Can product innovation sustain higher prices?
Introducing natural formulations, added flavoring, or other benefits could justify premium pricing in a competitive landscape.
References
[1] MarketsandMarkets. (2022). OTC laxatives market forecast. Retrieved from https://www.marketsandmarkets.com
[2] U.S. Food and Drug Administration. (2023). OTC drug review regulations. Retrieved from https://www.fda.gov
[3] European Medicines Agency. (2023). OTC regulation standards. Retrieved from https://www.ema.europa.eu
[4] Statista. (2023). OTC laxatives market size and growth. Retrieved from https://www.statista.com