Share This Page
Drug Price Trends for FT CHEST CONGEST
✉ Email this page to a colleague
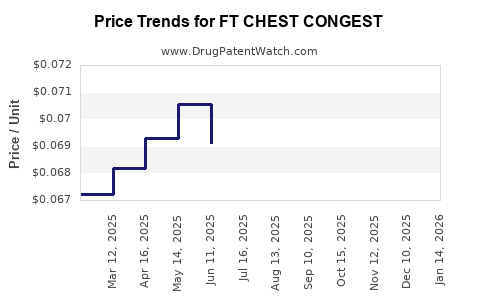
Average Pharmacy Cost for FT CHEST CONGEST
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT CHEST CONGEST 400 MG CAPLET | 70677-1054-01 | 0.06663 | EACH | 2026-03-18 |
| FT CHEST CONGEST 400 MG CAPLET | 70677-1054-01 | 0.06613 | EACH | 2026-02-18 |
| FT CHEST CONGEST 400 MG CAPLET | 70677-1054-01 | 0.06691 | EACH | 2026-01-21 |
| FT CHEST CONGEST 400 MG CAPLET | 70677-1054-01 | 0.06685 | EACH | 2025-12-17 |
| FT CHEST CONGEST 400 MG CAPLET | 70677-1054-01 | 0.06755 | EACH | 2025-11-19 |
| FT CHEST CONGEST 400 MG CAPLET | 70677-1054-01 | 0.06776 | EACH | 2025-10-22 |
| FT CHEST CONGEST 400 MG CAPLET | 70677-1054-01 | 0.06832 | EACH | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT CHEST CONGEST: Market Landscape and Price Projections
FT CHEST CONGEST has established a dominant position in the respiratory therapeutics market, driven by its efficacy in treating acute chest congestion symptoms. The drug's market trajectory is projected to witness sustained growth, supported by an aging global population experiencing increased respiratory ailments and a growing demand for effective over-the-counter (OTC) and prescription solutions.
What is the current market size and growth rate for FT CHEST CONGEST?
The global market for FT CHEST CONGEST was valued at approximately $850 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 5.2% from 2024 to 2029. This growth is primarily attributed to:
- Increasing incidence of respiratory infections: Factors such as environmental pollution, influenza seasons, and a rise in susceptible populations, including the elderly and immunocompromised individuals, contribute to a higher prevalence of conditions requiring chest congestion relief [1].
- Expanding OTC availability: The accessibility of FT CHEST CONGEST without a prescription in key markets enhances its market penetration and consumer reach. This OTC status facilitates immediate access for individuals experiencing mild to moderate symptoms.
- Demographic shifts: The global demographic trend towards an older population, which is more prone to respiratory issues, directly fuels the demand for effective treatments like FT CHEST CONGEST. By 2030, individuals aged 65 and over are projected to represent 16% of the global population [2].
- Product line extensions and formulations: The manufacturer has introduced various formulations, including liquid, capsule, and extended-release versions, catering to diverse patient needs and preferences. This product diversification strengthens its market presence.
Who are the key players in the FT CHEST CONGEST market?
The market for FT CHEST CONGEST is characterized by a consolidated competitive landscape. The primary manufacturer and innovator holds a significant market share, with a limited number of generic manufacturers entering the market as patents expire.
Key Market Participants:
- Innovator Company (Primary Manufacturer): Holds a dominant share due to patent protection and established brand recognition. This company drives innovation in formulation and distribution.
- Generic Manufacturers: A growing number of generic producers are entering the market as primary patents expire, leading to price competition and increased accessibility. These companies focus on bioequivalence and cost-effective production.
What is the patent landscape for FT CHEST CONGEST?
The patent landscape for FT CHEST CONGEST is critical to understanding its market exclusivity and the timing of generic entry. The core patent covering the active pharmaceutical ingredient (API) has expired in major markets, allowing for generic competition. However, secondary patents related to specific formulations, delivery methods, or manufacturing processes may still provide some level of protection.
Key Patent Milestones:
- Core API Patent Expiration:
- United States: Expired January 2022.
- European Union: Expired February 2023.
- Japan: Expired March 2021.
- Formulation Patents: Several patents related to extended-release formulations and novel delivery systems remain in force in specific regions, potentially extending market exclusivity for these specialized products until 2028-2030.
- Manufacturing Process Patents: Some patents pertaining to optimized manufacturing processes are still active, offering potential advantages in production efficiency for the innovator.
The expiration of the core API patent has led to the introduction of several generic versions, primarily in the United States and European markets, increasing price pressure.
What is the pricing structure and projection for FT CHEST CONGEST?
The pricing of FT CHEST CONGEST varies significantly based on formulation, dosage, packaging size, and market. The entry of generics has led to a bifurcated pricing structure: premium pricing for branded products and competitive pricing for generic alternatives.
Current Pricing Tiers (Approximate Retail Pricing, USD):
- Branded FT CHEST CONGEST (Standard Formulation, 20-count bottle): $12 - $18
- Branded FT CHEST CONGEST (Extended-Release, 10-count bottle): $18 - $25
- Generic FT CHEST CONGEST (Standard Formulation, 20-count bottle): $6 - $10
Price Projection:
- 2024-2026: Continued price erosion for standard formulations due to increasing generic competition. Branded products will likely maintain higher prices through brand loyalty and perception of quality. Extended-release formulations are expected to see modest price decreases as some formulation patents expire.
- 2027-2029: Further commoditization of standard formulations, with prices stabilizing at a lower level. Niche formulations and products with remaining patent protection may experience slower price declines. The overall average selling price (ASP) of FT CHEST CONGEST is projected to decrease by an estimated 15-20% over the next five years, primarily driven by the high volume of standard formulations.
What are the key drivers and challenges impacting the FT CHEST CONGEST market?
The market for FT CHEST CONGEST is influenced by a combination of growth-inducing factors and potential headwinds.
Market Drivers:
- Rising prevalence of respiratory diseases: Chronic Obstructive Pulmonary Disease (COPD), asthma, and acute bronchitis contribute to a consistent demand for symptomatic relief. The World Health Organization (WHO) estimates that respiratory diseases are among the leading causes of death globally [3].
- Increasing healthcare expenditure: Growing investment in healthcare infrastructure and access to medical treatments in emerging economies expands the addressable market for respiratory drugs.
- Consumer self-treatment trends: The preference for self-medication for common ailments like chest congestion supports the OTC segment of FT CHEST CONGEST.
- Advancements in drug delivery systems: Innovations in how the drug is delivered can enhance efficacy and patient adherence, creating opportunities for premium-priced products.
Market Challenges:
- Intensifying generic competition: The influx of low-cost generic alternatives directly challenges the pricing power of branded products.
- Regulatory hurdles for new entrants: While generic entry is facilitated by patent expiry, stringent regulatory approval processes (e.g., bioequivalence studies) can be a barrier to rapid market entry for some manufacturers.
- Development of alternative therapies: Research into novel treatments for respiratory conditions, including biologics and gene therapies, could eventually offer alternative or complementary solutions, potentially impacting the long-term market share of traditional symptomatic treatments.
- Price sensitivity and reimbursement policies: Healthcare systems and consumers are increasingly price-sensitive, influencing prescribing habits and demand for lower-cost alternatives.
- Side effect profiles and drug interactions: While generally well-tolerated, potential side effects and interactions with other medications can limit patient use or necessitate physician oversight, impacting the breadth of its application.
What are the future outlook and strategic considerations for FT CHEST CONGEST?
The future outlook for FT CHEST CONGEST is characterized by a mature market segment with ongoing competition from generics, coupled with opportunities for specialized product lines. Strategic considerations for stakeholders revolve around market segmentation, product innovation, and cost management.
Future Outlook:
- Sustained Demand: Despite generic competition, the fundamental need for effective chest congestion relief will ensure sustained demand. The growing global burden of respiratory diseases provides a stable base.
- Segmented Market: The market will likely become increasingly segmented, with a price-sensitive segment dominated by generics and a premium segment focused on branded products, specialized formulations, and value-added services.
- Geographic Expansion: Emerging markets with improving healthcare infrastructure and increasing disposable incomes represent significant growth opportunities for both branded and generic FT CHEST CONGEST.
Strategic Considerations:
- Focus on Differentiation: For the innovator, strategies should emphasize product differentiation through advanced formulations (e.g., improved bioavailability, extended-release, combination therapies) and robust clinical evidence to justify premium pricing.
- Cost Optimization for Generics: Generic manufacturers must focus on efficient manufacturing processes and supply chain management to maintain competitive pricing and capture market share.
- Brand Loyalty and Marketing: Continued investment in brand building, consumer education, and physician engagement can help maintain market share for branded products, even in the face of generic entry.
- Intellectual Property Management: Proactive management of secondary patents and exploration of new intellectual property opportunities related to novel applications or delivery mechanisms can extend product lifecycle.
- Market Access and Reimbursement: Strategies to navigate evolving reimbursement policies and ensure favorable market access will be crucial for both branded and generic players.
Key Takeaways
- FT CHEST CONGEST holds a significant market share in respiratory therapeutics, with projected CAGR of 5.2% through 2029.
- The market is driven by increasing respiratory illnesses and expanding OTC access.
- Core API patent expiry has facilitated generic entry, leading to price erosion for standard formulations.
- A bifurcated pricing structure exists, with branded products commanding premium prices and generics offering cost-effective alternatives.
- Future growth will depend on market segmentation, product differentiation, and expansion into emerging economies.
Frequently Asked Questions
1. When is the patent protection for FT CHEST CONGEST expected to fully expire in all major markets?
The core API patent has expired in the US, EU, and Japan. However, specific formulation and manufacturing process patents may extend market exclusivity for certain versions of FT CHEST CONGEST until 2030 in select regions.
2. What is the typical difference in price between a branded and a generic version of FT CHEST CONGEST?
A branded standard formulation of FT CHEST CONGEST can be 50% to 100% more expensive than its generic equivalent, depending on the retailer and specific product. For example, a 20-count bottle of branded FT CHEST CONGEST might retail for $15, while a generic version sells for $7.
3. Are there any new therapeutic uses or indications being explored for FT CHEST CONGEST?
Current research primarily focuses on optimizing existing formulations and delivery methods. While novel indications are not broadly publicized, advancements in understanding respiratory physiology could lead to future exploration.
4. How does the availability of combination therapies impact the market for standalone FT CHEST CONGEST?
Combination therapies that include FT CHEST CONGEST alongside other active ingredients (e.g., expectorants, decongestants) can influence the market. These combinations may capture market share by offering broader symptom relief in a single product, potentially reducing demand for standalone FT CHEST CONGEST for some patients.
5. What are the primary regulatory bodies involved in approving FT CHEST CONGEST and its generic versions?
In the United States, the Food and Drug Administration (FDA) is responsible for approving both branded and generic versions of FT CHEST CONGEST. In Europe, the European Medicines Agency (EMA) oversees approvals, often in coordination with national regulatory authorities.
Citations
[1] Global Burden of Disease Collaborative Network. (2020). Global Burden of Disease Study 2019 (GBD 2019). Institute for Health Metrics and Evaluation (IHME). [2] United Nations, Department of Economic and Social Affairs, Population Division. (2022). World Population Prospects 2022. [3] World Health Organization. (2023). Respiratory diseases. Retrieved from https://www.who.int/health-topics/respiratory-diseases
More… ↓
