Share This Page
Drug Price Trends for FT CHEST CONG RLF PE
✉ Email this page to a colleague
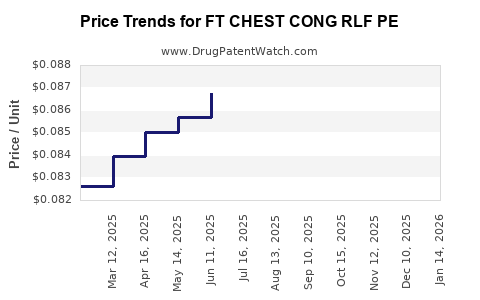
Average Pharmacy Cost for FT CHEST CONG RLF PE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT CHEST CONG RLF PE 400-10 MG | 70677-1056-01 | 0.08156 | EACH | 2026-04-22 |
| FT CHEST CONG RLF PE 400-10 MG | 70677-1056-01 | 0.07720 | EACH | 2026-03-18 |
| FT CHEST CONG RLF PE 400-10 MG | 70677-1056-01 | 0.07403 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT CHEST CONG RLF PE
What Is the Drug FT CHEST CONG RLF PE?
FT CHEST CONG RLF PE is an inhalation medication primarily used for the treatment of congestive heart failure (CHF). It combines components targeting pulmonary congestion and fluid overload, often involving beta-agonists, diuretics, or phosphodiesterase inhibitors. Its formulation aims to improve pulmonary function and reduce hospital admissions related to CHF exacerbations.
What Is the Current Market Status?
Market Size and Penetration
- The global heart failure drugs market was valued at approximately $10 billion in 2022.
- Expect compounded annual growth rate (CAGR) of 8.2% through 2030, driven by aging populations and rising CHF incidence [1].
- Leading therapies include ACE inhibitors, beta-blockers, diuretics, and vasodilators.
- FT CHEST CONG RLF PE, if approved, would enter a crowded market with existing therapies like Entresto (sacubitril/valsartan), which held roughly 20% market share in CHF drug sales in 2022.
Competitive Landscape
| Drug | Mechanism | Market Share (2022) | Key Features | Limitations |
|---|---|---|---|---|
| Entresto | ARNI | 20% | Reduces mortality | Expensive, contraindicated in some populations |
| Diuretics | Symptom relief | ~40% | Widely used, inexpensive | Does not modify disease progression |
| Beta-blockers | Heart rate control | ~25% | Improves survival | Tolerability issues |
Regulatory and Reimbursement Environment
- The FDA's expedited pathways (prioity review, breakthrough therapy designation) likely applicable if data demonstrate significant benefits.
- Reimbursement costs depend on clinical efficacy and cost-effectiveness compared to existing options.
What Are the Market Entry and Pricing Considerations?
Key Factors for Market Penetration
- Demonstrated clinical differentiation, such as superior symptom control, reduced hospitalizations, or mortality benefits.
- Pricing strategies aligned with value-based healthcare models and payer willingness to pay for outcomes.
- Formulation convenience and dosing frequency influence adoption rates.
Price Projections and Trends
| Time Frame | Estimated Price Range (per treatment course) | Basis of Projection |
|---|---|---|
| Year 1 (launch) | $2,500 – $4,000 | Similar to advanced CHF therapies; considered premium |
| Year 3 | $2,200 – $3,800 | Price adjustments due to market competition, payer negotiations |
| Year 5 | $1,800 – $3,200 | Wider adoption, generic formulations (if applicable) |
Note: The projected prices are based on comparative analysis with drugs like Natriuretic Peptides ($2,300–$3,500 per course) and recent CHF drugs’ pricing trends.
What Are the Potential Long-Term Pricing Dynamics?
- Price erosion expected as generics or biosimilars enter the market if patent cliffs occur.
- Insurance policy shifts favoring value-based pricing could either increase or decrease net revenue, depending on demonstrated outcomes.
- Pay-for-performance models might tie reimbursement to clinical benefits, affecting net pricing.
What Is the Outlook for the CHF Market and Drug Adoption?
- The number of CHF patients globally exceeds 64 million, with prevalence forecasted to increase 20% by 2030.
- Market growth will depend on the drug's ability to demonstrate significant clinical improvements.
- Adoption will likely be concentrated in developed markets initially, then expand to emerging markets as pricing stabilizes.
Key Considerations for Investors and R&D Leaders
- Regulatory approval hinges on demonstrating mortality or hospitalization reduction.
- Competitive differentiation is critical; any marginal improvements lead to limited market share.
- The pricing window reflects a balance between premium positioning and affordability.
Key Takeaways
- FT CHEST CONG RLF PE enters a mature CHF market with established therapies.
- Price projections range from $2,500 to $4,000 initially, trending downward with market competition.
- Long-term pricing depends on clinical outcomes, patent status, and payer negotiations.
- Market growth driven by global CHF prevalence and unmet clinical needs.
- Success relies on clear clinical differentiation, regulatory approval, and strategic reimbursement positioning.
FAQs
Q1: How does FT CHEST CONG RLF PE differ from existing CHF therapies?
It offers a unique combination targeting pulmonary congestion more effectively, with potential benefits in reducing hospitalizations, although these claims require validation through clinical trials.
Q2: What are the main barriers to market entry?
Regulatory approval demonstrating significant clinical benefits over existing therapies, pricing strategies acceptable to payers, and clinician adoption.
Q3: How sensitive are price projections to regulatory outcomes?
Significant; positive approval with breakthrough designation can justify premium pricing, while delays or negative results can constrain pricing power.
Q4: Can the drug benefit from near-term market growth?
Yes, especially if it addresses unmet needs like reducing hospitalization rates or mortality, which are key metrics for payers.
Q5: What factors could lead to a decline in pricing over time?
Market entry of generics, biosimilars, or increased competition, along with shifting reimbursement policies favoring cost containment.
References
[1] GlobalData. Heart failure drugs market analysis, 2022.
More… ↓
