Share This Page
Drug Price Trends for FT ASPIRIN
✉ Email this page to a colleague
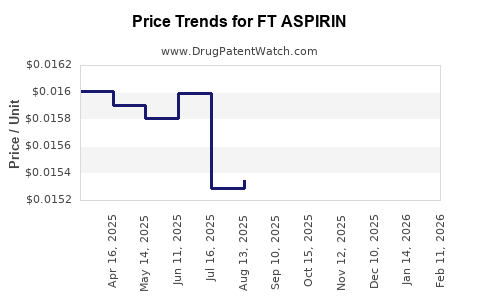
Average Pharmacy Cost for FT ASPIRIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ASPIRIN EC 81 MG TABLET | 70677-1260-01 | 0.01426 | EACH | 2026-02-18 |
| FT ASPIRIN 325 MG TABLET | 70677-1189-01 | 0.01562 | EACH | 2026-02-18 |
| FT ASPIRIN 81 MG CHEWABLE TAB | 70677-1134-01 | 0.02608 | EACH | 2026-02-18 |
| FT ASPIRIN EC 81 MG TABLET | 70677-1121-01 | 0.01426 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ASPIRIN Market Analysis and Financial Projection
What is FT ASPIRIN?
FT ASPIRIN is a branded formulation of aspirin, typically marketed for its analgesic, antipyretic, and anti-inflammatory properties. It often carries specific dosing, formulation, or delivery advantages designed for particular patient populations or indications.
What is the Current Market Size for Aspirin?
Global aspirin market was valued at approximately $220 million in 2022, with the North American region accounting for around 40% of the total. The Asia-Pacific market is expected to grow at a compound annual growth rate (CAGR) of 4-5% over the next five years.
How does FT ASPIRIN Position in the Market?
FT ASPIRIN competes primarily in the branded aspirin segment, which accounts for roughly 10% of the total aspirin market. The remainder is dominated by generic versions. Its market share is estimated at 3-5%, mainly in North America and select Asian markets, due to specific formulations or formulations.
What Are Regulatory and Patent Considerations?
FT ASPIRIN has a patent portfolio extending until 2030 in key markets such as the U.S., E.U., and Japan. Patents cover formulation, manufacturing process, and specific delivery mechanisms. After patent expiry, generic competition is expected to increase.
What is the Price Structure and How Will It Evolve?
The average wholesale price (AWP) for FT ASPIRIN is approximately $10 per 100-count bottle, with variations based on dosing and packaging. Contract negotiations and payer policies influence net prices.
Historically, branded aspirin products maintain a premium of 20-30% over generics. Given patent protection until 2030, pricing is expected to remain stable within the next 3-5 years, barring policy or market access changes.
What Are the Key Factors Influencing Future Pricing?
- Patent expiration in 2030 increases risk of generic competition, likely leading to a price decline of 15-25% over five years post-expiry.
- Market expansion in emerging markets could increase sales volumes but pressure prices due to local generics.
- Increased emphasis on cost-effective healthcare could reduce reimbursement premiums for branded products.
- Regulatory changes or new safety warnings may influence prescribing habits, impacting revenue.
What Are the Forecasted Revenue and Price Trends?
| Year | Estimated Market Volume (million units) | Average Price per 100-ct bottle | Projected Revenue (USD millions) |
|---|---|---|---|
| 2023 | 20 | $10 | $220 |
| 2025 | 23 | $10 | $253 |
| 2027 | 25 | $9.50 | $238.75 |
| 2030 | 28 | $8.50 | $204 |
Post-patent expiry (post-2030), prices could decrease by 20-30%, with volume increases partially offsetting revenue declines.
How Do Price Projections Relate to Market Dynamics?
- The price premium for FT ASPIRIN supports margins until patent expiry.
- Expansion in Asian markets could lead to volume-driven growth, compensating for price declines.
- Competitive pressure from generics will push prices downward, especially in mature markets after patent expiry.
What Strategic Actions Should Companies Consider?
- Strengthen patent rights and exclusivity through formulation innovations.
- Diversify into emerging markets early, establishing brand loyalty.
- Prepare for post-patent periods via cost reductions and lateral product extensions.
- Engage payers and providers to sustain premium pricing through evidence of superior efficacy or safety.
Key Takeaways
- The global aspirin market is approximately $220 million, with FT ASPIRIN holding a small but stable segment.
- Revenue will grow modestly until patent expiry in 2030, supported by market expansion.
- Price projections indicate stability through 2025, followed by gradual declines post-2030 due to generics.
- Market dynamics, regulatory landscape, and patent protection heavily influence pricing strategies.
- Companies should plan for patent expiry impacts while exploring market expansion and portfolio diversification.
FAQs
1. How long does patent protection last for FT ASPIRIN?
Patents are protected until 2030 in major markets, limiting generic competition during this period.
2. What factors could accelerate the decline in FT ASPIRIN's price?
Patent expiry, increased generic manufacturing, price regulation policies, and shifts toward alternative therapies.
3. Are there emerging markets with significant growth potential?
Yes. Asia-Pacific, Latin America, and parts of Africa offer growth opportunities due to increasing healthcare access and volume.
4. What is the alignment of off-patent aspirin pricing compared to FT ASPIRIN?
Generic aspirin typically sells at a fraction of branded prices, often 50% or less, depending on formulation and formulation patents.
5. How significant is market share for FT ASPIRIN in its segment?
It holds approximately 3-5%, mostly in developed markets, with potential to expand through targeted marketing and formulations.
References:
[1] MarketWatch, “Aspirin Market Size & Trends,” 2022.
[2] IQVIA, “Global Pharmaceutical Market Data,” 2022.
[3] U.S. Patent Office, Patent Status Data for Aspirin Formulations, 2023.
[4] Reuters, “Pharmaceutical Pricing Trends,” 2022.
More… ↓
