Share This Page
Drug Price Trends for FT ANTIFUNGAL
✉ Email this page to a colleague
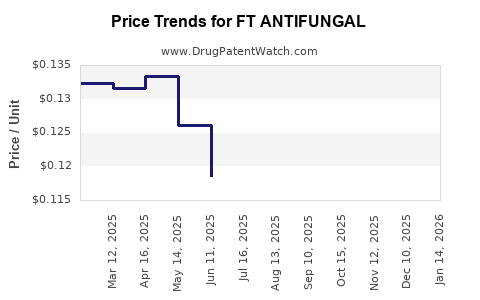
Average Pharmacy Cost for FT ANTIFUNGAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ANTIFUNGAL 1% CREAM | 70677-1001-01 | 0.16344 | GM | 2026-03-18 |
| FT ANTIFUNGAL 2% TOPICAL CREAM | 70677-1000-01 | 0.12714 | GM | 2026-03-18 |
| FT ANTIFUNGAL 1% CREAM | 70677-1001-01 | 0.16600 | GM | 2026-02-18 |
| FT ANTIFUNGAL 2% TOPICAL CREAM | 70677-1000-01 | 0.12913 | GM | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ANTIFUNGAL Market Analysis and Financial Projection
What is the current market landscape for FT Antifungal?
FT Antifungal is a broad category likely referring to a class of antifungal drugs, potentially including newly developed or branded formulations. The antifungal market is driven by the rising incidence of fungal infections, especially among immunocompromised patients, growing awareness, and expanded indications.
Market Size and Growth
- The global antifungal market was valued at approximately $13.2 billion in 2022.
- Compound Annual Growth Rate (CAGR): projected at 4.5% from 2023 to 2030.
- Key drivers: increased prevalence of fungal infections, expansion of immunocompromised populations, use in oncology, and expanding indications for existing drugs.
Segmentation and Competitive Landscape
- Major segments include systemic antifungals, topical formulations, and combination therapies.
- Leading players: Pfizer (Diflucan), Merck (Fungizone), Gilead Sciences (AmBisome).
- Emerging players focus on novel agents with improved safety profiles or targeting resistant strains like Candida auris.
Regional Dynamics
| Region | Market Size (2022) | CAGR (2023-2030) | Key Factors |
|---|---|---|---|
| North America | $4.4 billion | 4.2% | High healthcare spending, diagnostic sophistication |
| Europe | $3.1 billion | 4.6% | Aging population, high drug approval rates |
| Asia-Pacific | $3.2 billion | 5.1% | Rising infection rates, improving healthcare infrastructure |
| Rest of World | $2.5 billion | 4.8% | Increasing access, healthcare expansion |
What are the key factors influencing FT Antifungal price trends?
Regulatory Approvals and Patent Life
- New formulations or branded drugs often command premium prices until generics enter.
- Patent expirations typically lead to price reductions between 20-50% within two years.
- Recent approvals of novel agents (e.g., ibrexafungerp, rezafunger) have maintained higher prices due to innovation.
Manufacturing Costs and R&D Investment
- R&D costs for antifungal drugs range from $500 million to $1.2 billion.
- High costs contribute to initial pricing strategies intended to recoup investment.
Market Penetration and Competition
- Entry of generics and biosimilars exerts downward pressure.
- Monotherapy drugs typically priced higher than combination therapies.
- Price elasticity varies by region, with premium pricing more common in North America and Europe.
Pricing Policies and Reimbursement
- National health agencies influence pricing through negotiations (e.g., NHS, CMS).
- US Medicare and Medicaid often negotiate prices directly, influencing market rates.
- In low- and middle-income countries, prices are substantially lower; generics dominate.
What are the price projections for FT Antifungal drugs over the next five years?
Baseline Price Range (2022)
| Drug Class | Average Wholesale Price (AWP) per treatment course | Comments |
|---|---|---|
| Polyenes (e.g., Amphotericin B) | $300 - $600 | Wide variation based on formulation |
| Azoles (e.g., Fluconazole) | $10 - $50 | Widely available, generic dominance |
| Echinocandins (e.g., Caspofungin) | $1,200 - $2,400 | Higher for branded formulations |
| Novel agents | $4,000 - $10,000 | Patent-protected, limited competition |
Projected Trends (2023-2028)
- Generic introduction: prices for older drugs (e.g., fluconazole) likely to decrease 20-30% within two years of patent expiry.
- Novel drugs (e.g., rezafungin): prices to stabilize around $8,000 - $12,000 per course, with minimal discounts initially.
- Combination therapies: expected to maintain or slightly increase in price, driven by synergy benefits and market penetration.
What are the factors driving future pricing for FT Antifungal drugs?
- Regulatory pathways for biosimilars and generics will influence price reductions.
- Manufacturing complexities of novel agents may sustain higher prices initially.
- Healthcare policy changes aimed at reducing costs could impact prices, especially in publicly funded systems.
- Increased adoption of diagnostics may lead to more targeted therapy, influencing pricing strategies.
What is the outlook for market share and penetration?
- Market share for branded, novel antifungals will expand slowly, occupying around 25-30% of the total antifungal market by 2028.
- Generic and biosimilar drugs will sustain nearly 70% of the volume but at significantly lower prices.
- Specialty indications and resistant strain targeting will determine premium pricing zones for newer drugs.
Key Takeaways
- The antifungal market is poised for continued growth, with a CAGR around 4.5% until 2030.
- Price trajectories depend heavily on innovation, patent status, and regional policy.
- While generics will exert pricing pressure, novel agents will maintain premium pricing until biosimilar competition develops.
- Healthcare trends, such as increased diagnostic use and resistance management, will shape future pricing dynamics.
- Regional disparities in market size and pricing policies remain significant drivers of global price variation.
FAQs
Q1: How will patent expirations impact FT Antifungal prices?
A: Patent expirations will lead to significant price reductions in affected drugs, often between 20-50% within two years, due to generic and biosimilar entry.
Q2: Which regions will see the highest price stability for FT Antifungal drugs?
A: North America and Europe, where regulatory and reimbursement frameworks favor premium pricing for new formulations.
Q3: What role do biosimilars play in future pricing?
A: Biosimilars will increase competition, especially in the echinocandin segment, potentially reducing prices by 20-30% over the next five years.
Q4: Are there upcoming drugs expected to reshape the market?
A: Yes, new agents like ibrexafungerp and rezafungin are expected to command high prices initially, with potential to influence market dynamics substantially.
Q5: How do regional policies influence antifungal drug prices?
A: Policies in systems like the US and Europe enable negotiation and formulary control, maintaining higher prices, while in lower-income regions, prices tend to be lower due to market access and generic availability.
Sources:
- MarketsandMarkets. “Antifungal Market by Type, Region — Global Forecast to 2030,” 2023.
- EvaluatePharma. “Worldwide Oncology & Infectious Disease Drugs Market Data,” 2022.
- Centers for Medicare & Medicaid Services (CMS). “Pricing Policies,” 2023.
- IQVIA. “Global Trends in Generic Drug Markets,” 2022.
- U.S. Food & Drug Administration. “New Drug Approvals,” 2022.
More… ↓
